Numerous studies have indicated that endothelial cells
|
|
|
- Patience Stokes
- 5 years ago
- Views:
Transcription
1 Lateral Zone of Cell-Cell Adhesion as the Major Fluid Shear Stress Related Signal Transduction Site Yumiko Kano, Kazuo Katoh, Keigi Fujiwara Abstract It has been proposed previously that actin filaments and cell adhesion sites are involved in mechanosignal transduction. In this study, we present certain morphological evidence that supports this hypothesis. The 3D disposition of actin filaments and phosphotyrosine-containing proteins in endothelial cells in situ was analyzed by using confocal microscopy and image reconstruction techniques. Surgical coarctations were made in guinea pig aortas, and the same 3D studies were conducted on such areas 1 week later. Stress fibers (SFs) were present at both basal and apical regions of endothelial cells regardless of coarctation, and several phosphotyrosine-containing proteins were associated with SF ends. Apical SFs had one end attached to the apical cell membrane and the other attached to either the basal membrane or the lateral cell border. Within the coarctation area, the actin filament containing and vinculin-containing structures became prominent, especially at the apical and the lateral regions. Substantially higher levels of anti-phosphotyrosine and anti-src staining were detected in the constricted area, particularly at the cell-cell apposition, whereas the anti focal adhesion kinase, anti CT10-related kinase, anti platelet endothelial cell adhesion molecule-l, anti-vinculin, and phalloidin staining intensities increased only slightly after coarctation. We propose that apical SFs directly transmit the mechanical force of flow from the cell apex to the lateral and/or basal SF anchoring sites and that the SF ends associated with signaling molecules are sites of signal transduction. Our results support the idea that the cell apposition area is the major fluid shear stress dependent mechanosignal transduction site in endothelial cells. (Circ Res. 2000;86: ) Key Words: endothelium mechanotransduction stress fibers phosphotyrosine coarctation Numerous studies have indicated that endothelial cells (ECs) have some mechanism for responding to fluid flow. 1 When cultured ECs are exposed to the flow of culture medium for a short period of time, a number of so-called early flow responses occur, including NO release, 1 G-protein activation, 2 4 Ca 2 mobilization, 5 8 protein kinase C activation, 9 11 increased levels of tyrosine phosphorylation, 1,12 platelet endothelial cell adhesion molecule-l (PECAM-l) tyrosine phosphorylation, 13,14 mitogen-activated protein kinase activation, 11,12 and directed cell migration. 15,16 These events are followed by later responses that become detectable typically after several hours of flow application, including changes in various gene expressions, 1 changes in cell shape, and cytoskeletal reorganization. 1,23 27 It is not certain whether and how any of the early types of responses relate to any of the later events. It is also uncertain whether these flow-induced responses are expressed by in situ ECs that are constantly exposed to blood flow. Although these observations demonstrate the ability of ECs to respond to fluid flow, the mechanism for flow sensing and signal transduction remains largely unknown. Fluid shear stress is the frictional force that acts on the apical cell surface. If the cell surface is easily deformable, the force will mostly be dissipated by deformation and may not be able to elicit flow responses in cells. Thus, shear stress dependent signal transduction is expected to begin at sites where cell surface deformation is less likely to occur. One possible place is the apical stress fiber (SF) plasma membrane attachment site (the apical plaque), 28 although there is no evidence that the apical plaque is a site for mechanosignal transduction. There are other rigid structures associated with the plasma membrane, such as the cell-substrate adhesion site (focal adhesion) and the cell-cell adhesion site. Bundled actin filaments are associated with all of these sites, and via the actin bundle network, the force of shear stress acting on the apical plaque may be transmitted to the other fixed parts of the cell, where activation of specific signaling molecules may occur. This scheme suggests the possibility that some flow-dependent signaling events are initiated at the focal adhesion site, which is not directly exposed to flow. Indeed, in cultured cells, flow-dependent and cell adhesion dependent mitogen-activated protein kinase activation has been reported, 11,12 and there are apical SFs that appear to span the distance between the apical and the basal cell surfaces. 28 This scheme also predicts signal transduction at the cell-cell attachment site. This laboratory has shown that PECAM-1, a cell-cell adhesion molecule, is tyrosine-phosphorylated when ECs are exposed to flow. 13,14 This PECAM-l phosphorylation could Received August 19, 1999; accepted December 3, From the Department of Structural Analysis, National Cardiovascular Center Research Institute, Suita, Osaka, Japan. Correspondence to Dr Yumiko Kano, Department of Structural Analysis, National Cardiovascular Center Research Institute, 5 Fujishiro-dai, Suita, Osaka, , Japan American Heart Association, Inc. Circulation Research is available at 425
2 426 Circulation Research March 3, 2000 Figure 1. Stereo pairs of in situ ECs in a control guinea pig aorta doubly stained by phalloidin (A) and anti-phosphotyrosine (PY-20) (B). A, 3D distribution of actin filaments. Thick SFs are observed at the base, and thin SFs are observed at the apical portion. Some apical SFs run into the actin circumferential ring (arrowheads), and some attach to the basal part of cells (arrows). B, PY-20 staining associated with the plasma membrane, especially the lateral portion. Anti-phosphotyrosine staining without corresponding actin filament staining was rarely observed. be triggered by the apical SF system that links the apical plaque and the cell adhesion site. Indeed, we have shown that PECAM-1 tyrosine phosphorylation does not occur in ECs treated with cytochalasin D. 14 Whether apical SFs terminate on the cell-cell adhesion site requires investigation. This signal transduction involving actin filaments predicts a certain SF organization in the cell. More specifically, it predicts the presence of strategically placed SF plasma membrane attachment sites on the apical, lateral, and basal parts of ECs and the association of signaling molecules with the lateral and basal sites. Previously, we have provided evidence for the presence of apical SFs and plaques in ECs in situ. 29 In the present study, by using guinea pig aortas and confocal microscopy together with image reconstruction techniques, we show the 3D organization of actin filament containing structures in ECs in situ. This overall actin cytoskeletal organization is consistent with the proposed model for mechanosignal transduction. We found that the apical SFs run between the apical cell surface and the lateral and the basal part of the cell but that the basal SFs stay on the basal part of Figure 2. Stereo pairs of in situ ECs in the coarctation zone doubly stained with phalloidin (A and C) and anti-vinculin (B). A and C, Compared with Figure 1A, the actin cytoskeleton is more developed. Arrows indicate apical SFs connecting to the basal portions of ECs. At high magnification (C), an apical plaque is shown by an arrowhead. B, Vinculin localization is shown. Bright and large staining spots are observed at both the cell apex and base. Bar 20 m. the cell, presumably providing tight adhesion of ECs to the basement membrane. When the flow rate (as well as fluid shear stress) in the vessel is increased by making a region of coarctation, SF expression is increased, especially the apical type. Also increased in the coarctation area is the expression of Src and tyrosine-phosphorylated proteins, especially in the cell-cell overlap area. These results are consistent with the idea that the lateral cell apposition zone is a major fluid flow related mechanosignal transduction site. Materials and Methods En Face Aortic Sample Preparation Guinea pig abdominal aortas were used. A small silicon tube was placed over some of the aortas for a week to analyze the effect of increased fluid shear stress on various morphological parameters. Aortas were fixed, longitudinally cut, and spread flat to expose the endothelium. 29
3 Kano et al Cell-Cell Adhesion and Signal Transduction 427 Figure 3. A stereo pair of ECs stained with anti-vinculin in a control vessel. Although dotty staining was present in the specimen at both the apical and the basal portions, weak apical staining was lost during the image reconstruction procedure. Microscopy and 3D Image Reconstruction En face aortic samples were stained with various antibodies and observed under a laser scanning microscope. To make 3D images, serial optical sections were made and, with the use of computer software, reconstructed into stereo images. Each 3D image is presented as a pair of sized-matched appropriately mounted micrographs that should be viewed through a special stereo viewer. An expanded Materials and Methods section is available online at Results Actin Filament Organization Whole-mount preparations of guinea pig abdominal aortas were stained with fluorescein-labeled phalloidin, and 3D images were reconstructed from confocal serial optical sections (Figure 1A). Each EC was delineated by a ring of strong staining near the lumenal surface, presumably representing actin filaments of the adherens junction. However, there were also actin rings near the base of the cell. Whether adherens junctions are present near the base of ECs requires investigation. SFs were generally aligned in the direction of blood flow. Thick SFs were present at the base, and some of them were quite short. Thin SFs were found in the apical portion of the cell. These apical SFs tended to span the whole length of the cell and appeared to run into the actin ring of the apical location (Figure 1A, arrowheads). Some SFs ran vertically, connecting the apical and the basal surfaces (Figure 1A, arrows). Weakly fluorescent structures were lost during 3D image processing of confocal optical serial sections. Thus, structures consisting of a small number of actin filaments are not represented in the reconstructed images. It is known that in situ SF expression is high in the region of increased fluid shear stress, 22,27,30,31 and indeed, in the guinea pig aorta, the actin cytoskeletal architecture was more exaggerated near bifurcations (data not shown). We had attempted to take advantage of this exaggerated actin filament organization for our analyses, but it was difficult to obtain flat whole-mount preparations of branching areas. Thus, to create an area with increased fluid shear stress that could be easily made into a whole-mount Figure 4. FAK and Crk localization in ECs. Optical sections were made near the cell base. A and B, Anti-FAK staining in the control area (A) and the coarctation zone (B), with an increased size and number of staining spots in panel B. C, Anti-Crk stained focal adhesions (arrowheads) and the general cytoplasm (seen as smooth staining). preparation, we made a coarctation in the straight region of the abdominal aorta. A stereo pair of the constricted area showed ECs with increased staining with rhodaminelabeled phalloidin. Apical SFs terminating on the circumferential actin ring were clearly demonstrated. Also clearly seen were the SFs that ran between the apical and the basal surfaces (Figure 2A, arrows). Stereo pairs showed that the actin cytoskeleton was membrane-associated, forming a cagelike structure underneath the plasma membrane and surrounding the entire body of cytoplasm. Increased shear stress upregulated the expression of apical SFs. Figure 2C, a more magnified stereo pair, shows the centrally located
4 428 Circulation Research March 3, 2000 Figure 5. Phosphotyrosine localization in the coarctation zone. A, Low-magnification micrograph showing the downstream edge of a coarctation (arrows). In the coarctation area (left), PY-20 staining is significantly increased. Coarctations formed folds in the vessel wall in the unconstricted area, and the top of folds showed increased labeling. B, Confocal image of the coarctation zone optically sectioned at the middle level of ECs. Staining is at the cell-cell junction. C, Side view of the endothelium. A confocal image obtained from a folded area shows strong staining at the cell-cell border (arrows) and weak linear staining along the apical plasma membrane (arrowheads). Bar 20 m. apical plaque (arrowhead) 28 and SFs running between the apical plaque and the base of the cell (arrows). These studies demonstrate that apical SFs are connected to either the cell-cell adhesion site or the basal plasma membrane. Vinculin Anti-vinculin staining associated with both basal and apical plaques of in situ ECs has been shown. 28,29 Stereo pairs of normal aortic ECs showed basal anti-vinculin spots but not the apical spots, presumably because of their low level of labeling (Figure 3). The extent of cell delineation by antivinculin was minimal. On the other hand, the level of anti-vinculin labeling in the coarctation area was considerably higher, and each staining spot in Figure 2B was significantly larger than the spots shown in Figure 3. The lateral and the apical vinculin spots also became noticeably prominent, indicating that heightened shear stress increased the size of vinculin-containing structures. FAK and Other Focal Adhesion-Associated Proteins Focal adhesion kinase (FAK) association with focal adhesions but not with apical plaques was reported in cultured cells. 28 In ECs in vivo, this kinase was localized to discrete spots at the base of cells both in control (Figure 4A) and constricted (Figure 4B) vessels. They are presumably focal adhesions that are arranged linearly along the direction of blood flow. Under increased shear stress, the number and size of anti-fak spots increased, suggesting that ECs reenforced their adhesion to the basement membrane by making more and larger focal adhesions. CT10-related kinase (Crk), an adopter protein for both paxillin and pp130 cas, also binds to FAK. Anti-Crk stained the basal portion of ECs, most notably focal adhesions (Figure 4C). Paxillin and pp130 cas were also immunolocalized to focal adhesions (data not shown). Our data indicate colocalization of FAK, Crk, paxillin, and pp130 cas in the focal adhesion of ECs in situ. Phosphotyrosine Because protein tyrosine phosphorylation occurs during intracellular signaling, we investigated phosphotyrosine levels in ECs in the constricted region (Figure 5A). There was a clear demarcation in anti-phosphotyrosine staining at the edge of coarctation. Although immunostaining in the low fluid shear stress area was low (Figure 5A, right), the area of increased shear stress exhibited a high level of staining (Figure 5A, left), which was closely associated with the lateral plasma membrane (Figure 5B). The similar sharp staining demarcation was observed at the proximal edge of coarctations. The lateral staining did not always form a sharp line, which might be expected if the adherens junction were labeled (Figure 5B). Instead, sheetlike staining patterns were observed, suggesting that the lateral cell membrane overlap area was labeled. A side view of ECs clearly showed staining along the entire cell-cell apposition (Figure 5C, arrows). Dotty staining was associated with the base of the cell and the apical cell membrane (Figure 5C, arrowheads). A stereo pair of anti-phosphotyrosine stained endothelium showed immunoreactivity being spread throughout the lateral plasma membrane overlap (Figure 1B). The specimen was also stained for actin filaments and showed colocalization of phosphotyrosine with actin filaments (Figure 1A). Although spotty staining corresponding to focal adhesions and apical
5 Kano et al Cell-Cell Adhesion and Signal Transduction 429 Figure 6. Localization of Src family kinases. A to D, Cryosections (A and B) and whole-mount preparations (C and D) stained with 1 of the 2 types of Src antibodies, SRC-2 (A and C) and N-16 (B and D). Src staining was mainly observed at cell-cell junctions but also along the top and base of cells in a dotted manner. IE indicates internal elastic lamina; SM, smooth muscle cells. E and F, ECs doubly stained with anti-vinculin (green) and anti-src (red). Superimposed images show yellow spots indicating colocalization of the 2 antigens in focal adhesions and apical plaques (arrows). An en face view of the basal portion (E) and a side view (F) are shown. Bar 10 m. plaques was present, SFs were not labeled. These results suggest that the cell-cell border is an active site of signaling events and that increased shear stress upregulates signaling activities. Src Src family proteins are tyrosine-phosphorylated at the time of signal transduction and are tyrosine kinases. Localization of Src was investigated by using 2 antibodies with different specificity: SRC-2 (recognizing the Src family kinases) and N-16 (recognizing only c-src). Cryosections of the aortic wall stained with SRC-2 showed labeling of the cell border and some basal spots, presumably focal adhesions (Figure 6A). The cell apex was also labeled in a dotted manner (Figure 6A). Cryosections stained with N-16 exhibited similar staining patterns, but anti c-src images were better defined than SRC-2 images (Figure 6B). Like anti-phosphotyrosine staining, the anti-src signal was spread along the entire length of cell-cell apposition. When these 2 antibodies were treated with appropriate synthetic Src peptides and then used to stain specimens, no labeling was observed (data not shown). A confocal optical section at the mid level of ECs showed dotty staining by SRC-2 at the cell-cell border (Figure 6C). N-16 gave a similar but better defined staining pattern (Figure 6D). An en face preparation stained doubly with anti-vinculin and SRC-2 was optically sectioned at the base of cells, where anti-vinculin stained focal adhesions. Figure 6E is a merged image of anti-vinculin (red) and anti-src (green) signals and shows colocalization (yellow) of the 2 antigens at focal adhesions. A double-labeled confocal side view is similarly illustrated in Figure 6F, demonstrating localization of Src kinases at focal adhesions. This micrograph also demonstrates the localization of Src kinases at apical plaques (arrows). Src expression was investigated in the coarctation zone. A low-magnification photograph showed increased anti- Src staining in the area of coarctation (Figure 7A). The border between low and high levels of Src expression was sharp and corresponded well with the edge of coarctation (Figure 7B). Increased staining occurred at all localization sites, including the general cytoplasm, but staining was particularly strong at the cell border (Figure 7C). The localization of Src kinases was remarkably similar to the distribution of phosphotyrosine-containing proteins. Csk C-terminal Src kinase (Csk) is a nonreceptor-type tyrosine kinase that phosphorylates a tyrosine residue in the C-terminal region of Src family kinases and inactivates them.
6 430 Circulation Research March 3, 2000 Figure 7. Localization of Src in the coarctation zone. A, Low-magnification view of the entire coarctation area. The boundary between the low and the high shear stress areas is clearly observed. B, Upstream edge of a coarctation showing a sharp staining boundary. C, Confocal optical section at the middle level of ECs, showing staining at the cell-cell boundary. Strong anti-csk staining was present at the cell-cell apposition (Figure 8). This staining was broad, inasmuch as it was seen with anti-src kinases and anti-phosphotyrosine. Discussion The response of ECs to fluid flow has mostly been studied by using in vitro systems. Although this approach has provided information regarding the basic property of this cellular response, it is commonly believed that in vitro data may not totally reflect the situation in vivo. Therefore, it is of paramount importance that ECs in situ be closely studied. Understandably, however, performing experiments on cells in vivo is difficult, and available data on in situ ECs are severely limited. In the present study, using guinea pig abdominal aortic ECs in situ, we obtained certain morphological information pertinent to mechanosignal transduction by these cells. Although these immunolocalization studies do not provide definitive solutions to the mechanism of flow-induced signaling by ECs, we suggest that they provide insights into the way these cells respond to flow. Actin Bundle Organization and Signal Transduction A role of the cytoskeleton in mechanosignal transduction by ECs has been proposed. 1,32 36 We have suggested that the actin cytoskeleton might transmit mechanical forces directly from one part of the cell to another. Such a scheme predicts a certain pattern of actin bundle disposition. In the present study, we have demonstrated the presence of SFs that run in the specific ways that have been predicted by our mechanosignal transduction hypothesis. Of particular interest are the SFs located in the apical part of ECs. In cultured fibroblasts, one of the ends of the apical SF is attached to the apical plasma membrane via the apical plaque, and the other end appears to terminate at the basal plasma membrane. 28 Likewise, one end of the apical SF in ECs in situ is attached to the apical plaque, whose molecular makeup is similar to that of the apical plaque of cultured cells. 29 However, it was not clear where the other end of the apical SF was located. In the present study, we demonstrated that this end was attached to either the basal focal adhesion or the cell-cell apposition site. The apical SF attachment to the focal adhesion site is presumably mediated by the integrin system. The lateral termination sites were clearly above the base of the cell, indicating that they were not the cellsubstrate attachment sites. This brings up a new set of questions regarding the structure and the molecular makeup of this lateral SF plasma membrane binding site, which may Figure 8. Anti-Csk staining at the cell-cell border. A confocal optical section at the middle height of cells is shown.
7 Kano et al Cell-Cell Adhesion and Signal Transduction 431 be different from the binding sites involved in the integrinbased anchoring of SFs to the membrane. Integrin is not concentrated at the cell-cell adhesion site. Cadherin and PECAM-l are the major transmembrane proteins at the lateral cell adhesion site, and both proteins have a -catenin binding activity Actin filament bundles running into the lateral portion of cultured cells has been reported, and -catenin is localized to such sites. 41 There is a circumferential ring of actin filaments associated with the lateral plasma membrane of ECs forming a monolayer. In many instances, apical SFs appeared to run into this structure, at the level of resolution of a light microscope. Whether these 2 actin filament bundles are physically linked is an intriguing question, which we are now investigating. As we have outlined in the introductory section of the present study, fluid shear stress acts on the rigid part of cell. The apical plaque is such a part because it is anchored by the SF system. Thus, it is a candidate site for shear stress sensing and initial signal transduction. However, it is also possible that through the SF system, the force is transmitted to focal adhesions and cell-cell apposition sites, where appropriate signaling pathways can be activated. Thus, any individual region or combinations of these 3 regions are good candidates for fluid shear stress sensing and signal initiation sites. The present study has provided a structural basis for this hypothesis. Localization of Signaling Molecules Tyrosine residues of many proteins involved in signal transduction are phosphorylated at the time of their activation or inactivation. 1,42 Thus, the level of tyrosine phosphorylation at a given area in a cell can be a reflection of local signal transduction activities. Anti-phosphotyrosine staining was detected at apical plaques, regions of cell-cell apposition, and focal adhesions. The focal adhesion has been identified as a site of signal transduction, and several tyrosine-phosphorylated polypeptides, such as FAK, vinculin, paxillin, Crk, pp130 cas, and Csk, have been localized Vinculin and paxillin are also concentrated at apical plaques. 28,29 These proteins may be responsible for anti-phosphotyrosine staining of focal adhesions and apical plaques, but other proteins, perhaps specific for shear stress signal transduction, may also contribute to this staining. Anti-phosphotyrosine staining was much stronger at the cell-cell contact site than at the other 2 locations, suggesting more active signaling events at the lateral cell overlap. Because increased shear stress significantly increased this staining, the region of cell-cell apposition may be the most active signal transduction site of shear stress. We have reported previously that when a confluent monolayer of cultured ECs is exposed to 5 dyne/cm 2 of shear stress or osmotic changes, PECAM-1 is tyrosine-phosphorylated within 1 minute. 13,53 PECAM-1 is an EC adhesion molecule localized at the membrane overlap region between neighboring cells. Thus, a portion of the phosphotyrosine localized at the cell-cell border may be in PECAM-l. Unlike vascular endothelial (VE)-cadherin localization, which appears as a line at the cell-cell overlap, anti PECAM-l staining was seen as a belt. 14,54 As shown in the present study, antiphosphotyrosine staining was broad along the cell-cell border. Src family kinases and Csk were also localized to the entire cell-cell overlap. Thus, PECAM-1, Src, and Csk, all of which are phosphotyrosine-containing molecules, are localized in a nearidentical pattern to the cell-cell border. These results suggest that all or some of these molecules work together for mechanosignal transduction at the cell-cell border. Indeed, our in vitro study shows that c-src is an effective PECAM-1 kinase. 13,53 Immunolocalization also suggests that the activity of Src kinases is tightly controlled at the cell-cell border by Csk. Effect of Coarctation A coarctation was made in the straight region of the abdominal aorta to create a region of increased fluid shear stress that can be easily made into an en face specimen for microscopy. The same approach has been taken by other investigators. 55 Precise fluid mechanical analyses on the blood flow pattern and assessment of the magnitude of the increase of fluid shear stress within the constricted region were not performed. Because we have used a straight region of the blood vessels, it is expected that the flow pattern within the coarctation is generally laminar (as is assumed for a nonbranching straight blood vessel) and that the level of fluid shear stress in the constricted zone is significantly heightened. The flow pattern in the region immediately distal to the coarctation is expected to be nonlaminar, and accordingly, the fluid shear stress level there is expected to be lower than that of the normal flow area. Although these local differences in fluid shear stress levels are likely, ECs are expected to be under higher shear stress in the constricted region than in the areas immediately proximal and distal to a coarctation. Thus, we expect that the expression of any shear stress dependent events is heightened in the area of coarctation. The basic localization patterns of various proteins were the same between regions with and without a coarctation. However, the actin cytoskeleton and the structures labeled with anti-vinculin, anti-paxillin, and anti-fak were enlarged in ECs in the coarctation area. Inasmuch as most of these latter structures are focal adhesions, our results suggest that cells under increased shear stress reenforce their adhesion to the substrate by making larger and possibly more numerous adhesion sites. All these changes, however, were detectable only at a high magnification. When en face specimens stained with anti- FAK, anti-vinculin, anti-paxillin, or fluorescent phalloidin were observed at low magnifications, there was no fluorescence boundary between the low and the high shear stress areas. This indicates that the extent of increased expression of these proteins in the coarctation area is not incredibly high. However, in the specimens stained with anti-phosphotyrosine or anti-src, a clear demarcation was detected between the low and the high shear stress regions, indicating that Src expression is substantially upregulated by shear stress and that the amount of proteins with phosphorylated tyrosines is also highly concentrated in the high shear stress area. Observations at high magnifications revealed that increased staining was mostly associated with the lateral cell overlap region. It is not clear at this time what types of signaling pathways are activated at the cell border, but they are presumably fluid shear stress dependent signaling events perhaps involving PECAM-1, Src, and Csk. These data suggest the cell-cell
8 432 Circulation Research March 3, 2000 apposition to be an important site for mechanosignal transduction in ECs. Many types of EC flow responses, especially the early ones, are transient. The present study indicates that although the ECs in the coarctation zone have been exposed to increased shear stress for 1 week, certain types of signaling events are still occurring. Consistent with this interpretation is our observation that both anti-src and anti-phosphotyrosine heavily stain ECs at arterial branching, where the average fluid shear stress is always higher than the straight region of the same vessel. Some of the long-lasting responses are related to cell morphology, including cell-shape changes and SF alignment, and these responses are known to be reversible. To maintain the expression of a stimulus-dependent, reversible response, cells may need to continuously monitor the level of the stimulus and to generate a positive signal for the response. Thus, we suggest that the increased expression of SFs and focal adhesions and the elongated cell shape are maintained in ECs in the region of high shear stress because the fluid shear stress dependent signal transduction machinery of these cells is continuously active and keeps generating positive signals. This may be the reason for the increased expression of phosphotyrosine in the zone of high shear stress. We propose that the expression and organization of the SF system, which includes the SF plasma membrane attachment site and possibly of other morphological parameters in ECs in situ, are dynamically maintained by fluid shear stress dependent signals transmitted continuously from the area of cell-cell adhesion. Acknowledgments This study was supported in part by grants-in-aid for Scientific Research from the Ministry of Education, Science, and Culture of Japan, grants from the Ministry of Health and Welfare of Japan, and a grant from the Organization for Pharmaceutical Safety and Research. References 1. Davies PF. Flow-mediated endothelial mechanotransduction. Physiol Rev. 1995;75: Ohno M, Gibbons GH, Dzau VJ, Cooke JP. Shear stress elevates endothelial cgmp: role of a potassium channel and G protein coupling. Circulation. 1993;88: Kuchan MJ, Hanjoong JO, Frangos AJ. Role of G protein in shear stress-mediated nitric oxide production by endothelial cells. Am J Physiol. 1994;267:C753 C Gudi SRP, Clark CB, Frangos JA. Fluid flow rapidly activates G proteins in human endothelial cells: involvement of G proteins in mechanochemical signal transduction. Circ Res. 1996;79: Mo M, Eskin SG, Schilling WP. Flow-induced changes in Ca 2 signaling of vascular endothelial cells: effects of shear stress and ATP. Am J Physiol. 1991;260:H1698 H Ando J, Komatsuda T, Kamiya A. Intracellular calcium response to directly applied mechanical shearing force in cultured vascular endothelial cells. Biorheology. 1994;31: Shen J, Luscinskas FW, Connolly A, Dewey CFJ, Gimbrone MAJ. Fluid shear stress modulates cytosolic free calcium in vascular endothelial cells. Am J Physiol. 1992;262:C384 C Geiger RV, Berk BC, Alexander RW, Nerem RM. Flow-induced calcium transients in single endothelial cells: spatial and temporal analysis. Am J Physiol. 1984;262:C1441 C Kuchan MJ, Frangos JA. Shear stress regulates endothelin-1 release via protein kinase C and cgmp in cultured endothelial cells. Am J Physiol. 1993;264:H150 H Romer LH, McLean N, Turner CE, Burridge K. Tyrosine kinase activity, cytoskeletal organization, and motility in human vascular endothelial cells. Mol Biol Cell. 1994;5: Tseng H, Peterson TE, Berk BC. Fluid shear stress stimulates mitogenactivated protein kinases in bovine aortic endothelial cells. Circ Res. 1995;77: Ishida M, Peterson TE, Kovach NL, Berk BC. MAP kinase activation by flow in endothelial cells: role of integrins and tyrosine kinases. Circ Res. 1996;79: Osawa M, Masuda M, Harada N, Lopes RB, Fujiwara K. Tyrosine phosphorylation of platelet endothelial cell adhesion molecule-1 (PECAM-1, CD31) in mechanically stimulated vascular endothelial cells. Eur J Cell Biol. 1997;72: Masuda M, Osawa M, Shigematsu H, Harada N, Fujiwara K. Platelet endothelial cell adhesion molecule-1 is a major SH-PTP2 binding protein in vascular endothelial cells. FEBS Lett. 1997;408: Masuda M, Fujiwara K. Cultured endothelial cells respond to fluid shear stress. In: Clark EB, Takao A, eds. Developmental Cardiology: Morphogenesis and Function. Mt Kisco, NY; Futura Publishing Co; 1990: Masuda M, Fujiwara K. The biased lamellipodium development and microtubule organizing center position in vascular endothelial cells migrating under the influence of fluid flow. Biol Cell. 1993;77: Flaherty JT, Pierce JE, Ferrans VJ, Patel DJ, Tucker WK, Fry DL. Endothelial nuclear patterns in the canine arterial tree with particular reference to hemodynamic events. Circ Res. 1972;30: Dewey CFJ, Bussolari SR, Gimbrone MAJ, Davies PF. The dynamic response of vascular endothelial cells to fluid shear stress. J Biomech Eng. 1981;103: Eskin SG, Ives CL, McIntire LV, Navarro LT. Response of cultured endothelial cells to steady flow. Microvasc Res. 1984;28: Reidy MA, Langille BL. The effect of local blood flow patterns on endothelial cell morphology. Exp Mol Pathol. 1980;32: Nerem RM, Levesque MJ, Cornhill JF. Vascular endothelial morphology as an indicator of the pattern of blood flow. J Biomech Eng. 1981;103: Yoshida Y, Okano M, Wang S, Kobayashi M, Kawasumi M, Hagiwara H, Mitsumata M. Hemodynamic-force-induced difference of interendothelial junctional complexes. Ann N Y Acad Sci. 1995; 748: Kim DW, Gotlieb AI, Langille BL. In vivo modulation of endothelial F-actin microfilaments by experimental alterations in shear stress. Arteriosclerosis 1989;9: Ookawa K, Sato M, Ohshima N. Changes in the microstructure of cultured porcine aortic endothelial cells in the early stage after applying a fluid-imposed shear stress. J Biomech. 1992;25: White GE, Gimbrone MAJ, Fujiwara K. Factors influencing the expression of stress fibers in vascular endothelial cells in situ. J Cell Biol. 1983;97: Franke RP, Gräfe M, Schnittler H, Seiffge D, Mittermayer C. Induction of human vascular endothelial stress fibers by fluid shear stress. Nature. 1984;16: Wong AJ, Pollard TD, Herman IM. Actin filament stress fibers in vascular endothelial cells in vivo. Science. 1983;219: Katoh K, Masuda M, Kano Y, Jinguji Y, Fujiwara K. Focal adhesion proteins associated with apical stress fibers of human fibroblasts. Cell Motil Cytoskeleton. 1995;31: Kano Y, Katoh K, Masuda M, Fujiwara K. Macromolecular composition of stress fiber plasma membrane attachment sites in endothelial cells in situ. Circ Res. 1996;79: White GE, Fujiwara K. Expression and intracellular distribution of stress fibers in aortic endothelium. J Cell Biol. 1986;103: Kim DW, Langille BL, Wong MKK, Gotlieb AI. Patterns of endothelial microfilament distribution in the rabbit aorta in situ. Circ Res. 1989;64: Wechezak AR, Wight TN, Viggers RF, Sauvage LR. Endothelial adherence under shear stress is dependent upon microfilament reorganization. J Cell Physiol. 1989;139: Morita T, Kurihara H, Maemura K, Yoshizumi M, Yazaki Y. Disruption of cytoskeletal structures mediates shear stress-induced endothelin-1 gene expression in cultured porcine aortic endothelial cells. J Clin Invest. 1993;92: Knudsen H, Frangos JA. Role of cytoskeleton in shear stress-induced endothelial nitric oxide production. Am J Physiol. 1997;273:H347 H355.
9 Kano et al Cell-Cell Adhesion and Signal Transduction Takahashi M, Berk BC. Mitogen-activated protein kinase (ERK1/2) activation by shear stress and adhesion in endothelial cells. J Clin Invest. 1996;98: Busse R, Fleming I. Pulsatile stretch and shear stress: physical stimuli determining the production of endothelium-derived relaxing factors. J Vasc Res. 1998;35: Matsumura T, Wolff K, Petzelbauer P. Endothelial cell tube formation depends on cadherin 5 and CD31 interactions with filamentous actin. J Immunol. 1997;158: McCrea PD, Gumbiner BM. Purification of a 92-kDa cytoplasmic protein tightly associated with the cell-cell adhesion molecule E-cadherin (Uvomorulin). J Biol Chem. 1991;266: Ozawa M, Baribault H, Kemler R. The cytoplasmic domain of the cell adhesion molecule uvomorulin associates with three independent proteins structurally related in different species. EMBO J. 1989;8: Kusano K, Masuda M, Lopes RB, Osawa M, Fujiwara K. Evidence for direct interaction between PECAM-1 and beta-catenin. Mol Biol Cell. 1998;9:85a. Abstract. 41. Gloushankova NA, Krendel MF, Alieva NO, Bonder EM, Feder HH, Vasiliev JM, Gelfand IM. Dynamics of contacts between lamellae of fibroblasts: essential role of the actin cytoskeleton. Proc Natl Acad Sci USA. 1998;95: Lampugnani MG, Corada M, Andriopoulou P, Esser S, Risau W, Dejana E. Cell confluence regulates tyrosine phosphorylation of adherens junction components in endothelial cells. J Cell Sci. 1997;110: Clark EA, Brugge JS. Integrins and signal transduction pathways: the road taken. Science. 1995;268: Schwartz MA, Schaller MD, Ginsberg MH. Integrins: emerging paradigms of signal transduction. Annu Rev Cell Dev Biol. 1995;11: Burridge K, Chrzanowska-Wodnicka M. Focal adhesions, contractility and signaling. Annu Rev Cell Dev Biol. 1996;12: Schaller MD, Borgman CA, Cobb BS, Vines RR, Reynold AB, Parsons JT. pp125 FAK a structurally distinctive protein tyrosine kinase associated with focal adhesions. Proc Natl Acad Sci U S A. 1992;89: Sefton BM, Hunter T, Ball EH, Singer SJ. Vinculin: a cytoskeletal target of the transforming protein of Rous sarcoma virus. Cell. 1981;24: Burridge K, Feramisco JR. Microinjection and localization of a 130K protein in living fibroblasts: a relationship to actin and fibronectin. Cell. 1980;19: Turner CE, Miller JT. Primary sequence of paxillin contains putative SH2 and SH3 domain binding motifs and multiple LIM domains: identification of a vinculin and pp125 FAK -binding region. J Cell Sci. 1994;107: Nievers MG, Birge RB, Greulich H, Verkleij AJ, Hanafusa H. v-crkinduced cell transformation: changes in focal adhesion composition and signaling. J Cell Sci. 1997;110: Petch LA, Bockholt SM, Bouton A, Parsons JT, Burridge K. Adhesioninduced tyrosine phosphorylation of the 130 SRC substrate. J Cell Sci. 1995;108: Bergman M, Joukov V, Virtanen I, Alitalo K. Overexpressed Csk tyrosine kinase is localized in focal adhesions, causes reorganization of v 5 integrin, and interferes with HeLa cell spreading. Mol Cell Biol. 1995; 15: Harada N, Masuda M, Fujiwara K. Fluid flow and osmotic stress induce tyrosine phosphorylation of an endothelial cell 128 kda surface protein. Biochem Biophys Res Commun. 1995;214: Albelda SM, Oliver PD, Romer LH, Buck CA. Endo-CAM: a novel endothelial cell-cell adhesion molecules. J Cell Biol. 1990;110: Gabriels JE, Paul DL. Connexin43 is highly localized to sites of disturbed flow in rat aortic endothelium but connexin37 and connexin40 are more uniformly distributed. Circ Res. 1998;83:
Numerous studies have indicated that endothelial cells
 Lateral Zone of Cell-Cell Adhesion as the Major Fluid Shear Stress Related Signal Transduction Site Yumiko Kano, Kazuo Katoh, Keigi Fujiwara Downloaded from http://circres.ahajournals.org/ by guest on
Lateral Zone of Cell-Cell Adhesion as the Major Fluid Shear Stress Related Signal Transduction Site Yumiko Kano, Kazuo Katoh, Keigi Fujiwara Downloaded from http://circres.ahajournals.org/ by guest on
Linda Lowe Krentz Bioscience in the 21 st Century October 22, 2010
 Cell Biomechanics Linda Lowe Krentz Bioscience in the 21 st Century October 22, 2010 Outline for today Tubes Vessel mechanics and disease Models Lung mechanics Technology integration Atherosclerosis is
Cell Biomechanics Linda Lowe Krentz Bioscience in the 21 st Century October 22, 2010 Outline for today Tubes Vessel mechanics and disease Models Lung mechanics Technology integration Atherosclerosis is
20 2 Stomach Fig. 2.1 An illustration showing different patterns of the myenteric plexus peculiar to the regions in the guinea-pig stomach stained wit
 Stomach 2 The stomach is unique in that ICC have a different distribution in proximal and distal regions of the same organ. ICC-CM and ICC-LM are densely distributed throughout the thick circular and longitudinal
Stomach 2 The stomach is unique in that ICC have a different distribution in proximal and distal regions of the same organ. ICC-CM and ICC-LM are densely distributed throughout the thick circular and longitudinal
Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue
 Megan McCain ES240 FEM Final Project December 19, 2006 Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue Cardiac myocytes are the cells that constitute the working muscle of the heart.
Megan McCain ES240 FEM Final Project December 19, 2006 Stretching Cardiac Myocytes: A Finite Element Model of Cardiac Tissue Cardiac myocytes are the cells that constitute the working muscle of the heart.
Localization of talin in skeletal and cardiac muscles
 Volume 200, number 1 FEBS 3591 May 1986 Localization of talin in skeletal and cardiac muscles A.M. Belkin, N.I. Zhidkova and V.E. Koteliansky* Laboratory of Molecular and Cellular Cardiology, Institute
Volume 200, number 1 FEBS 3591 May 1986 Localization of talin in skeletal and cardiac muscles A.M. Belkin, N.I. Zhidkova and V.E. Koteliansky* Laboratory of Molecular and Cellular Cardiology, Institute
The dynamic regulation of blood vessel caliber
 INVITED BASIC SCIENCE REVIEW The dynamic regulation of blood vessel caliber Colleen M. Brophy, MD, Augusta, Ga BACKGROUND The flow of blood to organs is regulated by changes in the diameter of the blood
INVITED BASIC SCIENCE REVIEW The dynamic regulation of blood vessel caliber Colleen M. Brophy, MD, Augusta, Ga BACKGROUND The flow of blood to organs is regulated by changes in the diameter of the blood
Histology = the study of tissues. Tissue = a complex of cells that have a common function
 { EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
{ EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility
 Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
CELL BIOLOGY - CLUTCH CH CELL JUNCTIONS AND TISSUES.
 !! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
!! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
Principles of Genetics and Molecular Biology
 Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
04_polarity. The formation of synaptic vesicles
 Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
Shear Stress Induces Changes in the Morphology and Cytoskeleton Organisation of Arterial Endothelial Cells
 Eur J Vasc Endovasc Surg 9, 86-92 (1995) Shear Stress Induces Changes in the Morphology and Cytoskeleton Organisation of Arterial Endothelial Cells Alessandra Cucina, Antonio V. Sterpetti, Guntars Pupelis,
Eur J Vasc Endovasc Surg 9, 86-92 (1995) Shear Stress Induces Changes in the Morphology and Cytoskeleton Organisation of Arterial Endothelial Cells Alessandra Cucina, Antonio V. Sterpetti, Guntars Pupelis,
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Blood Vessel Mechanics
 Blood Vessel Mechanics Ying Zheng, Ph.D. Department of Bioengineering BIOEN 326 11/01/2013 Blood Vessel Structure A Typical Artery and a Typical Vein Pressure and Blood Flow Wall stress ~ pressure Poiseuille
Blood Vessel Mechanics Ying Zheng, Ph.D. Department of Bioengineering BIOEN 326 11/01/2013 Blood Vessel Structure A Typical Artery and a Typical Vein Pressure and Blood Flow Wall stress ~ pressure Poiseuille
Muscle Tissue. Muscle Development and Repair. Development: fusion of myoblasts. Repair: Satellite cells (S) 3 Types of Muscle
 ANNOUNCEMENTS Review Session Every Friday at 12:20 Muscle Tissue 3 Types of Muscle Function: Force generation Lab Practical Coming up! October 26 th, 27 th Muscle Tissue Striated Nonstriated Skeletal Smooth
ANNOUNCEMENTS Review Session Every Friday at 12:20 Muscle Tissue 3 Types of Muscle Function: Force generation Lab Practical Coming up! October 26 th, 27 th Muscle Tissue Striated Nonstriated Skeletal Smooth
KALNINS* noted that the centrioles in porcine thoracic aortic endothelial. cells fixed in situ are preferentially located on the heart
 Proc. Nati. Acad. Sci. USA Vol. 82, pp. 3272-3276, May 1985 Cell Biology Preferential orientation of centrioles toward the heart in endothelial cells of major blood vessels is reestablished after reversal
Proc. Nati. Acad. Sci. USA Vol. 82, pp. 3272-3276, May 1985 Cell Biology Preferential orientation of centrioles toward the heart in endothelial cells of major blood vessels is reestablished after reversal
Bioen Lecture 28: Mechanotransduction. Biological Significance Examples of Mechanosensors
 Bioen 326 2014 Lecture 28: Mechanotransduction Biological Significance Examples of Mechanosensors 1 Mechanotransduction Conversion of mechanical signal to a biochemical signals Specialized cells sense
Bioen 326 2014 Lecture 28: Mechanotransduction Biological Significance Examples of Mechanosensors 1 Mechanotransduction Conversion of mechanical signal to a biochemical signals Specialized cells sense
Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function
 ONLINE DATA SUPPLEMENTS Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function Supplementary Figures Figure S1 Effect of Ad-p27-126TS on the expression
ONLINE DATA SUPPLEMENTS Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function Supplementary Figures Figure S1 Effect of Ad-p27-126TS on the expression
About This Chapter. Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Pearson Education, Inc.
 About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo
 Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
Phospholipase C γ Prof. Graham Carpenter
 Graham Carpenter, h.d. rofessor of Biochemistry Cornelia Crooke Department of Biochemistry Vanderbilt University School of Medicine, Nashville, TN 1 Receptor Tyrosine Kinases GF Extracellular M Intracellular
Graham Carpenter, h.d. rofessor of Biochemistry Cornelia Crooke Department of Biochemistry Vanderbilt University School of Medicine, Nashville, TN 1 Receptor Tyrosine Kinases GF Extracellular M Intracellular
Roles of Flow Mechanics in Vascular Cell Biology in Health and Disease
 Roles of Flow Mechanics in Vascular Cell Biology in Health and Disease Shu Chien Dept. of Bioengineering & Medicine UC, San Diego Presented by Ming-Shaung Ju Dept. of Mech. Eng., NCKU, Tainan Background
Roles of Flow Mechanics in Vascular Cell Biology in Health and Disease Shu Chien Dept. of Bioengineering & Medicine UC, San Diego Presented by Ming-Shaung Ju Dept. of Mech. Eng., NCKU, Tainan Background
Signaling. Dr. Sujata Persad Katz Group Centre for Pharmacy & Health research
 Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Hemodynamic forces have long been implicated in the
 Temporal Gradients in Shear, but Not Spatial Gradients, Stimulate Endothelial Cell Proliferation Charles R. White,* PhD; Mark Haidekker,* PhD; Xuping Bao, MD; John A. Frangos, PhD Background The effect
Temporal Gradients in Shear, but Not Spatial Gradients, Stimulate Endothelial Cell Proliferation Charles R. White,* PhD; Mark Haidekker,* PhD; Xuping Bao, MD; John A. Frangos, PhD Background The effect
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Muscle Tissue. General concepts. Classification of muscle. I. Functional classification is based on the type of neural control.
 Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
Muscle Tissue LEARNING OBJECTIVES 1. Identify the three types of muscle tissue at the light microscopic level. 2. List and compare the structural and functional features of each of the three muscle fiber
Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted
 Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted Ctns -/- mice. Cells immunolabeled for the proliferation marker (Ki-67) were counted in sections (n=3 WT, n=4
Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted Ctns -/- mice. Cells immunolabeled for the proliferation marker (Ki-67) were counted in sections (n=3 WT, n=4
Cell signaling. How do cells receive and respond to signals from their surroundings?
 Cell signaling How do cells receive and respond to signals from their surroundings? Prokaryotes and unicellular eukaryotes are largely independent and autonomous. In multicellular organisms there is a
Cell signaling How do cells receive and respond to signals from their surroundings? Prokaryotes and unicellular eukaryotes are largely independent and autonomous. In multicellular organisms there is a
ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY
 Nagoya J. Med. Sci. 72. 139 ~ 144, 1 ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY FENG XU, SATOKO ITO, MICHINARI HAMAGUCHI and TAKESHI
Nagoya J. Med. Sci. 72. 139 ~ 144, 1 ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY FENG XU, SATOKO ITO, MICHINARI HAMAGUCHI and TAKESHI
Organismic Biology Bio 207. Lecture 6. Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics. Prof.
 Organismic Biology Bio 207 Lecture 6 Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics Prof. Simchon Today s Agenda Skeletal muscle Neuro Muscular Junction
Organismic Biology Bio 207 Lecture 6 Muscle and movement; sliding filaments; E-C coupling; length-tension relationships; biomechanics Prof. Simchon Today s Agenda Skeletal muscle Neuro Muscular Junction
sfigure 1: Detection of L-fucose in normal mouse renal cortex using the plant lectin LTL
 sfigure 1: Detection of L-fucose in normal mouse renal cortex using the plant lectin LTL LTL staining Negative control Fluorescence microscopy of normal (CL-11 +/+ ) mouse renal tissue after staining with
sfigure 1: Detection of L-fucose in normal mouse renal cortex using the plant lectin LTL LTL staining Negative control Fluorescence microscopy of normal (CL-11 +/+ ) mouse renal tissue after staining with
Mechanical assessment by magnetocytometry of the cytosolic and cortical cytoskeletal compartments in adherent epithelial cells
 Biorheology 40 (2003) 235 240 235 IOS Press Mechanical assessment by magnetocytometry of the cytosolic and cortical cytoskeletal compartments in adherent epithelial cells Valérie M. Laurent, Emmanuelle
Biorheology 40 (2003) 235 240 235 IOS Press Mechanical assessment by magnetocytometry of the cytosolic and cortical cytoskeletal compartments in adherent epithelial cells Valérie M. Laurent, Emmanuelle
CVS HISTOLOGY. Dr. Nabil Khouri.
 CVS HISTOLOGY Dr. Nabil Khouri http://anatomy.kmu.edu.tw/blockhis/block3/slides/block4_24.html The Heart Wall Contract as a single unit Cardiac Muscle Simultaneous contraction due to depolarizing at the
CVS HISTOLOGY Dr. Nabil Khouri http://anatomy.kmu.edu.tw/blockhis/block3/slides/block4_24.html The Heart Wall Contract as a single unit Cardiac Muscle Simultaneous contraction due to depolarizing at the
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and
 Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Supplementary Figure 1: Signaling centers contain few proliferating cells, express p21, and exclude YAP from the nucleus. (a) Schematic diagram of an E10.5 mouse embryo. (b,c) Sections at B and C in (a)
Tissues. tissue = many cells w/ same structure and function. cell shape aids its function tissue shape aids its function
 Tissues tissue = many cells w/ same structure and function cell shape aids its function tissue shape aids its function Histology = study of tissues 4 types of tissues Epithelial coverings contact openings
Tissues tissue = many cells w/ same structure and function cell shape aids its function tissue shape aids its function Histology = study of tissues 4 types of tissues Epithelial coverings contact openings
Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq
 Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
Skeletal Muscle Contraction 4/11/2018 Dr. Hiwa Shafiq Skeletal Muscle Fiber About 40 per cent of the body is skeletal muscle, and 10 per cent is smooth and cardiac muscle. Skeletal muscles are composed
Muscle and Neuromuscular Junction. Peter Takizawa Department of Cell Biology
 Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
Muscle and Neuromuscular Junction Peter Takizawa Department of Cell Biology Types and structure of muscle cells Structural basis of contraction Triggering muscle contraction Skeletal muscle consists of
In Vitro Reendothelialization
 In Vitro Reendothelialization Microfilament Bundle Reorganization in Migrating Porcine Endothelial Cells Avrum I. Gotlieb, Wendy Spector, Michael K. K. Wong, and Carolyn Lacey An experimentally induced
In Vitro Reendothelialization Microfilament Bundle Reorganization in Migrating Porcine Endothelial Cells Avrum I. Gotlieb, Wendy Spector, Michael K. K. Wong, and Carolyn Lacey An experimentally induced
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering
 nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
nature methods Organelle-specific, rapid induction of molecular activities and membrane tethering Toru Komatsu, Igor Kukelyansky, J Michael McCaffery, Tasuku Ueno, Lidenys C Varela & Takanari Inoue Supplementary
8 - Muscular System. Introduction Taft College Human Physiology
 8 - Muscular System Introduction Taft College Human Physiology Muscular System - Introduction The bones provide the levers and structure of the skeleton but it is the muscles that cause movement. Motion
8 - Muscular System Introduction Taft College Human Physiology Muscular System - Introduction The bones provide the levers and structure of the skeleton but it is the muscles that cause movement. Motion
Supplements. Figure S1. B Phalloidin Alexa488
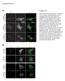 Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
Supplements A, DMSO, PP2, PP3 Crk-myc Figure S1. (A) Src kinase activity is necessary for recruitment of Crk to Nephrin cytoplasmic domain. Human podocytes expressing /7-NephrinCD () were treated with
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System
 Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale
 Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale (b) show stronger immunolabeling for CB 1 than those in
Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale (b) show stronger immunolabeling for CB 1 than those in
Signal Transduction: Information Metabolism. Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire
 Signal Transduction: Information Metabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire Introduction Information Metabolism How cells receive, process and respond
Signal Transduction: Information Metabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire Introduction Information Metabolism How cells receive, process and respond
Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were
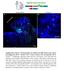 Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were electroporated with β- Catenin S33Y in PiggyBac expression
Supplementary Figure 1. Electroporation of a stable form of β-catenin causes masses protruding into the IV ventricle. HH12 chicken embryos were electroporated with β- Catenin S33Y in PiggyBac expression
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Muscle Cells & Muscle Fiber Contractions. Packet #8
 Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
The role of death-associated protein kinase in endothelial apoptosis under fluid shear stress
 Purdue University Purdue e-pubs Open Access Dissertations Theses and Dissertations Spring 2015 The role of death-associated protein kinase in endothelial apoptosis under fluid shear stress Keith R. Rennier
Purdue University Purdue e-pubs Open Access Dissertations Theses and Dissertations Spring 2015 The role of death-associated protein kinase in endothelial apoptosis under fluid shear stress Keith R. Rennier
Extra notes for lab- 1 histology. Slide 1 : cross section in the elastic artery ( aortic arch, ascending aorta, descending aorta )
 Extra notes for lab- 1 histology Slide 1 : cross section in the elastic artery ( aortic arch, ascending aorta, descending aorta ) - twin of ascending aorta is the pulmonary trunk. Ascending aorta represents
Extra notes for lab- 1 histology Slide 1 : cross section in the elastic artery ( aortic arch, ascending aorta, descending aorta ) - twin of ascending aorta is the pulmonary trunk. Ascending aorta represents
#1 20. physiology. Muscle tissue 30/9/2015. Ahmad Adel Sallal. Mohammad Qudah
 # 20 physiology Muscle tissue Ahmad Adel Sallal 30/9/205 Mohammad Qudah MUSCLES PHYSIOLOGY Awn, welcome to the first physiology lecture in the MSS, I wish you a perfect exams with high grades, and never
# 20 physiology Muscle tissue Ahmad Adel Sallal 30/9/205 Mohammad Qudah MUSCLES PHYSIOLOGY Awn, welcome to the first physiology lecture in the MSS, I wish you a perfect exams with high grades, and never
Muscle Dr. Ted Milner (KIN 416)
 Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Cardiovascular Division, Brigham and Women s Hospital, Harvard Medical School
 Low Endothelial Shear Stress Upregulates Atherogenic and Inflammatory Genes Extremely Early in the Natural History of Coronary Artery Disease in Diabetic Hyperlipidemic Juvenile Swine Michail I. Papafaklis,
Low Endothelial Shear Stress Upregulates Atherogenic and Inflammatory Genes Extremely Early in the Natural History of Coronary Artery Disease in Diabetic Hyperlipidemic Juvenile Swine Michail I. Papafaklis,
Novel roles for ICAM1 in leukocyte transendothelial migration. Michael John Allingham
 Novel roles for ICAM1 in leukocyte transendothelial migration Michael John Allingham A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of
Novel roles for ICAM1 in leukocyte transendothelial migration Michael John Allingham A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of
Rearrangements of the Actin Cytoskeleton and E-Cadherin Based Adherens Junctions Caused by Neoplasic Transformation Change Cell Cell Interactions
 Rearrangements of the Actin Cytoskeleton and E-Cadherin Based Adherens Junctions Caused by Neoplasic Transformation Change Cell Cell Interactions Dmitry V. Ayollo., Irina Y. Zhitnyak., Jury M. Vasiliev,
Rearrangements of the Actin Cytoskeleton and E-Cadherin Based Adherens Junctions Caused by Neoplasic Transformation Change Cell Cell Interactions Dmitry V. Ayollo., Irina Y. Zhitnyak., Jury M. Vasiliev,
Effects of Second Messengers
 Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade)
 HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
HOMEWORK RUBRICS MECHANOTRANSDUCTION UNIT: HOMEWORK #1 (20 pts towards your grade) 1. Mesenchymal stem cells (MSC) cultured on extracellular matrices with different stiffness exhibit diverse lineage commitment
Overexpression of the focal adhesion kinase (p125 FAK ) in the vascular smooth muscle cells of intimal hyperplasia
 Overexpression of the focal adhesion kinase (p125 FAK ) in the vascular smooth muscle cells of intimal hyperplasia Lewis V. Owens, MD, b LiHui Xu, MD, d William A. Marston, MD, b XiHui Yang, BS, d Mark
Overexpression of the focal adhesion kinase (p125 FAK ) in the vascular smooth muscle cells of intimal hyperplasia Lewis V. Owens, MD, b LiHui Xu, MD, d William A. Marston, MD, b XiHui Yang, BS, d Mark
Supplementary Figure 1. Mother centrioles can reduplicate while in the close association
 C1-GFP distance (nm) C1-GFP distance (nm) a arrested HeLa cell expressing C1-GFP and Plk1TD-RFP -3 s 1 2 3 4 5 6 7 8 9 11 12 13 14 16 17 18 19 2 21 22 23 24 26 27 28 29 3 b 9 8 7 6 5 4 3 2 arrested HeLa
C1-GFP distance (nm) C1-GFP distance (nm) a arrested HeLa cell expressing C1-GFP and Plk1TD-RFP -3 s 1 2 3 4 5 6 7 8 9 11 12 13 14 16 17 18 19 2 21 22 23 24 26 27 28 29 3 b 9 8 7 6 5 4 3 2 arrested HeLa
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Chapter Skeletal Muscle Structure and Function
 Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
Cell Communication. Cell Communication. Cell Communication. Cell Communication. Cell Communication. Chapter 9. Communication between cells requires:
 Chapter 9 Communication between cells requires: ligand: the signaling molecule receptor protein: the molecule to which the receptor binds -may be on the plasma membrane or within the cell 2 There are four
Chapter 9 Communication between cells requires: ligand: the signaling molecule receptor protein: the molecule to which the receptor binds -may be on the plasma membrane or within the cell 2 There are four
Neuronal plasma membrane
 ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
Elastic Skeleton of Intracranial Cerebral Aneurysms in Rats
 1722 Elastic Skeleton of Intracranial Cerebral Aneurysms in Rats Naohiro Yamazoe, MD, Nobuo Hashimoto, MD, Haruhiko Kikuchi, MD, and Fumitada Hazama, MD In an attempt to clarify the developmental mechanism
1722 Elastic Skeleton of Intracranial Cerebral Aneurysms in Rats Naohiro Yamazoe, MD, Nobuo Hashimoto, MD, Haruhiko Kikuchi, MD, and Fumitada Hazama, MD In an attempt to clarify the developmental mechanism
Cell Motility. Junfeng Ji ( 纪俊峰 ), PhD
 Cell Motility Junfeng Ji ( 纪俊峰 ), PhD Professor Laboratory of Somatic Reprogramming and Stem Cell Aging Center for Stem Cell and Tissue Engineering School of Medicine Zhejiang University Email: jijunfeng@zju.edu.cnedu
Cell Motility Junfeng Ji ( 纪俊峰 ), PhD Professor Laboratory of Somatic Reprogramming and Stem Cell Aging Center for Stem Cell and Tissue Engineering School of Medicine Zhejiang University Email: jijunfeng@zju.edu.cnedu
F-actin VWF Vinculin. F-actin. Vinculin VWF
 a F-actin VWF Vinculin b F-actin VWF Vinculin Supplementary Fig. 1. WPBs in HUVECs are located along stress fibers and at focal adhesions. (a) Immunofluorescence images of f-actin (cyan), VWF (yellow),
a F-actin VWF Vinculin b F-actin VWF Vinculin Supplementary Fig. 1. WPBs in HUVECs are located along stress fibers and at focal adhesions. (a) Immunofluorescence images of f-actin (cyan), VWF (yellow),
Src-INACTIVE / Src-INACTIVE
 Biology 169 -- Exam 1 February 2003 Answer each question, noting carefully the instructions for each. Repeat- Read the instructions for each question before answering!!! Be as specific as possible in each
Biology 169 -- Exam 1 February 2003 Answer each question, noting carefully the instructions for each. Repeat- Read the instructions for each question before answering!!! Be as specific as possible in each
Comptes Rendus Physique
 C. R. Physique 14 (2013) 479 496 Contents lists available at SciVerse ScienceDirect Comptes Rendus Physique www.sciencedirect.com Living fluids/fluides vivants Blood flow and arterial endothelial dysfunction:
C. R. Physique 14 (2013) 479 496 Contents lists available at SciVerse ScienceDirect Comptes Rendus Physique www.sciencedirect.com Living fluids/fluides vivants Blood flow and arterial endothelial dysfunction:
Chapter 11 Intercellular Communication and Tissue Architecture
 Part III Organization of Cell Populations Chapter 11 Multicellular organisms such as the human body consist of various tissues such as epithelial tissues, bones and nerves, and organs such as heart and
Part III Organization of Cell Populations Chapter 11 Multicellular organisms such as the human body consist of various tissues such as epithelial tissues, bones and nerves, and organs such as heart and
Neuronal plasma membrane
 ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
Outline. Bio 105: Muscular System. Muscular System. Types of Muscles. Smooth Muscle. Cardiac Muscle 4/6/2016
 Outline Bio 105: Muscular System Lecture 11 Chapter 6 Characteristics of muscles 3 types of muscles Functions of muscles Structure of skeletal muscles Mechanics of muscle contraction Energy sources for
Outline Bio 105: Muscular System Lecture 11 Chapter 6 Characteristics of muscles 3 types of muscles Functions of muscles Structure of skeletal muscles Mechanics of muscle contraction Energy sources for
Integrin mechanotransduction stimulates caveolin-1 phosphorylation and recruitment of Csk to mediate actin reorganization
 Am J Physiol Heart Circ Physiol 288: H936 H945, 2005. First published October 7, 2004; doi:10.1152/ajpheart.00519.2004. Integrin mechanotransduction stimulates caveolin-1 phosphorylation and recruitment
Am J Physiol Heart Circ Physiol 288: H936 H945, 2005. First published October 7, 2004; doi:10.1152/ajpheart.00519.2004. Integrin mechanotransduction stimulates caveolin-1 phosphorylation and recruitment
Cell Cell Communication
 IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
Muscular Tissue. Functions of Muscular Tissue. Types of Muscular Tissue. Skeletal Muscular Tissue. Properties of Muscular Tissue
 Muscular Tissue Functions of Muscular Tissue Muscle makes up a large percentage of the body s weight (40-50%) Their main functions are to: Create motion muscles work with nerves, bones, and joints to produce
Muscular Tissue Functions of Muscular Tissue Muscle makes up a large percentage of the body s weight (40-50%) Their main functions are to: Create motion muscles work with nerves, bones, and joints to produce
Arterial Branching in Man and Monkey
 Published Online: 1 March, 1982 Supp Info: http://doi.org/10.1085/jgp.79.3.353 Downloaded from jgp.rupress.org on December 13, 2018 Arterial Branching in Man and Monkey M. ZAMIR and J. A. MEDEIROS From
Published Online: 1 March, 1982 Supp Info: http://doi.org/10.1085/jgp.79.3.353 Downloaded from jgp.rupress.org on December 13, 2018 Arterial Branching in Man and Monkey M. ZAMIR and J. A. MEDEIROS From
Muscle tissues. Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD
 Muscle tissues Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD Muscle tissue is a soft tissue that composes muscles in animal bodies, and gives rise to muscles' ability to contract. Muscle tissue
Muscle tissues Dr. Hersh Abdul Ham-Karim BVM&S, PG Dip, MSc and PhD Muscle tissue is a soft tissue that composes muscles in animal bodies, and gives rise to muscles' ability to contract. Muscle tissue
Cell Cell Communication
 IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
IBS 8102 Cell, Molecular, and Developmental Biology Cell Cell Communication January 29, 2008 Communicate What? Why do cells communicate? To govern or modify each other for the benefit of the organism differentiate
(d) are made mainly of lipids and of proteins that lie like thin sheets on the membrane surface
 Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
Nature Methods: doi: /nmeth.4257
 Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb /DC1
 Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued
 Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
Cell structure of Eukaryotic cells Lecture 6 9/17 Dr. Hirsh Organization of Cells, continued Lots of double-membraned organelles Existence of an Endo-membrane system separation of areas of cell, transport
Protein kinases are enzymes that add a phosphate group to proteins according to the. ATP + protein OH > Protein OPO 3 + ADP
 Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Chapter 7. (7-1 and 7-2) A Tour of the Cell
 Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
Arterial enlargement, tortuosity, and intimal thickening in response to sequential exposure to high and low wall shear stress
 BASIC RESEARCH STUDIES Arterial enlargement, tortuosity, and intimal thickening in response to sequential exposure to high and low wall shear stress Eiketsu Sho, MD, PhD, a,b Hiroshi Nanjo, MD, PhD, a
BASIC RESEARCH STUDIES Arterial enlargement, tortuosity, and intimal thickening in response to sequential exposure to high and low wall shear stress Eiketsu Sho, MD, PhD, a,b Hiroshi Nanjo, MD, PhD, a
Influenza virus exploits tunneling nanotubes for cell-to-cell spread
 Supplementary Information Influenza virus exploits tunneling nanotubes for cell-to-cell spread Amrita Kumar 1, Jin Hyang Kim 1, Priya Ranjan 1, Maureen G. Metcalfe 2, Weiping Cao 1, Margarita Mishina 1,
Supplementary Information Influenza virus exploits tunneling nanotubes for cell-to-cell spread Amrita Kumar 1, Jin Hyang Kim 1, Priya Ranjan 1, Maureen G. Metcalfe 2, Weiping Cao 1, Margarita Mishina 1,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Nanoneedle-Mediated Stimulation of Cell Mechanotransduction Machinery Catherine S Hansel, Spencer W Crowder, Samuel Cooper, Sahana Gopal, Maria João Pardelha da Cruz, Leonardo
SUPPLEMENTARY INFORMATION Nanoneedle-Mediated Stimulation of Cell Mechanotransduction Machinery Catherine S Hansel, Spencer W Crowder, Samuel Cooper, Sahana Gopal, Maria João Pardelha da Cruz, Leonardo
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture
 Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Skeletal muscle. General features :
 Muscular tissues In the first embryonic life the muscular tissues arise from mesoderm, The function of movement in multicellular organisms is usually assumed by specialized cells called muscle fibers which
Muscular tissues In the first embryonic life the muscular tissues arise from mesoderm, The function of movement in multicellular organisms is usually assumed by specialized cells called muscle fibers which
Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions.
 Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions. There are several important themes that transcends just the chemistry and bring the
Lecture 1, Fall 2014 The structure of the eukaryotic cell, as it relates to cells chemical and biological functions. There are several important themes that transcends just the chemistry and bring the
Chapter 6: A Tour of the Cell. 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures
 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
1. Studying Cells. Concepts of Microscopy 11/7/2016. Chapter 6: A Tour of the Cell
 Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
PECAM-1 Mediates NO-Dependent Dilation of Arterioles to High Temporal Gradients of Shear Stress
 PECAM-1 Mediates NO-Dependent Dilation of Arterioles to High Temporal Gradients of Shear Stress Zsolt Bagi, John A. Frangos, Jiunn-Chern Yeh, Charles R. White, Gabor Kaley, Akos Koller Objective In response
PECAM-1 Mediates NO-Dependent Dilation of Arterioles to High Temporal Gradients of Shear Stress Zsolt Bagi, John A. Frangos, Jiunn-Chern Yeh, Charles R. White, Gabor Kaley, Akos Koller Objective In response
Cochlear anatomy, function and pathology III. Professor Dave Furness Keele University
 Cochlear anatomy, function and pathology III Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of this lecture Focus (3) on the cochlear lateral wall and Reissner s membrane:
Cochlear anatomy, function and pathology III Professor Dave Furness Keele University d.n.furness@keele.ac.uk Aims and objectives of this lecture Focus (3) on the cochlear lateral wall and Reissner s membrane:
Chapter 4: Cell Structure and Function
 Chapter 4: Cell Structure and Function Robert Hooke Fig. 4-2, p.51 The Cell Smallest unit of life Can survive on its own or has potential to do so Is highly organized for metabolism Senses and responds
Chapter 4: Cell Structure and Function Robert Hooke Fig. 4-2, p.51 The Cell Smallest unit of life Can survive on its own or has potential to do so Is highly organized for metabolism Senses and responds
Sodium and cell rigidity
 Sodium and cell rigidity Hans Oberleithner Institute of Physiology II University Münster, Germany Calcium and the cytoskeleton, Minisymposium, Casapueblo, November 3, 2007; 11:30 a.m. This is what we need
Sodium and cell rigidity Hans Oberleithner Institute of Physiology II University Münster, Germany Calcium and the cytoskeleton, Minisymposium, Casapueblo, November 3, 2007; 11:30 a.m. This is what we need
Histology of the Cardiac System. Dr. Nabil Khoury Anatomy Department
 Histology of the Cardiac System Dr. Nabil Khoury Anatomy Department Objectives 1. Identify the 3 layers of the heart endocardium, myocardium, epicardium 2. Differentiate cardiacmuscle 3. Define intercalated
Histology of the Cardiac System Dr. Nabil Khoury Anatomy Department Objectives 1. Identify the 3 layers of the heart endocardium, myocardium, epicardium 2. Differentiate cardiacmuscle 3. Define intercalated
