Constitutively active Stat5A and Stat5B promote adipogenesis
|
|
|
- Beverley Jennings
- 5 years ago
- Views:
Transcription
1 Environ Health Prev Med (2011) 16: DOI /s REGULAR ARTICLE Constitutively active Stat5A and Stat5B promote adipogenesis Hiroshi Wakao Rika Wakao Atsushi Oda Hiroyoshi Fujita Received: 13 September 2010 / Accepted: 1 November 2010 / Published online: 1 December 2010 Ó The Japanese Society for Hygiene 2010 Abstract Objective The metabolic syndrome is an important social problem affecting many people in developed countries. Obesity is a leading cause of this syndrome, hence understanding molecular mechanisms underlying obesity is of prime importance for preventive medicine to develop novel methods to alleviate the corresponding social cost as well as for pharmaceutical companies to develop antimetabolic drugs. Methods Since adipocytes play an important role in obesity, we explored the signaling pathways leading to differentiation of adipocytes. We used a preadipocyte cell line to monitor the differentiation of adipocytes, and virusmediated gene transfer to assess the role of the transcription factor Stat5 in adipogenesis. Adipocyte differentiation was assessed by Northern blot and Western blot analyses as well as accumulation of fat droplets in cells. Promoter activity of the proadipogenic transcription factor peroxisome proliferator-activated receptor-gamma (PPARc) was evaluated by luciferase assay. Results Virus-mediated gene transfer of the constitutively active form of both Stat5A and Stat5B resulted in enhanced adipocyte differentiation in the absence of fetal bovine serum (FBS) as judged by expression of proadipogenic factors as well as accumulation of fat droplets in cells. H. Wakao (&) A. Oda H. Fujita Department of Environmental Biology, School of Medicine, Hokkaido University, North 15 West 7, Kita-ku, Sapporo , Japan hwakao@med.hokudai.ac.jp R. Wakao Pharmaceutical and Medical Devices Agency, Kasumigaseki, Chiyoda-ku, Tokyo , Japan Such a proadipogenic effect of Stat5 is, in part, mediated by its ability to enhance transcription of PPARc, a master transcriptional regulator in adipogenesis. Conclusion The constitutively active form of Stat5A and Stat5B promoted adipocyte differentiation in the absence of FBS via induction of PPARc. Keywords Obesity Adipocytes Stat5 Signal transduction PPARc Introduction At present, more than 1.7 billion people are considered to be obese [1]. Obesity is caused by excess calorie intake and is a major worldwide health concern. The World Health Organization reports that at least 170 million people worldwide suffer from diabetes due to obesity. Obesity is characterized by dysregulated metabolism, dyslipidemia, insulin resistance, metabolic syndrome, nonalcoholic fatty liver disease, hyperglycemia, hypertension, some forms of cancer, and increased risk for development of type 2 diabetes (T2D) and its most serious comorbidity, cardiovascular disease. It is estimated that around 90% of T2D is due to obesity [2]. If not reversed, exacerbating obesity will lead to an epidemic of these comorbidities that will challenge many health care delivery systems. Thus, obesity poses a great challenge for public health. To date, many studies have shed light on obesogenic genes, transcriptional processes, and signaling pathways that control weight gain; however, we are far from understanding the whole process of obesity [3, 4]. Since excess adipocyte size and number are tightly linked to obesity, it is important to decipher the molecular mechanisms underlying such differentiation.
2 248 Environ Health Prev Med (2011) 16: Differentiation of preadipocyte cell lines such as 3T3L-1 and 3T3-F422A is stimulated in response to adipogenic hormone cocktails comprising insulin, glucocorticoids, cyclic adenosine monophosphate (camp)-producing factors, and fetal bovine serum (FBS). Such treatment allows growth-arrested preadipocytes to re-enter the cell cycle with another one or two cell cycles, which is known as mitotic clonal expansion. The transcription factor CCAAT/ enhancer binding proteins (C/EBP) b and d are induced during this period, and induction of these factors is a prerequisite for expression of PPARc, a master regulator of adipogenesis [3, 4]. PPARc in turn stimulates expression of C/EBPa. PPARc and C/EBPa promote adipogenesis by turning on adipocyte-specific genes such as ap2, leptin, and adiponectin. Terminally differentiated adipocytes harbor fat droplets within the cells, which is a hallmark of energystoring cells. PPARc is a nuclear hormone receptor whose agonists such as thiazolidinedione (TZD) troglitazone, and rosiglitazone are used as antidiabetic drugs, though the natural ligand in the body has yet to be determined. It is noteworthy that PPARc ligands are required for efficient adipogenesis of fibroblastic cells [5]. The role of glucocorticoids and camp-producing factors in adipogenesis is considered to be induction of C/EBPd and b, respectively, while that of insulin is believed to be cross-activation of insulin-like growth factor (IGF)-1 receptor. In contrast, the role of FBS in adipogenesis is more ambiguous, although FBS is essential for adipogenesis. It is proposed that IGF-1 in FBS is critical for adipogenesis; however, this remains to be formally proven. Stat family proteins Stat1, 2, 3, 4, 5A, 5B, and 6 are activated by many cytokines and play crucial roles in cell proliferation and differentiation [6]. That the size of the fat pad from Stat5A and Stat5B double-knockout mice is onefifth of that from control mice suggests that Stat5 plays a role in adipogenesis [7]. Concurrently we have shown that Stat5 is activated by FBS and adipogenic hormones, and conveys the extracellular stimulus to the nucleus, culminating in execution of the adipogenic program in 3T3-L1 cells. Activation of Stat5 tightly correlates with expression of PPARc2, an isoform of PPARc exclusively expressed in adipocytes [8]. In the present report, we show that Stat5 regulates murine PPARc2 promoter activity. Furthermore, we demonstrate that ectopic expression of the constitutively active form of Stat5A or Stat5B promotes expression of PPARc2 and adipogenesis in the absence of FBS. These data reveal a novel facet of transcriptional cascades in adipogenesis and highlight the role of Stat5 in fat cell differentiation. Materials and methods Retrovirus-mediated gene transduction in 3T3-L1 cells Retroviral vectors were constructed using plncx2 (Clontech). plncx2 harboring the constitutively active form of murine Stat5A (Stat5A*) and Stat5B (Stat5B*) [9] were generated using the Quick change mutation kit (Stratagene), and the mutated DNA sequence was verified (ABI, 310 sequencer). These Stat5s reside in the nucleus and show constitutive transcriptional activity in the absence of ligands. Recombinant retroviruses were produced using Ecopack2 packaging cells and used for transduction (Clontech). Neomycin-resistant 3T3-L1 cells were selected after 2 weeks. These cells were grown to confluence and subjected to adipogenic regimen. Cell culture and induction of differentiation 3T3-L1 cells were maintained in growth medium consisting of Dulbecco s modified Eagle s medium (DMEM) containing 10% normal calf serum (CS). To differentiate 3T3-L1 cells, they were grown to confluence and left for 2 days. Cells were then exposed to fresh differentiation medium consisting of DMEM, 10% CS, 1 lm dexamethasone (DEX), 0.5 mm methylisobutylxanthine (MIX), and 10 lg/ml insulin for 48 h. Thereafter, cells were refed every other day with DMEM containing 10% CS and 2.5 lg/ml insulin. All experiments were done with CS, except the transcriptional assay as shown in Fig. 1b, c, wherein FBS was used instead of CS. FBS contains some factors that activate Stat5. Transient transfection assay Murine PPARc2 promoter comprising?1 to-1047 or?1 to (devoid of Stat5 binding site) was cloned from the genomic DNA of C57BL/6 mice by polymerase chain reaction (PCR) with Pfu-turbo DNA polymerase (Stratagene) and ligated to pgl2-basic luciferase reporter gene (Clontech). PPARc2 promoter harboring a mutated Stat5 site (5 0 -TTCAGGGAA-3 0 to 5 0 -GGCAGGTAA-3 0 ) was created by PCR. Transfection of reporter gene was performed with Lipofectamine-plus (Invitrogen) together with Stat5A, Stat5B, Stat5A* or Stat5B* complementary DNA (cdna) in the presence of cdna for renilla luciferase as internal control. After transfection, both firefly and renilla luciferase activities were measured, and relative luciferase activity was calculated.
3 Environ Health Prev Med (2011) 16: RNA preparation Total RNA was prepared with Trizol solution (Invitrogen) at the indicated time points and subjected to Northern blot analysis with digoxigenin (DIG)-labeled antisense RNA probes as described [8]. Western blot analysis Total cell lysates from 3T3-L1 cells were prepared 10 days after differentiation. Proteins (50 lg) were separated on a sodium dodecyl sulfate (SDS) polyacrylamide gel and transferred to polyvinylidene fluoride (PVDF) membrane. Proteins were visualized by enhanced chemiluminescence (ECL) system (GE Biosciences). Anti-Stat5 (C-17), anti- PPARc (H-100), and anti-c/ebpa (14AA) antibodies were purchased (Santa Cruz Biotechnology). Results Fig. 1 Stat5 activates the PPARc2 promoter. a The sequence of murine and human PPARc2 gene promoter was analyzed, and a potential Stat5 binding site in the promoters is indicated by the closed box (upper panel). Also is indicated a consensus Stat5 binding site. Schematic representation of the reporter gene constructs containing a wild-type Stat5 binding site (wild type), mutated Stat5 binding site (mutated nucleotides are underlined) or no Stat5 binding site (deleted) (lower panel). b Stat5 transactivates PPARc2 promoter via Stat5- responsive element. Relative luciferase activity (firefly luciferase activity/renilla luciferase activity) was determined with the reporter constructs containing a Stat5 binding site, a mutated Stat5 binding site or no Stat5 binding site. After serum starvation, cells were left unstimulated or stimulated with FBS for 6 h. Results are expressed as fold induction and represent the mean ± standard deviation of three independent experiments. c Constitutively active Stat5A and Stat5B activate the PPARc2 promoter in an FBS-independent manner. 3T3- L1 cells were transfected with the PPARc2 reporter construct containing a Stat5 binding site together with wild-type or constitutively active Stat5 complementary DNA (cdna) (Stat5A* or Stat5B*). After starvation, cells were left unstimulated or stimulated with 10% FBS for 6 h. Results are from three independent experiments performed in triplicates We have previously shown that Stat5 overexpression promotes PPARc2 expression, while ectopic expression of dominant negative Stat5 compromises it at both messenger RNA (mrna) and protein levels [8]. To gain insight into the molecular mechanism underlying PPARc2 mrna expression, we have aligned a putative promoter region of murine and human PPARc2 that is specific for adipocytes [10]. Sequence comparison revealed that they exhibited high similarity; in particular, several C/EBP binding sites known to be important for PPARc2 promoter activity were well conserved [11]. Intriguingly, sequences of mouse promoter comprising between and and the corresponding human region contained a putative Stat5 binding site (TTCNNNGAA, N: any nucleotide) [12] (Fig. 1a, upper panel). These data indicate that the Stat5 binding site might be important for PPARc2 promoter activity. We explored this by measuring the murine PPARc2 promoter-luciferase reporter activity. Reporter gene construct containing a putative Stat5 binding site was transfected into 3T3-L1 cells together with Stat5A or Stat5B cdna. After transfection, cells were left unstimulated or stimulated with FBS, and relative luciferase activity was measured. In the absence of FBS, luciferase activity was barely affected by exogenous Stat5A or Stat5B, but a certain level of basal activity was observed. This was most likely due to endogenous Stat5s or other Stat family proteins (Fig. 1b, none). While stimulation with FBS augmented luciferase activity, ectopic expression of Stat5A or Stat5B further increased luciferase activity by 2.5- or
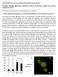
4 250 Environ Health Prev Med (2011) 16: fold, respectively (Fig. 1b, wild type, FBS). In sharp contrast, mutation in the putative Stat5 binding site severely compromised activity in response to FBS (Fig. 1b, mutated). Furthermore, ablation of the putative Stat5 binding site virtually abolished promoter activity, regardless of stimulation (Fig. 1b, deleted). These results indicate that Stat5 controls, at least partially, the activity of the murine PPARc2 promoter, and is responsible for mediating FBS-induced expression of PPARc2. Several reports have shown that FBS is an essential factor for adipogenesis, although its effector molecules in FBS have not yet been identified. Our previous data suggest that FBS plays a role in inducing PPARc2 in 3T3-L1 as well as NIH3T3 cells [13]. Since FBS enhanced PPARc2 promoter activity via Stat5 binding site (Fig. 1b), we hypothesized that constitutively active Stat5s might obviate the FBS dependence of 3T3-L1 cells in adipocyte differentiation. To explore this, we evaluated the effect of constitutively active Stat5 (hereafter designated Stat5A* or Stat5B*) on PPARc2 promoter activity. Intriguingly, introduction of Stat5A* and to a lesser extent Stat5B* resulted in further enhanced activity. In both cases, however, there was little difference in transcriptional activity regardless of stimulation by FBS (Fig. 1c). These results indicate that constitutively active Stat5 substitutes the function of FBS for inducing PPARc2 in 3T3-L1 cells. We then examined whether ectopic expression of Stat5A* or Stat5B* culminated in promotion of 3T3-L1 cell adipogenesis in the absence of FBS. To this end, Stat5A* or Stat5B* was introduced into 3T3-L1 cells by retrovirus. After transduction, cells were induced to differentiate in the presence of MIX, DEX, and insulin with CS. As a control, cells harboring the empty vector were subjected to an identical regimen. The effects of constitutively active Stat5 were assessed by Northern blot analysis for transcription factors relevant to adipogenesis, such as C/EBPb, d, and a, PPARc, and SREBP1c (Fig. 2a). Both Stat5A* and Stat5B* augmented C/EBPb mrna, while they had little impact on C/EBPd mrna during adipogenesis (Fig. 2a). As for C/EBPa mrna, neither Stat5A* nor Stat5B* showed any significant effect until day 4. However, both Stat5s maintained the high level of the transcript up to day 10, whereas the empty vector failed to do so. Stat5A* and Stat5B* led to an increment of SREBP1c mrna over the control, whilst the former exhibited slightly stronger effects than the latter. The impact of constitutively active Stat5 expression was most pronounced in PPARc2 mrna. Whereas the empty vector alone barely induced PPARc2 mrna, ectopic expression of Stat5A* and Stat5B* markedly stimulated it from day 4 to 10 without any decline (Fig. 2a). Together, these results demonstrated that constitutively active Stat5A and Stat5B enhanced mrna expression for proadipogenic factors such Fig. 2 Constitutively active Stat5A and Stat5B promote expression of proadipogenic factors. a Ectopic expression of constitutively active Stat5s induces proadipogenic transcription factors. 3T3-L1 cells were infected with control retrovirus vector (plncx2), vector harboring constitutively active Stat5A (Stat5A*) or Stat5B (Stat5B*). After G418 selection, cells were grown to confluence and subjected to the adipogenic regimen as described in the Materials and methods section. Total RNA was extracted at the indicated time, and 2 lg of each sample was subjected to Northern blot analysis with the antisense riboprobes corresponding to the genes. Three independent experiments were performed, and essentially identical results were obtained. b Induction of adipogenic marker genes in cells transduced with constitutively active Stat5s. Total RNA was isolated at 10 days post differentiation induction and subjected to Northern blot analysis with DIG-labeled riboprobes. Lane 1 cells transduced with the empty vector, lane 2 cells harboring Stat5A*, lane 3 cells harboring Stat5B*. c Increase of C/EBPa and PPARc proteins upon Stat5 transduction. Cells harboring empty vector, Stat5A* or Stat5B* were subjected to the adipogenic regimen. Ten days later, cells were harvested and cell lysates were prepared. After separation on SDS gel, the proteins were visualized by the indicated antibodies using an ECL system. For Stat5, position of Stat5A and Stat5B is depicted by arrow. Lane 1 cells transduced with the empty vector, lanes 2 and 3 cells harboring Stat5A* or Stat5B*, respectively as C/EBPb, SREBP1c, and PPARc2 in the absence of FBS in 3T3-L1 cells. We then investigated whether such an increase in proadipogenic factors led to adipogenesis by biochemical and morphological criteria. Several terminal markers for adipocytes were assessed by Northern blot analysis at the end of differentiation (Fig. 2b). Phosphoenolpyruvate carboxykinase (PEPCK) mrna was slightly but significantly elevated in cells harboring Stat5A* or Stat5B* over the control (Fig. 2b). Stat5A* more strongly induced mrna
5 Environ Health Prev Med (2011) 16: for ap2, adiponectin, adipsin, leptin, and glucose transporter 4 (Glut4) than did Stat5B* (Fig. 2b, lanes 1 3). It is worth noting that preferential activation of adipsin and Glut4 was also observed in NIH3T3 cells harboring Stat5A [14]. Nevertheless, such a biased increment of the transcript for ap2 and PPARc2 in NIH3T3 cells was not evident in 3T3-L1 cells harboring Stat5A* (Fig. 2a, b). Since PPARc2 and C/EBPa are critical factors for adipogenesis, the correlation between ectopic expression of Stat5 and resulting induction of such factors was evaluated by Western blot analysis (Fig. 2c). Ectopic expression of Stat5A* or Stat5B* proteins was evident (Fig. 2c, lanes 1 3). While empty vector alone led to a low level expression of PPARc2 and C/EBPa, ectopic expression of Stat5A* or Stat5B* significantly increased these proteins. Stat5A* was more potent than Stat5B* for inducing C/EBPa (Fig. 2c, lanes 1 3). These results mirrored the strong activity of Stat5A* in inducing mrna for ap2, adiponectin, adipsin, leptin, and Glut4. Finally, adipogenesis was assessed by Oil-Red-O staining, which detects neutral lipids. While little staining was observed in cells transduced with the empty vector, cells harboring Stat5A* or Stat5B* showed substantial staining and rounded-up morphology (Fig. 3, compare 1 3). These results were in accordance with the simultaneous augmentation of genes relevant to mature adipocytes such as PPARc, adiponectin, ap2, and PEPCK (Fig. 2a, b). Discussion Adipogenic hormones MIX, a camp inducer, and DEX are responsible for transcriptional induction of C/EBPb and Fig. 3 Impact of constitutively active Stat5 on triglyceride loading (Oil-Red-O staining of neutral lipids). 3T3-L1 cells were differentiated as described in Fig. 2. After 10 days, cells were stained with Oil- Red-O to visualize triglyceride vesicles. Upper panel microscopic view, lower panel macroscopic view. Lane 1 cells transduced with the empty vector, lanes 2 and 3 cells harboring Stat5A* or Stat5B*, respectively C/EBPd, respectively, at the early phase of adipocyte differentiation [15]. Ectopic expression of these factors leads to adipogenic conversion of NIH3T3 or 3T3-L1 cells by inducing PPARc, which then results in execution of adipogenesis [16, 17]. Conversely, ablation of these factors severely compromised adipocyte differentiation [3]. Furthermore, C/EBPb and d expression is a prerequisite for C/EBPa induction [17]. Intriguingly, PPARc turns on expression of C/EBPa. C/EBPa itself can bind to the C/EBP site in the PPARc promoter, providing a stable, positive forward regulatory loop [5]. In addition to the above classical C/EBP PPARc axis, several lines of evidence have suggested that Stat5 may play an important role in adipogenesis. Indeed, Stat5 is rapidly activated upon adipogenic hormone challenge in 3T3-L1 preadipocytes. Furthermore, overexpression of Stat5 results in promotion of adipogenesis, whilst expression of dominant negative Stat5 leads to inhibition of adipocyte differentiation. Intriguingly, the level of Stat5A and Stat5B protein increases as cells differentiate into adipocytes [8, 18]. Furthermore, FBS is a prerequisite for efficient induction of PPARc mrna in NIH3T3 as well as 3T3-L1 cells [13]. Thus, it is most likely that the requirement for FBS for efficient adipogenesis correlates with the activity of Stat5. In line with this hypothesis, prolactin and growth hormone, both of which activate Stat5, are shown to promote adipogenesis in the absence/presence of FBS in 3T3-L1 and 3T3-F422A cells, respectively [19 21]. In this regard, the weak induction of PPARc and C/EBPa in adipogenesis at both protein and mrna levels most likely reflects the fact that CS fails to activate Stat5s strongly with the control vector (Fig. 2a, c). Although prolactin and growth hormone are components of FBS, it is difficult to evaluate the relative contribution of these hormones to adipogenesis, as identification of the factor(s) responsible for Stat5 activation in FBS has yet to be achieved. Intriguingly, it is reported that only Stat5A but not Stat5B can confer the adipogenic potential to the cells [14]. In this regard, our present data indicating that constitutively active Stat5A as well as Stat5B enhance expression of PPARc2, and to a lesser extent C/EBPb, and promote adipogenesis without affecting C/EBPd expression shed light on a novel adipogenic transcriptional cascade. Though both Stat5A and Stat5B appear to control PPARc2 expression by interacting with its promoter, it is yet to be examined how these Stat5s impinge upon C/EBPb expression. The difference between Stat5A and Stat5B in terms of adipogenesis-enhancing potential indicates that they function differently in fat cell development (Fig. 2b). Our observation that Stat5A is more potent in inducing adipocyte genes such as PEPCK, ap2, adiponectin, adipsin, leptin, and GLUT4 is in accordance with the previous report that Stat5A can promote adipogenesis [14]. Since
6 252 Environ Health Prev Med (2011) 16: both Stat5A and Stat5B possess similar potential in inducing PPARc2 and adipocyte differentiation as judged by Western blot analysis and Oil-Red-O staining (Figs. 2c, 3), the target genes for each Stat5, which may be responsible for optimal adipogenesis, are yet to be delineated. Identification of such genes will shed light on the molecular mechanism underlying the different adipogenic potential between Stat5A and Stat5B and opens a novel avenue for increasing our understanding of adipogenesis, which ultimately may culminate in the development of antiobesity drugs. Conclusions Both constitutively active Stat5A and Stat5B could functionally bypass the FBS requirement for adipogenesis in 3T3-L1 cells. These Stat5s induced PPARc, a master regulator in adipogenesis, thus culminating execution of the adipogenic program. Our present data suggest that compounds that stimulate the Stat5 PPARc axis in adipocytes may be beneficial for promoting adipogenesis, which in turn increases fat storage in the body and alleviates metabolic syndrome [22]. Thus, the search for such compounds may open the door for future preventive medicines and contribute to improved social welfare. Acknowledgments the work. Conflict of interest References The authors thank Helix Institute for supporting No conflict of interest. 1. Haslam DW, James WP. Obesity. Lancet. 2005;366: Hossain P, Kawar B, El Nahas M. Obesity and diabetes in the developing world a growing challenge. N Engl J Med. 2007; 356: Rosen ED, Spiegelman BM. Molecular regulation of adipogenesis. Annu Rev Cell Dev Biol. 2000;16: Rosen ED, Spiegelman BM. Adipocytes as regulators of energy balance and glucose homeostasis. Nature. 2006;444: Tontonoz P, Spiegelman BM. Fat and beyond: the diverse biology of PPARgamma. Annu Rev Biochem. 2008;77: Darnell JE Jr. STATs and gene regulation. Science. 1997;277: Teglund S, McKay C, Schuetz E, van Deursen JM, Stravopodis D, Wang D, et al. Stat5a and Stat5b proteins have essential and nonessential, or redundant, roles in cytokine responses. Cell. 1998;93: Nanbu-Wakao R, Morikawa Y, Matsumura I, Masuho Y, Muramatsu MA, Senba E, et al. Stimulation of 3T3-L1 adipogenesis by signal transducer and activator of transcription 5. Mol Endocrinol. 2002;16: Onishi M, Nosaka T, Misawa K, Mui AL, Gorman D, McMahon M, et al. Identification and characterization of a constitutively active STAT5 mutant that promotes cell proliferation. Mol Cell Biol. 1998;18: Tontonoz P, Hu E, Graves RA, Budavari AI, Spiegelman BM. mppar gamma 2: tissue-specific regulator of an adipocyte enhancer. Genes Dev. 1994;8: Shi XM, Blair HC, Yang X, McDonald JM, Cao X. Tandem repeat of C/EBP binding sites mediates PPARgamma2 gene transcription in glucocorticoid-induced adipocyte differentiation. J Cell Biochem. 2000;76: Gouilleux F, Wakao H, Mundt M, Groner B. Prolactin induces phosphorylation of Tyr694 of Stat5 (MGF), a prerequisite for DNA binding and induction of transcription. EMBO J. 1994;13: Nanbu-Wakao R, Fujitani Y, Masuho Y, Muramatu M, Wakao H. Prolactin enhances CCAAT enhancer-binding protein-beta (C/ EBP beta) and peroxisome proliferator-activated receptor gamma (PPAR gamma) messenger RNA expression and stimulates adipogenic conversion of NIH-3T3 cells. Mol Endocrinol. 2000;14: Floyd ZE, Stephens JM. STAT5A promotes adipogenesis in nonprecursor cells and associates with the glucocorticoid receptor during adipocyte differentiation. Diabetes. 2003;52: Cao Z, Umek RM, McKnight SL. Regulated expression of three C/EBP isoforms during adipose conversion of 3T3-L1 cells. Genes Dev. 1991;5: Wu Z, Xie Y, Bucher NL, Farmer SR. Conditional ectopic expression of C/EBP beta in NIH-3T3 cells induces PPAR gamma and stimulates adipogenesis. Genes Dev. 1995;9: Yeh WC, Cao Z, Classon M, McKnight SL. Cascade regulation of terminal adipocyte differentiation by three members of the C/EBP family of leucine zipper proteins. Genes Dev. 1995;9: Stephens JM, Morrison RF, Pilch PF. The expression and regulation of STATs during 3T3-L1 adipocyte differentiation. J Biol Chem. 1996;271: Shang CA, Waters MJ. Constitutively active signal transducer and activator of transcription 5 can replace the requirement for growth hormone in adipogenesis of 3T3-F442A preadipocytes. Mol Endocrinol. 2003;17: Stewart WC, Baugh JE Jr, Floyd ZE, Stephens JM. STAT 5 activators can replace the requirement of FBS in the adipogenesis of 3T3-L1 cells. Biochem Biophys Res Commun. 2004;324: Kawai M, Namba N, Mushiake S, Etani Y, Nishimura R, Makishima M, et al. Growth hormone stimulates adipogenesis of 3T3- L1 cells through activation of the Stat5A/5B-PPARgamma pathway. J Mol Endocrinol. 2007;38: Sugii S, Olson P, Sears DD, Saberi M, Atkins AR, Barish GD, et al. PPARgamma activation in adipocytes is sufficient for systemic insulin sensitization. Proc Natl Acad Sci USA. 2009;106:
ab Adipogenesis Assay Kit (Cell-Based)
 ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
MBB317. Dr D MANGNALL OBESITY. Lecture 2
 MBB317 Dr D MANGNALL OBESITY Lecture 2 When the structure of the insulin receptor was first discovered it was assumed that the active beta subunit tyrosine kinase would phosphorylate some intracellular
MBB317 Dr D MANGNALL OBESITY Lecture 2 When the structure of the insulin receptor was first discovered it was assumed that the active beta subunit tyrosine kinase would phosphorylate some intracellular
SUPPLEMENTARY INFORMATION
 -. -. SUPPLEMENTARY INFORMATION DOI: 1.1/ncb86 a WAT-1 WAT- BAT-1 BAT- sk-muscle-1 sk-muscle- mir-133b mir-133a mir-6 mir-378 mir-1 mir-85 mir-378 mir-6a mir-18 mir-133a mir- mir- mir-341 mir-196a mir-17
-. -. SUPPLEMENTARY INFORMATION DOI: 1.1/ncb86 a WAT-1 WAT- BAT-1 BAT- sk-muscle-1 sk-muscle- mir-133b mir-133a mir-6 mir-378 mir-1 mir-85 mir-378 mir-6a mir-18 mir-133a mir- mir- mir-341 mir-196a mir-17
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Consecutive Positive Feedback Loops Create a Bistable Switch that Controls Preadipocyte-to-Adipocyte Conversion
 Cell Reports Article Consecutive Positive Feedback Loops Create a Bistable Switch that Controls Preadipocyte-to-Adipocyte Conversion Byung Ouk Park, 1 Robert Ahrends, 1 and Mary N. Teruel 1, * 1 Department
Cell Reports Article Consecutive Positive Feedback Loops Create a Bistable Switch that Controls Preadipocyte-to-Adipocyte Conversion Byung Ouk Park, 1 Robert Ahrends, 1 and Mary N. Teruel 1, * 1 Department
Endocrine Disrupters as Obesogens
 Endocrine Disrupters as Obesogens Is the environment making us fat? Bruce Blumberg, Ph.D. Department of Developmental and Cell Biology Department of Pharmaceutical Sciences Developmental Biology Center
Endocrine Disrupters as Obesogens Is the environment making us fat? Bruce Blumberg, Ph.D. Department of Developmental and Cell Biology Department of Pharmaceutical Sciences Developmental Biology Center
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent Protein.
 prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Metabolic Solutions Development Company, Kalamazoo, USA.
 New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
ENHANCEMENT OF BROWN ADIPOSE TISSUE DEVELOPMENT IN VIVO BY A NOVEL INSULIN SENSITIZER
 ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
In The Name Of God. In The Name Of. EMRI Modeling Group
 In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
Resistin overexpression impaired glucose tolerance in hepatocytes
 Eur. Cytokine Netw., Vol. 17 n 3, September 26, 189-95 189 Resistin overexpression impaired glucose tolerance in hepatocytes Lei Zhou, Yixing Li, Tao Xia, Shengqiu Feng, Xiaodong Chen, Zaiqing Yang Key
Eur. Cytokine Netw., Vol. 17 n 3, September 26, 189-95 189 Resistin overexpression impaired glucose tolerance in hepatocytes Lei Zhou, Yixing Li, Tao Xia, Shengqiu Feng, Xiaodong Chen, Zaiqing Yang Key
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
Supplemental Material:
 Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
A novel 3T3-L1 preadipocyte variant that expresses PPAR 2 and RXR but does not undergo differentiation
 A novel 3T3-L1 preadipocyte variant that expresses PPAR 2 and RXR but does not undergo differentiation Rebecca A. Baillie, Xiaoming Sha, Philippe Thuillier, and Steven D. Clarke 1 Institute for Cellular
A novel 3T3-L1 preadipocyte variant that expresses PPAR 2 and RXR but does not undergo differentiation Rebecca A. Baillie, Xiaoming Sha, Philippe Thuillier, and Steven D. Clarke 1 Institute for Cellular
PEPCK. The Regulation of Eukaryotic Gene Expression. Why choose PEPCK? PEPCK. PEPCK overexpression in muscle. The Supermouse.
 PEPK The Regulation of Eukaryotic Gene Expression..using the example of PEPK This is an acronym for an enzyme PhosphoEnol Pyruvate arboxykinase This enzyme is NLY regulated by gene expression! No allosteric
PEPK The Regulation of Eukaryotic Gene Expression..using the example of PEPK This is an acronym for an enzyme PhosphoEnol Pyruvate arboxykinase This enzyme is NLY regulated by gene expression! No allosteric
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
ACTH Enhances Lipid Accumulation in Bone-marrow derived Mesenchymal stem cells undergoing adipogenesis
 ACTH Enhances Lipid Accumulation in Bone-marrow derived Mesenchymal stem cells undergoing adipogenesis Thomas Rhodes a, Michelle Pazienza a and Jodi F. Evans a ACTH is a major hormone of the stress axis
ACTH Enhances Lipid Accumulation in Bone-marrow derived Mesenchymal stem cells undergoing adipogenesis Thomas Rhodes a, Michelle Pazienza a and Jodi F. Evans a ACTH is a major hormone of the stress axis
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
Project Summary. Funded by The Beef Checkoff. Regulation of Marbling Development in Beef Cattle by Specific Fatty Acids
 Project Summary Regulation of Marbling Development in Beef Cattle by Specific Fatty Acids Principal Investigators: S. Smith 1, B. Johnson 2 and M. Doumit 3 Texas A&M University 1, Texas Tech University
Project Summary Regulation of Marbling Development in Beef Cattle by Specific Fatty Acids Principal Investigators: S. Smith 1, B. Johnson 2 and M. Doumit 3 Texas A&M University 1, Texas Tech University
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Metabolic Syndrome. DOPE amines COGS 163
 Metabolic Syndrome DOPE amines COGS 163 Overview - M etabolic Syndrome - General definition and criteria - Importance of diagnosis - Glucose Homeostasis - Type 2 Diabetes Mellitus - Insulin Resistance
Metabolic Syndrome DOPE amines COGS 163 Overview - M etabolic Syndrome - General definition and criteria - Importance of diagnosis - Glucose Homeostasis - Type 2 Diabetes Mellitus - Insulin Resistance
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Brief Critical Review
 Brief Critical Review May 2007: 251 256 Serum Retinol-Binding Protein: A Link Between Obesity, Insulin Resistance, and Type 2 Diabetes George Wolf, DPhil Insulin resistance occurs under conditions of obesity,
Brief Critical Review May 2007: 251 256 Serum Retinol-Binding Protein: A Link Between Obesity, Insulin Resistance, and Type 2 Diabetes George Wolf, DPhil Insulin resistance occurs under conditions of obesity,
Akt-dependent Skp2 mrna translation is required for exiting contact inhibition, oncogenesis, and adipogenesis
 The EMBO Journal (2012) 31, 1134 1146 & 2012 European Molecular Biology Organization All Rights Reserved 0261-4189/12 www.embojournal.org Akt-dependent Skp2 mrna translation is required for exiting contact
The EMBO Journal (2012) 31, 1134 1146 & 2012 European Molecular Biology Organization All Rights Reserved 0261-4189/12 www.embojournal.org Akt-dependent Skp2 mrna translation is required for exiting contact
UNDERSTANDING THE BIOLOGY OF FAT METABOLISM is fundamental
 Image-Based High-Throughput Quantification of Cellular Fat Accumulation MIKE DRAGUNOW, 1,3 RACHEL CAMERON, 1 PRITIKA NARAYAN, 1,3 and SIMON O CARROLL 2 A number of biochemical methods are available for
Image-Based High-Throughput Quantification of Cellular Fat Accumulation MIKE DRAGUNOW, 1,3 RACHEL CAMERON, 1 PRITIKA NARAYAN, 1,3 and SIMON O CARROLL 2 A number of biochemical methods are available for
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
Supplementary Information
 Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
ADD1 SREBP1 activates PPAR through the production of endogenous ligand (fatty acid metabolism nuclear hormone receptor adipocyte differentiation)
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 4333 4337, April 1998 Cell Biology ADD1 SREBP1 activates PPAR through the production of endogenous ligand (fatty acid metabolism nuclear hormone receptor adipocyte
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 4333 4337, April 1998 Cell Biology ADD1 SREBP1 activates PPAR through the production of endogenous ligand (fatty acid metabolism nuclear hormone receptor adipocyte
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice
 Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway
 Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway Hui H. Zhang 1, Jingxiang Huang 1, Katrin Düvel 1, Bernard Boback 1, Shulin Wu 2, Rachel M. Squillace 3, Chin-Lee Wu 2, Brendan D. Manning
Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway Hui H. Zhang 1, Jingxiang Huang 1, Katrin Düvel 1, Bernard Boback 1, Shulin Wu 2, Rachel M. Squillace 3, Chin-Lee Wu 2, Brendan D. Manning
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Adipose Tissue Expandability in the Maintenance of Metabolic Homeostasis Sarah L. Gray, PhD, and Antonio J. Vidal-Puig, MD, PhD
 June 2007(II): S7 S12 Adipose Tissue Expandability in the Maintenance of Metabolic Homeostasis Sarah L. Gray, PhD, and Antonio J. Vidal-Puig, MD, PhD Adipose tissue expands to accommodate increased lipid
June 2007(II): S7 S12 Adipose Tissue Expandability in the Maintenance of Metabolic Homeostasis Sarah L. Gray, PhD, and Antonio J. Vidal-Puig, MD, PhD Adipose tissue expands to accommodate increased lipid
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS
 FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Chairoungdua et al., http://www.jcb.org/cgi/content/full/jcb.201002049/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Expression of CD9 and CD82 inhibits Wnt/ -catenin
Supplemental material Chairoungdua et al., http://www.jcb.org/cgi/content/full/jcb.201002049/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Expression of CD9 and CD82 inhibits Wnt/ -catenin
Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation
 Chemistry Volume 2015, Article ID 171570, 4 pages http://dx.doi.org/10.1155/2015/171570 Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation Jin Taek Hwang,
Chemistry Volume 2015, Article ID 171570, 4 pages http://dx.doi.org/10.1155/2015/171570 Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation Jin Taek Hwang,
LPA is a highly bioactive phospholipid which acid receptor signaling axis. (Harald M.
 Progress Report Description of activities: 1. mtor signaling in kidney cell with silenced HNF1B 2. Differentiation of 3T3 cells into adipose cells 3. Silencing of HNF1B gene in adipose cells and disturbing
Progress Report Description of activities: 1. mtor signaling in kidney cell with silenced HNF1B 2. Differentiation of 3T3 cells into adipose cells 3. Silencing of HNF1B gene in adipose cells and disturbing
Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene
 Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene YUELIN ZHANG, WEIHUA FAN, MARK KINKEMA, XIN LI, AND
Interaction of NPR1 with basic leucine zipper protein transcription factors that bind sequences required for salicylic acid induction of the PR-1 gene YUELIN ZHANG, WEIHUA FAN, MARK KINKEMA, XIN LI, AND
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
The expression of cardiotrophin-1 is differentially regulated in murine and human obesity type II diabetes
 Louisiana State University LSU Digital Commons LSU Master's Theses Graduate School 2011 The expression of cardiotrophin-1 is differentially regulated in murine and human obesity type II diabetes Kelesha
Louisiana State University LSU Digital Commons LSU Master's Theses Graduate School 2011 The expression of cardiotrophin-1 is differentially regulated in murine and human obesity type II diabetes Kelesha
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1 Schematic depiction of the tandem Fc GDF15. Supplementary Figure 2 Supplementary Figure 2 Gfral mrna levels in the brains of both wild-type and knockout Gfral
Supplementary Figure 1 Supplementary Figure 1 Schematic depiction of the tandem Fc GDF15. Supplementary Figure 2 Supplementary Figure 2 Gfral mrna levels in the brains of both wild-type and knockout Gfral
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein
 Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
PAX8-PPARγ Fusion Protein in thyroid carcinoma
 Sidney H. Ingbar Lecture, ATA 10/17/2013: PAX8-PPARγ Fusion Protein in thyroid carcinoma Ronald J. Koenig, MD, PhD Division of Metabolism, Endocrinology & Diabetes University of Michigan Learning Objectives
Sidney H. Ingbar Lecture, ATA 10/17/2013: PAX8-PPARγ Fusion Protein in thyroid carcinoma Ronald J. Koenig, MD, PhD Division of Metabolism, Endocrinology & Diabetes University of Michigan Learning Objectives
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments:
 Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
Supplementary Information
 Supplementary Information Notch deficiency decreases hepatic lipid accumulation by induction of fatty acid oxidation No-Joon Song,#, Ui Jeong Yun,#, Sunghee Yang, Chunyan Wu, Cho-Rong Seo, A-Ryeong Gwon,,
Supplementary Information Notch deficiency decreases hepatic lipid accumulation by induction of fatty acid oxidation No-Joon Song,#, Ui Jeong Yun,#, Sunghee Yang, Chunyan Wu, Cho-Rong Seo, A-Ryeong Gwon,,
People Living with And Inspired by Diabetes
 People Living with And Inspired by Diabetes IMPERFECTLY MOVING TOWARDS & FAINTING GOATS Vol. 2 No. 2 Fall 2016 Vol. 2 No. 2 Fall 2016 1 Review PARTNERING WITH SCIENCE: New Hope for the Effective Treatment
People Living with And Inspired by Diabetes IMPERFECTLY MOVING TOWARDS & FAINTING GOATS Vol. 2 No. 2 Fall 2016 Vol. 2 No. 2 Fall 2016 1 Review PARTNERING WITH SCIENCE: New Hope for the Effective Treatment
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Data Sheet. Notch Pathway Reporter Kit Catalog # 60509
 Data Sheet Notch Pathway Reporter Kit Catalog # 60509 6042 Cornerstone Court W, Ste B Background The Notch signaling pathway controls cell fate decisions in vertebrate and invertebrate tissues. NOTCH signaling
Data Sheet Notch Pathway Reporter Kit Catalog # 60509 6042 Cornerstone Court W, Ste B Background The Notch signaling pathway controls cell fate decisions in vertebrate and invertebrate tissues. NOTCH signaling
RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice
 SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
Pre-made Reporter Lentivirus for JAK-STAT Signaling Pathway
 Pre-made Reporter for JAK-STAT Signaling Pathway Cat# Product Name Amounts LVP937-P or: LVP937-P-PBS ISRE-GFP (Puro) LVP938-P or: LVP938-P-PBS ISRE-RFP (Puro) LVP939-P or: LVP939-P-PBS ISRE-Luc (Puro)
Pre-made Reporter for JAK-STAT Signaling Pathway Cat# Product Name Amounts LVP937-P or: LVP937-P-PBS ISRE-GFP (Puro) LVP938-P or: LVP938-P-PBS ISRE-RFP (Puro) LVP939-P or: LVP939-P-PBS ISRE-Luc (Puro)
Supplemental Table 1. Plasma NEFA and liver triglyceride levels in ap2-hif1ako and ap2-hif2ako mice under control and high fat diets.
 Supplemental Table 1. Plasma NEFA and liver triglyceride levels in Hif1aKO and Hif2aKO mice under control and high fat diets. Hif1a (n=6) Hif1aK O (n=6) Hif2a Hif2aK O Hif1a (n=5) Hif1aKO (n=5) Hif2a Hif2aK
Supplemental Table 1. Plasma NEFA and liver triglyceride levels in Hif1aKO and Hif2aKO mice under control and high fat diets. Hif1a (n=6) Hif1aK O (n=6) Hif2a Hif2aK O Hif1a (n=5) Hif1aKO (n=5) Hif2a Hif2aK
Insulin-induced gene 1 (insig-1) mrna increases dramatically in
 Insig-1 brakes lipogenesis in adipocytes and inhibits differentiation of preadipocytes Jinping Li*, Kiyosumi Takaishi*, William Cook*, Sara Kay McCorkle*, and Roger H. Unger* *Touchstone Center for Diabetes
Insig-1 brakes lipogenesis in adipocytes and inhibits differentiation of preadipocytes Jinping Li*, Kiyosumi Takaishi*, William Cook*, Sara Kay McCorkle*, and Roger H. Unger* *Touchstone Center for Diabetes
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
/06/$15.00/0 Molecular Endocrinology 20(9): Copyright 2006 by The Endocrine Society doi: /me
 0888-8809/06/$15.00/0 Molecular Endocrinology 20(9):2062 2079 Printed in U.S.A. Copyright 2006 by The Endocrine Society doi: 10.1210/me.2005-0316 Androgens, Progestins, and Glucocorticoids Induce Follicle-Stimulating
0888-8809/06/$15.00/0 Molecular Endocrinology 20(9):2062 2079 Printed in U.S.A. Copyright 2006 by The Endocrine Society doi: 10.1210/me.2005-0316 Androgens, Progestins, and Glucocorticoids Induce Follicle-Stimulating
A simple flow cytometric method to assess lipid accumulation in fat cells
 A simple flow cytometric method to assess lipid accumulation in fat cells Ying-Hue Lee 1 *, Su-Yin Chen 1, Rudolf J. Wiesner 2 and Yi-Fong Huang 1 1 Laboratory of Molecular Pathology, Institute of Molecular
A simple flow cytometric method to assess lipid accumulation in fat cells Ying-Hue Lee 1 *, Su-Yin Chen 1, Rudolf J. Wiesner 2 and Yi-Fong Huang 1 1 Laboratory of Molecular Pathology, Institute of Molecular
DOWNLOAD PDF ADIPOSE TISSUE AND ADIPOKINES IN HEALTH AND DISEASE (NUTRITION AND HEALTH)
 Chapter 1 : Adiposity, Adipokines, and Adiposopathy - Sick Fat Explained Adipose Tissue and Adipokines in Health and Disease, Second Edition is a useful resource for physicians interested in adipose tissue
Chapter 1 : Adiposity, Adipokines, and Adiposopathy - Sick Fat Explained Adipose Tissue and Adipokines in Health and Disease, Second Edition is a useful resource for physicians interested in adipose tissue
Exendin-4-mediated cell proliferation in beta-cells: focusing on the transcription factors
 Exendin-4-mediated cell proliferation in beta-cells: focusing on the transcription factors Seo-Yoon Chang, Jae Min Cho, Yang-Hyeok Jo, Myung-Jun Kim Departments of Physiology, College of Medicine, The
Exendin-4-mediated cell proliferation in beta-cells: focusing on the transcription factors Seo-Yoon Chang, Jae Min Cho, Yang-Hyeok Jo, Myung-Jun Kim Departments of Physiology, College of Medicine, The
Pelagia Research Library
 Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(3): 32-36 ISSN: 0976-8688 CODEN (USA): PSHIBD Study of the effect of ethanolic extract of Solanum xanthocarpum schrad &
Available online at www.pelagiaresearchlibrary.com Der Pharmacia Sinica, 2014, 5(3): 32-36 ISSN: 0976-8688 CODEN (USA): PSHIBD Study of the effect of ethanolic extract of Solanum xanthocarpum schrad &
Chapter 7 Conclusions
 VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
SUPPLEMENTARY INFORMATION. Supp. Fig. 1. Autoimmunity. Tolerance APC APC. T cell. T cell. doi: /nature06253 ICOS ICOS TCR CD28 TCR CD28
 Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
Mamofillin New aesthetic perspective
 New aesthetic perspective info@ White adipose tissue (WAT) White adipose tissue (WAT) is the prevalent type in human adults functioning as the major storage site for the lipids absorbed from daily intake
New aesthetic perspective info@ White adipose tissue (WAT) White adipose tissue (WAT) is the prevalent type in human adults functioning as the major storage site for the lipids absorbed from daily intake
Jumpstart your research with ViraPower Lentiviral Expression Systems
 ViraPower Lentiviral Expression Systems Jumpstart your research with ViraPower Lentiviral Expression Systems With ViraPower Lentiviral Systems you can: Efficiently transduce both dividing and non-dividing
ViraPower Lentiviral Expression Systems Jumpstart your research with ViraPower Lentiviral Expression Systems With ViraPower Lentiviral Systems you can: Efficiently transduce both dividing and non-dividing
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli
 BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
Interleukin-6; pathogenesis and treatment of autoimmune inflammatory diseases
 54 Review Article Interleukin-6; pathogenesis and treatment of autoimmune inflammatory diseases Toshio Tanaka 1, 2), Masashi Narazaki 3), Kazuya Masuda 4) and Tadamitsu Kishimoto 4, ) 1) Department of
54 Review Article Interleukin-6; pathogenesis and treatment of autoimmune inflammatory diseases Toshio Tanaka 1, 2), Masashi Narazaki 3), Kazuya Masuda 4) and Tadamitsu Kishimoto 4, ) 1) Department of
Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists
 Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists Marco Scarsi Biozentrum - Swiss Institute of Bioinformatics Drug discovery and development 12-15 years of
Combining molecular modelling with experiments: Sulfonylureas and glinides as new PPARγ agonists Marco Scarsi Biozentrum - Swiss Institute of Bioinformatics Drug discovery and development 12-15 years of
Pre-made Reporter Lentivirus for NF-κB Signal Pathway
 Pre-made Reporter for NF-κB Signal Pathway Cat# Product Name Amounts LVP965-P or: LVP965-P-PBS NFKB-GFP (Puro) LVP966-P or: LVP966-P-PBS NFKB-RFP (Puro) LVP967-P or: LVP967-P-PBS NFKB-Luc (Puro) LVP968-P
Pre-made Reporter for NF-κB Signal Pathway Cat# Product Name Amounts LVP965-P or: LVP965-P-PBS NFKB-GFP (Puro) LVP966-P or: LVP966-P-PBS NFKB-RFP (Puro) LVP967-P or: LVP967-P-PBS NFKB-Luc (Puro) LVP968-P
Naohito AOKI, Erina ARAKAWA and Miyuki ITO. Department of Life Science, Graduate School of Bioresources, Mie University Tsu ABSTRACT
 Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643
 Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
s u p p l e m e n ta ry i n f o r m at i o n
 Figure S1 Characterization of tet-off inducible cell lines expressing GFPprogerin and GFP-wt lamin A. a, Western blot analysis of GFP-progerin- or GFP-wt lamin A- expressing cells before induction (0d)
Figure S1 Characterization of tet-off inducible cell lines expressing GFPprogerin and GFP-wt lamin A. a, Western blot analysis of GFP-progerin- or GFP-wt lamin A- expressing cells before induction (0d)
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION
 Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
A New Preadipocyte Cell Line, AP-18, Established from Adult Mouse Adipose Tissue
 Tohoku J. Exp. Med., 2005, 207, 209-216 A New Preadipocyte Cell Line 209 A New Preadipocyte Cell Line, AP-18, Established from Adult Mouse Adipose Tissue HIDEYUKI DOI, NAKUI MASAKI, HITOSHI TAKAHASHI,
Tohoku J. Exp. Med., 2005, 207, 209-216 A New Preadipocyte Cell Line 209 A New Preadipocyte Cell Line, AP-18, Established from Adult Mouse Adipose Tissue HIDEYUKI DOI, NAKUI MASAKI, HITOSHI TAKAHASHI,
Supplementary Information. Induction of p53-independent apoptosis by ectopic expression of HOXA5
 Supplementary Information Induction of p53-independent apoptosis by ectopic expression of in human liposarcomas Dhong Hyun Lee 1, *, Charles Forscher 1, Dolores Di Vizio 2, 3, and H. Phillip Koeffler 1,
Supplementary Information Induction of p53-independent apoptosis by ectopic expression of in human liposarcomas Dhong Hyun Lee 1, *, Charles Forscher 1, Dolores Di Vizio 2, 3, and H. Phillip Koeffler 1,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Certificate of Analysis
 Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
TITLE: Characterization of the Mechanisms of IGF-I-Mediated Stress-Activated Protein Kinase Activation in Human Breast Cancer Cell MCF-7
 AD Award Number DAMD17-98-1-8066 TITLE: Characterization of the Mechanisms of IGF-I-Mediated Stress-Activated Protein Kinase Activation in Human Breast Cancer Cell MCF-7 PRINCIPAL INVESTIGATOR: Wei Liu,
AD Award Number DAMD17-98-1-8066 TITLE: Characterization of the Mechanisms of IGF-I-Mediated Stress-Activated Protein Kinase Activation in Human Breast Cancer Cell MCF-7 PRINCIPAL INVESTIGATOR: Wei Liu,
Receptors Functions and Signal Transduction- L4- L5
 Receptors Functions and Signal Transduction- L4- L5 Faisal I. Mohammed, MD, PhD University of Jordan 1 PKC Phosphorylates many substrates, can activate kinase pathway, gene regulation PLC- signaling pathway
Receptors Functions and Signal Transduction- L4- L5 Faisal I. Mohammed, MD, PhD University of Jordan 1 PKC Phosphorylates many substrates, can activate kinase pathway, gene regulation PLC- signaling pathway
* This work was supported, in whole or in part, by National Institutes of Health Grants HL076684, HL62984 (to N. L. W.), and ES (to S. H.
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 31, pp. 27836 27847, August 5, 2011 Printed in the U.S.A. Histone Deacetylase 9 Is a Negative Regulator of Adipogenic Differentiation * S Received for
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 31, pp. 27836 27847, August 5, 2011 Printed in the U.S.A. Histone Deacetylase 9 Is a Negative Regulator of Adipogenic Differentiation * S Received for
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
