Unit 1: Science of Life 1. Define the following terms: Hypothesis: Testable explanation for a phenomenon
|
|
|
- Christian Cannon
- 5 years ago
- Views:
Transcription
1 UCS BIOLOGY STUDY GUIDE FOR 1 ST SEMESTER MIDTERM EXAM Unit 1: Science of Life 1. Define the following terms: Hypothesis: Testable explanation for a phenomenon Experiment: an orderly procedure carried out with the goal of verifying, refuting, or establishing the validity of a hypothesis. Controlled experiments provide insight into cause-and-effect by demonstrating what outcome occurs when a particular factor is manipulated. Theory : an explanation of a scientific phenomenon that has been tested by many scientists over many years Homeostasis: Maintaining an internal balance regardless of external conditions Observation: Examining something carefully to gain information Peer review: the evaluation of work by one or more people of similar competence to the producers of the work Characteristics of Life: Metabolism, composed of cells, growth and development, change over time, homeostasis, response to stimuli, reproduction
2 2. What is the order of structures in living organisms from the simplest to the most complex (order of organization)? Cells, tissues, organs, organ systems, organisms Unit 2: Metabolism 3. What is the one element that is found in all living things? Carbon (organic compounds) 4. What are the 4 most common elements in all living things? Carbon, hydrogen, oxygen, and nitrogen 5. Where is energy stored in a molecule? Carbon-hydrogen bonds 6. How is energy in a molecule released? Break the bonds hydrolysis (adding water to break bonds)
3 7. How is the energy in a molecule of ATP released? 8. What are enzymes? What is their function? How do enzymes maintain the homeostasis of living things? Proteins that are biological catalysts that speed up chemical reactions by lowering activation energy
4 9. What are the structural differences between macromolecules? Nucleic acids Carbs Lipids
5 10. What are the monomers and polymers of carbohydrates? Monosaccharide Polysaccharide 11. What are the monomers and polymers of lipids? Fatty Acids Saturated and unsaturated fats 12. What are the monomers and polymers of proteins? Amino Acids Proteins 13. What is the purpose of lipids in the animal body? Fats, oils, waxes, hormones Unit 3: Cells 14. Define organelle. a specialized subunit within a cell that has a specific function 15. Describe the functions of the nucleus, ER, Golgi apparatus, cell membrane, cell wall, mitochondria, chloroplasts, central vacuole and ribosomes. Nucleus: Contain genetic material
6 ER: process proteins Golgi apparatus: package materials Cell membrane: control what goes in and out of the cell Cell wall: Protects the cell, keeps its shape Mitochondria: cellular respiration (provide energy for the cell) Chloroplasts: photosynthesis Central vacuole: Water and nutrient storage Ribosomes: protein factory 16. Describe the cell membrane and its functions. The cell membrane separates the contents of the cell from its environment. Composed of mostly of lipids. Controls what goes in and out of the cell (security guard) 17. Differentiate between animal and plant cell structures. Plant cells have a cell wall, chloroplasts, and a large central (permanent) vacuole. Both have a cell membrane, nucleus, and membrane bound organelles.
7 18. Compare and contrast chloroplasts and mitochondria (include processes and DNA). Mitochondria: cellular respiration- provides energy for the cell Chloroplasts: photosynthesis- makes food for the cell (glucose) Both contain DNA because they originated as prokaryotes that became a part of a larger cell.
8 19. What is a prokaryote? Single-celled organism with free floating DNA because they do not have a nucleus. Bacteria are prokaryotes 20. Compare the structures of prokaryotic and eukaryotic cells. 21. What is a virus? Non-living infectious agent. Requires a host cell to replicate. Does not carry out metabolism.
9 22. What are the 2 main structures each virus contains? Protein coat and genetic material (DNA or RNA) 23. Compare the main structures and nucleic acids in bacteria and viruses. Virus: protein coat & genetic material Bacteria: Unit 4: Cellular Respiration and Photosynthesis 24. What is the source of all energy on Earth? The Sun!
10 25. What is the chemical equation for photosynthesis? 26. What are the 2 reactants needed for photosynthesis to take place? Reactants (before the arrow) = carbon dioxide and water 27. How do plants obtain these needed 2 reactants into their systems? Carbon dioxide from the air and H2O from the ground through the roots 28. Explain the function of the guard cells, stomata, and roots.
11 Stomata: gas exchange Guard cells: open and close the stomata Roots: anchor the plant and get water from the soil 29. What is the chemical equation for cellular respiration? 30. What is the purpose of cellular respiration? To create ATP and store the energy found in the bond of glucose and transfers it to the bonds of ATP. The energy currency of the cell. 31. What is the purpose of ATP? ATP is the high-energy molecule of the cell. The bonds of ATP are broken to convert the chemical energy stored in mechanical energy for movement. 32. What do cells breakdown during cellular respiration in order to produce ATP? Glucose!
12 33. Compare the chemical equations for photosynthesis and cellular respiration. (products/reactant relationships) 34. Explain how photosynthesis and cellular respiration are interrelated. 35. Why do plants have chloroplasts and mitochondria? Plants must make their food (glucose) in chloroplasts that can then be used in cellular respiration to make ATP to power the cells in the mitochondria.
13 Unit 5: Body Systems and Homeostasis 36. Explain how different organisms accomplish the same result using different specializations (gills vs. lungs vs. membranes, skeletal (endoskeleton vs. exoskeleton), respiratory (Oxygen absorption). 37. What is the main function of the human cardiovascular system? To transport oxygen and nutrients to your cells and transport CO2 and materials to be excreted
14 38. What is the main function of the muscular system? To contract and relax for movement. 39. What is the main function of the digestive system? To break down food into nutrients that can be absorbed by the blood stream and used for cellular respiration and growth. 40. How does the nervous system send and receive messages to the muscular system (reflexes)? Through connections between nerve cells that send an impulse around the body. The brain sends an impulse through nerves to your muscle to make them move (contract) 41. How does gas exchange occur in the lungs? In the alveoli. Gas diffuses across the membrane to and from the capillaries
15 42. How does each human body system help our bodies maintain homeostasis? Go over body systems graphic organizer 43. How do the human body systems work together to maintain homeostasis? Go over body systems graphic organizer
Renaissance Biology Midterm Study Guide Answers
 Renaissance Biology Midterm Study Guide Answers 2016-2017 LEARNING TARGET 1: List the characteristics of life Made of one or more cells Organization cells -> tissues -> organs -> organ systems -> organisms
Renaissance Biology Midterm Study Guide Answers 2016-2017 LEARNING TARGET 1: List the characteristics of life Made of one or more cells Organization cells -> tissues -> organs -> organ systems -> organisms
Unit 2 - Characteristics of Living Things
 Living Environment Answer Key to Practice Exam- Parts A and B-1 1. A fully functioning enzyme molecule is arranged in a complex three-dimensional shape. This shape determines the A) specific type of molecule
Living Environment Answer Key to Practice Exam- Parts A and B-1 1. A fully functioning enzyme molecule is arranged in a complex three-dimensional shape. This shape determines the A) specific type of molecule
Macromolcules, Enzymes, & Cells Intro
 Name: Date: 1. The distortion (change in shape) of enzyme molecules which occurs at high temperatures is known as 5. A characteristic shared by all enzymes, hormones, and antibodies is that their function
Name: Date: 1. The distortion (change in shape) of enzyme molecules which occurs at high temperatures is known as 5. A characteristic shared by all enzymes, hormones, and antibodies is that their function
***Non-living things may show one or more of these Characteristics, but NEVER ALL of them
 -Living things are highly ORGANIZED -Living things are made up of one or more CELLS -Living things use ENERGY -Living things GROW and develop -Living things RESPOND to changes in the environment -Living
-Living things are highly ORGANIZED -Living things are made up of one or more CELLS -Living things use ENERGY -Living things GROW and develop -Living things RESPOND to changes in the environment -Living
Mid Term Review. 1. step 1, only 3. both step 1 and step 2 2. step 2, only 4. neither step 1 nor step 2
 Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
Test Review Worksheet 1 Name: Per:
 Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
Directions: Use Aim # 7-13 (Unit 2) to complete this study guide. Part I. Aim # 7 Inorganic and Organic Molecules. Building Blocks (monomers)
 Name: Date: Period: Living Environment Unit 2 Biochemistry and The Cell Study Guide Due Date: Test Date: Unit 2 Important Topics: Aim # 7: Inorganic and Organic Molecules Aim # 8: Organic Macromolecules
Name: Date: Period: Living Environment Unit 2 Biochemistry and The Cell Study Guide Due Date: Test Date: Unit 2 Important Topics: Aim # 7: Inorganic and Organic Molecules Aim # 8: Organic Macromolecules
Introduction to Biochemistry
 Life is Organized in Increasing Levels of Complexity Introduction to Biochemistry atom simple molecule What is the chemical makeup of living things? macromolecule organ organ system organism organelle
Life is Organized in Increasing Levels of Complexity Introduction to Biochemistry atom simple molecule What is the chemical makeup of living things? macromolecule organ organ system organism organelle
Macromolecules Chapter 2.3
 Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
Macromolecules Chapter 2.3 E.Q. What are the 4 main macromolecues found in living things and what are their functions? Carbon-Based Molecules Why is carbon called the building block of life? Carbon atoms
Cells & Cell Organelles
 Cells & Cell Organelles The Building Blocks of Life AP Biology 2008-2009 Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal
Cells & Cell Organelles The Building Blocks of Life AP Biology 2008-2009 Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal
Copyright 2014 Edmentum - All rights reserved.
 Study Island Copyright 2014 Edmentum - All rights reserved. Generation Date: 04/01/2014 Generated By: Cheryl Shelton Title: Science- biology Cells 1. Below is an image of a plant cell. What processes require
Study Island Copyright 2014 Edmentum - All rights reserved. Generation Date: 04/01/2014 Generated By: Cheryl Shelton Title: Science- biology Cells 1. Below is an image of a plant cell. What processes require
1. Structure A is the a. Cell wall b. Cell membrane c. Vacuole d. Lysosome
 Figure 1 Use Figure 1 to answer the following questions: 1. Structure A is the a. Cell wall b. Cell membrane c. Vacuole d. Lysosome 2. Structure E controls cellular functions. It is the a. Nucleolus b.
Figure 1 Use Figure 1 to answer the following questions: 1. Structure A is the a. Cell wall b. Cell membrane c. Vacuole d. Lysosome 2. Structure E controls cellular functions. It is the a. Nucleolus b.
BALSIGER BIOLOGY STUDY GUIDES (SEMESTER 1)
 BALSIGER BIOLOGY STUDY GUIDES (SEMESTER 1) Table of Contents 1 III. Parts of an Experiment 1) Write a brief definition for each of these components of an experiment: manipulated variable, treatments, experimental
BALSIGER BIOLOGY STUDY GUIDES (SEMESTER 1) Table of Contents 1 III. Parts of an Experiment 1) Write a brief definition for each of these components of an experiment: manipulated variable, treatments, experimental
Macromolecules. SC.912.L.18.1 Describe the basic molecular structures and primary functions of the four major categories of biological macromolecules.
 Macromolecules SC.912.L.18.1 Describe the basic molecular structures and primary functions of the four major categories of biological macromolecules. FOUR MAJOR BIOLOGICAL MACROMOLECULES 1.Carbohydrates
Macromolecules SC.912.L.18.1 Describe the basic molecular structures and primary functions of the four major categories of biological macromolecules. FOUR MAJOR BIOLOGICAL MACROMOLECULES 1.Carbohydrates
Cells & Cell Organelles
 Cells & Cell Organelles Doing Life s Work 2009 2010 1 Types of cells bacteria cells Prokaryote no organelles animal cells Eukaryotes organelles plant cells 2 Cell size comparison Animal cell Bacterial
Cells & Cell Organelles Doing Life s Work 2009 2010 1 Types of cells bacteria cells Prokaryote no organelles animal cells Eukaryotes organelles plant cells 2 Cell size comparison Animal cell Bacterial
Organic Compounds. B-3.5 Students will be able to summarize the functions of proteins, carbohydrates, and fats in the human body.
 Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
C) amount of carbon dioxide absorbed by the animal B) rate of respiration of the animal
 Name: 1) A model of a section of a cell membrane is represented below. 4034-1 - Page 1 Which type of molecule is indicated by the arrow? A) carbohydrate B) protein C) lipid D) nucleotide 2) The movement
Name: 1) A model of a section of a cell membrane is represented below. 4034-1 - Page 1 Which type of molecule is indicated by the arrow? A) carbohydrate B) protein C) lipid D) nucleotide 2) The movement
The building blocks for this molecule are A) amino acids B) simple sugars C) fats D) molecular bases
 1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
To be able to answer a question
 1. State Problem or question 2. Gather information 3. State a hypothesis 4. Conduct Experiment 5. Observe, collect, & analyze data 6. State a conclusion7. Repeat many times Observation =Recognizing a FACT
1. State Problem or question 2. Gather information 3. State a hypothesis 4. Conduct Experiment 5. Observe, collect, & analyze data 6. State a conclusion7. Repeat many times Observation =Recognizing a FACT
2.3: Carbon-Based Molecules Notes
 2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
2.3: Carbon-Based Molecules Notes Carbon-based molecules are the of life. Bonding Properties of Carbon Carbon forms bonds with up to other atoms, including other carbon atoms. QUESTION: What types of elements
A. There are about 100 elements; 25 of them are necessary for life. B. Carbon atoms can form long chains, leading to a huge number of possible
 Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
Ch. 2 How Cells Function 2.1 Chemical reactions take place inside cells. 1. All cells are made of the same elements. A. There are about 100 elements; 25 of them are necessary for life. B. The smallest
2. Eukaryote plant, animal, protozoa and fungal cells; complex and organized; organelles; true nucleus
 BIOLOGY TERMS AND CONCEPTS TO KNOW: 2010-2011. Prokaryote unicellular organism; bacteria; lacks a true nucleus but does contain dna; cell membrane and often a flage 2. Eukaryote plant, animal, protozoa
BIOLOGY TERMS AND CONCEPTS TO KNOW: 2010-2011. Prokaryote unicellular organism; bacteria; lacks a true nucleus but does contain dna; cell membrane and often a flage 2. Eukaryote plant, animal, protozoa
Ms. Golub & Ms. Sahar Date: Unit 2- Test #1
 Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
The Structure and Function of Biomolecules
 The Structure and Function of Biomolecules The student is expected to: 9A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic
The Structure and Function of Biomolecules The student is expected to: 9A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic
Jeopardy Q $100 Q $100 Q $100 Q $100 Q $100 Q $200 Q $200 Q $200 Q $200 Q $200 Q $300 Q $300 Q $300 Q $300 Q $300 Q $400 Q $400 Q $400 Q $400 Q $400
 Jeopardy Proteins Carbohydrates Lipids Nucleic Acids Energy & Reactions Q $100 Q $200 Q $300 Q $400 Q $500 Q $100 Q $100 Q $100 Q $100 Q $200 Q $200 Q $200 Q $200 Q $300 Q $300 Q $300 Q $300 Q $400 Q $400
Jeopardy Proteins Carbohydrates Lipids Nucleic Acids Energy & Reactions Q $100 Q $200 Q $300 Q $400 Q $500 Q $100 Q $100 Q $100 Q $100 Q $200 Q $200 Q $200 Q $200 Q $300 Q $300 Q $300 Q $300 Q $400 Q $400
CP Biology Chapter 2: Molecules of Life Name Amatuzzi #1: Carbohydrates pp Period Homework
 Amatuzzi #1: Carbohydrates pp. 46-47 Period 1. Which elements make up carbohydrates? a. In which ratio? 2. How do living things use most of their carbohydrates? 3. How do cells get energy from carbs? a.
Amatuzzi #1: Carbohydrates pp. 46-47 Period 1. Which elements make up carbohydrates? a. In which ratio? 2. How do living things use most of their carbohydrates? 3. How do cells get energy from carbs? a.
Chapter 3 CELL PROCESSES AND ENERGY
 Chapter 3 CELL PROCESSES AND ENERGY Section 1: Chemical Compounds in Cells Elements= Any substance that cannot be broken down into a simpler form Made up of only one kind of atom Found in the body Carbon
Chapter 3 CELL PROCESSES AND ENERGY Section 1: Chemical Compounds in Cells Elements= Any substance that cannot be broken down into a simpler form Made up of only one kind of atom Found in the body Carbon
Molecule - two or more atoms held together by covalent bonds. Ex. = water, H O
 ORGANIC CHEMISTRY NOTES Why study carbon? ORGANIC CHEMISTRY NOTES Why study carbon? * All of life is built on carbon * Cells are made up of about 72% water 3% salts (NaCl, and K) 25% carbon compounds which
ORGANIC CHEMISTRY NOTES Why study carbon? ORGANIC CHEMISTRY NOTES Why study carbon? * All of life is built on carbon * Cells are made up of about 72% water 3% salts (NaCl, and K) 25% carbon compounds which
Standard 2 Exam Biology. 2. This macromolecule is responsible for short term energy storage and structural support in plants
 1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
CELLS CELL THEORY. There are two types of cells: 9/26/2016 CELLULAR COMPONENTS & PROCESES. Which is more complicated? REMEMBER YOU ARE EUKARYOTIC!
 CELLS CELLULAR COMPONENTS & PROCESES CELL THEORY 1. All living things are made up of one or more cells 2. The cell is the smallest unit of life. 3. All new cells come from pre-existing cells. There are
CELLS CELLULAR COMPONENTS & PROCESES CELL THEORY 1. All living things are made up of one or more cells 2. The cell is the smallest unit of life. 3. All new cells come from pre-existing cells. There are
Biology SAGE Review Standard dos
 Biology SAGE Review Standard dos Name Period 1-7 Main Idea How I will prove my infinite knowledge The major chemical elements that compose cells have an acronym: CHNOPS (carbon, hydrogen, nitrogen, oxygen,
Biology SAGE Review Standard dos Name Period 1-7 Main Idea How I will prove my infinite knowledge The major chemical elements that compose cells have an acronym: CHNOPS (carbon, hydrogen, nitrogen, oxygen,
Living Environment. Scientific Inquiry Exam
 Name: Class: 1. Which elements are present in all organic compounds? 1) nitrogen and carbon 3) hydrogen and oxygen 2) nitrogen and oxygen 4) hydrogen and carbon 2. Which substances are inorganic compounds?
Name: Class: 1. Which elements are present in all organic compounds? 1) nitrogen and carbon 3) hydrogen and oxygen 2) nitrogen and oxygen 4) hydrogen and carbon 2. Which substances are inorganic compounds?
Cells are the smallest units of life CH 3
 Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
1. Which of the following structures is not found in bacteria?
 Untitled Document EOC Macromolecules 1. Which of the following structures is not found in bacteria? A. ribosome B. cytoplasm C. cell membrane D. nuclear membrane 4. Plants and animals are composed of organic
Untitled Document EOC Macromolecules 1. Which of the following structures is not found in bacteria? A. ribosome B. cytoplasm C. cell membrane D. nuclear membrane 4. Plants and animals are composed of organic
Chemical Formulas. Chemical Formula CH 3 COCHCHOCHClCHNH Lewis Dot Structure
 Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
3. Describe the study in mimicry, using king snakes and coral snakes. Identify the control in the experiment.
 Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
EQ What are the 4 biomolecules and their monomers?
 EQ What are the 4 biomolecules and their monomers? Monomers: Fatty Acids Amino acids Nucleotides Monosaccharides Drawings Chemistry of Life Chapter 3 Biomolecules Chapter 3 Section 3 Building Blocks of
EQ What are the 4 biomolecules and their monomers? Monomers: Fatty Acids Amino acids Nucleotides Monosaccharides Drawings Chemistry of Life Chapter 3 Biomolecules Chapter 3 Section 3 Building Blocks of
10. The diagram below shows two different kinds of substances, A and B, entering a cell.
 1. In the binomial system of nomenclature, which two classification groups provide the scientific name of an organism? A) kingdom and phylum B) phylum and species C) kingdom and genus D) genus and species
1. In the binomial system of nomenclature, which two classification groups provide the scientific name of an organism? A) kingdom and phylum B) phylum and species C) kingdom and genus D) genus and species
Digestive and Excretory Systems
 Digestive and Excretory Systems Homeostasis Q: How are the materials that enter and leave your body related to the processes that maintain homeostasis? 30.1 How is the human body organized and regulated?
Digestive and Excretory Systems Homeostasis Q: How are the materials that enter and leave your body related to the processes that maintain homeostasis? 30.1 How is the human body organized and regulated?
Do Now Makeups. 4. In which organelle would water and dissolved materials be stored? A. 1 B. 2 C. 3 D. 5. A. mitochondria B.
 Do Now Makeups Name: Date: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell? A. mitochondria B. centrosomes
Do Now Makeups Name: Date: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell? A. mitochondria B. centrosomes
Biochemistry. 2. Besides carbon, name 3 other elements that make up most organic compounds.
 Biochemistry Carbon compounds Section 3-1 1. What is an organic compound? 2. Besides carbon, name 3 other elements that make up most organic compounds. 3. Carbon dioxide, CO 2, is NOT an organic compound.
Biochemistry Carbon compounds Section 3-1 1. What is an organic compound? 2. Besides carbon, name 3 other elements that make up most organic compounds. 3. Carbon dioxide, CO 2, is NOT an organic compound.
High School Science MCA Item Sampler Teacher Guide
 High School Science MCA Item Sampler Teacher Guide Overview of Item Samplers Item samplers are one type of student resource provided to help students and educators prepare for test administration. While
High School Science MCA Item Sampler Teacher Guide Overview of Item Samplers Item samplers are one type of student resource provided to help students and educators prepare for test administration. While
Cells and Osmosis BCT Questions. Questions taken from 2-12 to 2-18
 Cells and Osmosis BCT Questions Questions taken from 2-12 to 2-18 1. Which of the following would be least affected by defective receptor proteins on a cell membrane? a. Homeostasis b. Muscle activity
Cells and Osmosis BCT Questions Questions taken from 2-12 to 2-18 1. Which of the following would be least affected by defective receptor proteins on a cell membrane? a. Homeostasis b. Muscle activity
Practice Midterm» Form A (Master Copy)» Teacher Version
 Directions: Please choose the best answer choice for each of the following questions. 1. Anton van Leeuwenhoek concluded that all plants are composed of cells. True False Correct ItemID DD-HOLT.1037649
Directions: Please choose the best answer choice for each of the following questions. 1. Anton van Leeuwenhoek concluded that all plants are composed of cells. True False Correct ItemID DD-HOLT.1037649
(A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids. A. Incorrect! Both prokaryotic and eukaryotic cells have cell membranes.
 High School Biology - Problem Drill 03: The Cell No. 1 of 10 1. Which of the following is NOT found in prokaryotic cells? #01 (A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids Both prokaryotic
High School Biology - Problem Drill 03: The Cell No. 1 of 10 1. Which of the following is NOT found in prokaryotic cells? #01 (A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids Both prokaryotic
2 3 Carbon Compounds Slide 1 of 37
 1 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with
1 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Carbon atoms have four valence electrons that can join with
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
CP Biology Midterm Review. Scientific Method
 CP Biology Midterm Review Scientific Method 1. Define the following terms: Independent variable Dependent variable Control Constant variables Quantitative Observation Qualitative Observation Variable being
CP Biology Midterm Review Scientific Method 1. Define the following terms: Independent variable Dependent variable Control Constant variables Quantitative Observation Qualitative Observation Variable being
The Cell and Its Chemical Compounds
 Cell Theory Cell - The basic unit of structure and function in living things. All of an organism s process or functions are carried out in the cell. Robert Hooke - One of the first people to observe cells
Cell Theory Cell - The basic unit of structure and function in living things. All of an organism s process or functions are carried out in the cell. Robert Hooke - One of the first people to observe cells
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
2.3 Carbon-Based Molecules. KEY CONCEPT Carbon-based molecules are the foundation of life.
 KEY CONCEPT Carbon-based molecules are the foundation of life. Carbon atoms have unique bonding properties. Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. Carbon-based
KEY CONCEPT Carbon-based molecules are the foundation of life. Carbon atoms have unique bonding properties. Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. Carbon-based
Biochemistry. Biome. & Compound. Macromolecules
 Biochemistry Biome Macromolecules & Compound 1 ATOMS the smallest unit of an element. Ex: Carbon- C MOLECULE A molecule is formed when two or more atoms join together chemically. EX: O 2 (Oxygen Gas) 2
Biochemistry Biome Macromolecules & Compound 1 ATOMS the smallest unit of an element. Ex: Carbon- C MOLECULE A molecule is formed when two or more atoms join together chemically. EX: O 2 (Oxygen Gas) 2
Anatomy & Physiology I. Macromolecules
 Anatomy & Physiology I Macromolecules Many molecules in the human body are very large, consisting of hundreds or even thousands of atoms. These are called macromolecules. Four types of macromolecules are
Anatomy & Physiology I Macromolecules Many molecules in the human body are very large, consisting of hundreds or even thousands of atoms. These are called macromolecules. Four types of macromolecules are
Unit 2 Warm Ups. Equilibrium
 Unit 2 Warm Ups Equilibrium 1. Cell wall 2. Mitochondria 3. Chloroplast 4. Vesicle 5. Vacuole 6. Rough Endoplasmic Reticulum 7. Smooth Endoplasmic Reticulum 8. Cytoskeleton 9. Lysosomes 10.Cell Membrane
Unit 2 Warm Ups Equilibrium 1. Cell wall 2. Mitochondria 3. Chloroplast 4. Vesicle 5. Vacuole 6. Rough Endoplasmic Reticulum 7. Smooth Endoplasmic Reticulum 8. Cytoskeleton 9. Lysosomes 10.Cell Membrane
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
Objectives. Carbon Bonding. Carbon Bonding, continued. Carbon Bonding
 Biochemistry Table of Contents Objectives Distinguish between organic and inorganic compounds. Explain the importance of carbon bonding in biological molecules. Identify functional groups in biological
Biochemistry Table of Contents Objectives Distinguish between organic and inorganic compounds. Explain the importance of carbon bonding in biological molecules. Identify functional groups in biological
9. At about 0 C., most enzymes are (1.) inactive (2.) active (3.) destroyed (4.) replicated
 Study Guide 1. Which of the following enzymes would digest a fat? (1.) sucrase (2.) fatase (3.) protease (4.) lipase 2. At high temperatures, the rate of enzyme action decreases because the increased heat
Study Guide 1. Which of the following enzymes would digest a fat? (1.) sucrase (2.) fatase (3.) protease (4.) lipase 2. At high temperatures, the rate of enzyme action decreases because the increased heat
Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together.
 Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
Biomolecules Macromolecules are large molecules. Macromolecules are large structures made of many smaller structures linked together. The small single structure is a monomer (mono=one). The larger structure
2.3: Carbon- Based Molecules
 2.3: Carbon- Based Molecules Carbon-based molecules are the foundation of life. Bonding Properties of Carbon Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. 1 3,
2.3: Carbon- Based Molecules Carbon-based molecules are the foundation of life. Bonding Properties of Carbon Carbon forms covalent bonds with up to four other atoms, including other carbon atoms. 1 3,
2.2 Properties of Water
 2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
Biochemistry Name: Practice Questions
 Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
A Tour of the Cell. Ch. 7
 A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
2-2 Properties of Water
 2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
The chemistry of life
 The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
Genetics Unit Bell Work September 27 & 28, 2016
 Name: Date: Genetics Unit Bell Work September 27 & 28, 2016 nswer the following questions about the process shown above. 1. What are the reactants in this process? 2. What are the products in this process?
Name: Date: Genetics Unit Bell Work September 27 & 28, 2016 nswer the following questions about the process shown above. 1. What are the reactants in this process? 2. What are the products in this process?
Biology. Slide 1 of 37. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Life s molecular diversity is based on the. properties of carbon. Chain Ring Branching chain
 Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
BIOMOLECULES. Ms. Bosse Fall 2015
 BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
Cell Processes Review
 1. Most green algae are able to obtain carbon dioxide from the environment and use it to synthesize organic compounds. This activity is an example of 1) hydrolysis 2) saprophytism 3) cellular respiration
1. Most green algae are able to obtain carbon dioxide from the environment and use it to synthesize organic compounds. This activity is an example of 1) hydrolysis 2) saprophytism 3) cellular respiration
Biomolecules. Unit 3
 Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
The Components of Life
 The omponents of Life Section 1.2 Organic hemistry Pre-View 1.2 hemistry Review MAAP-EO Biology I 8 Section 1.2 The omponents of Life Section 1.2, continued Organic hemistry Each line represents a bond.
The omponents of Life Section 1.2 Organic hemistry Pre-View 1.2 hemistry Review MAAP-EO Biology I 8 Section 1.2 The omponents of Life Section 1.2, continued Organic hemistry Each line represents a bond.
Name # Class Regents Review: Characteristics of Life and Biochemistry
 Name # Class Regents Review: Characteristics of Life and Biochemistry 6. Some processes that occur in a cell are listed below. A. utilize energy B. detect changes in the environment C. rearrange and synthesize
Name # Class Regents Review: Characteristics of Life and Biochemistry 6. Some processes that occur in a cell are listed below. A. utilize energy B. detect changes in the environment C. rearrange and synthesize
the contents of the cell from the environment.
 Name: Date: Period: Living Environment Unit 3: Cellular Processes Study Guide Due Date: Test Date: Unit 3 Important Topics: I. Aim # 14 Cell Membrane II. Aim # 15 NYS Diffusion Lab III. Aim # 16 Photosynthesis
Name: Date: Period: Living Environment Unit 3: Cellular Processes Study Guide Due Date: Test Date: Unit 3 Important Topics: I. Aim # 14 Cell Membrane II. Aim # 15 NYS Diffusion Lab III. Aim # 16 Photosynthesis
Carbon. p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms
 Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
Organic Compounds Carbon p Has four valence electrons p Can bond with many elements p Can bond to other carbon atoms n Gives carbon the ability to form chains that are almost unlimited in length. p Organic
Cells & Cell Organelles. Doing Life s Work
 Cells & Cell Organelles Doing Life s Work AP Biology 2009-2010 Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cells & Cell Organelles Doing Life s Work AP Biology 2009-2010 Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Human Epithelial Cells
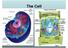 The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
Chapter 3 Review Assignment
 Class: Date: Chapter 3 Review Assignment Multiple Choice 40 MC = 40 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following organelles produces transport
Class: Date: Chapter 3 Review Assignment Multiple Choice 40 MC = 40 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following organelles produces transport
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head
 CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
Chapter 3: Biochemistry Adapted from PPT by S. Edwards. By PresenterMedia.com
 Chapter 3: Biochemistry Adapted from PPT by S. Edwards By PresenterMedia.com CARBON COMPOUNDS CHAPTER 3 SECTION 1 By PresenterMedia.com Compounds LOOK NO Carbon!!! ORGANIC COMPOUNDS Compounds that contain
Chapter 3: Biochemistry Adapted from PPT by S. Edwards By PresenterMedia.com CARBON COMPOUNDS CHAPTER 3 SECTION 1 By PresenterMedia.com Compounds LOOK NO Carbon!!! ORGANIC COMPOUNDS Compounds that contain
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
Competitive Inhibitor
 is a substance that reduces the activity of an enzyme by entering the active site in place of the substrate whose structure it mimics. Competitive Inhibitor Identify the following molecule: Polysaccharide
is a substance that reduces the activity of an enzyme by entering the active site in place of the substrate whose structure it mimics. Competitive Inhibitor Identify the following molecule: Polysaccharide
All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds:
 Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic compounds
Organic Chemistry All living things are mostly composed of 4 elements: H, O, N, C honk Compounds are broken down into 2 general categories: Inorganic Compounds: Do not contain carbon Organic compounds
Carbon. Has four valence electrons Can bond with many elements. Can bond to other carbon atoms. Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
 Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Refresher: What do we remember about CARBON? What makes it special? Nickname? Where do we find it?
 2.3: Carbon Based Molecules Situation: You are tasked with making Chicken Parm and ziti for you entire family (aunts, uncles, cousins, etc). There are 92 different ingredients you have access to in the
2.3: Carbon Based Molecules Situation: You are tasked with making Chicken Parm and ziti for you entire family (aunts, uncles, cousins, etc). There are 92 different ingredients you have access to in the
Journey to the World of Cells
 Journey to the World of Cells Starting Point for Life All living organisms on Earth are divided into pieces called cells. Formed from Proteins Organelles Make larger pieces called Tissues and systems Main
Journey to the World of Cells Starting Point for Life All living organisms on Earth are divided into pieces called cells. Formed from Proteins Organelles Make larger pieces called Tissues and systems Main
WELCOME TO BIOLOGY 11. Mr. Gandha
 WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
Study Guide for Biology Chapter 5
 Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
BIOMOLECULES. (AKA MACROMOLECULES) Name: Block:
 BIOMOLECULES (AKA MACROMOLECULES) Name: Block: BIOMOLECULES POGIL All living things share the same chemical building blocks and depend on chemical processes for survival. Life without carbon (C) would
BIOMOLECULES (AKA MACROMOLECULES) Name: Block: BIOMOLECULES POGIL All living things share the same chemical building blocks and depend on chemical processes for survival. Life without carbon (C) would
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS
 BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
BIOCHEMISTRY NOTES PT. 3 FOUR MAIN TYPES OF ORGANIC MOLECULES THAT MAKE UP LIVING THINGS 1. 2. 3. 4. CARBOHYDRATES LIPIDS (fats) PROTEINS NUCLEIC ACIDS We call these four main types of carbon- based molecules
Chapter 3. Table of Contents. Section 1 Carbon Compounds. Section 2 Molecules of Life. Biochemistry
 Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Lesson Overview 2.3 THINK ABOUT IT In the early 1800s, many chemists called the compounds created by organisms organic, believing they were fundamentally different from compounds in nonliving things. We
Midterm Exam Review 2012
 UNIT EXAM 1 Part I. Multiple choice. Write the letter of the correct answer in the multiple choice section of your answer sheet. (2 points each, 42 points) 1. Which of these units is best for measuring
UNIT EXAM 1 Part I. Multiple choice. Write the letter of the correct answer in the multiple choice section of your answer sheet. (2 points each, 42 points) 1. Which of these units is best for measuring
Lesson Overview. Carbon Compounds. Lesson Overview. 2.3 Carbon Compounds
 Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Lesson Overview 2.3 The Chemistry of Carbon What elements does carbon bond with to make up life s molecules? Carbon can bond with many elements, including Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
Biomolecules. Organic compounds of life
 Biomolecules Organic compounds of life TEKS 9A: Students will Compare the structure and function of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids 9D: Students
Biomolecules Organic compounds of life TEKS 9A: Students will Compare the structure and function of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids 9D: Students
2 3 Carbon Compounds (Macromolecules)
 2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
Animal & Plant Cells Biology 20
 Animal & Plant Cells Biology 20 Structures in Cells ALL cells start out as fully functional living things They must be able to create and maintain substances (compounds, ATP, ADP) and structures (membranes,
Animal & Plant Cells Biology 20 Structures in Cells ALL cells start out as fully functional living things They must be able to create and maintain substances (compounds, ATP, ADP) and structures (membranes,
Cell are made up of organelles. An ORGANELLE is a specialized subunit within a cell that has a specific function.
 Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Biology Final Review Answer Key
 Name: Date: Hour: Cell Energy (Cellular Respiration) Biology Final Review 14-15 Answer Key 1. What is the equation for cellular respiration in both words and formulas? Words: Glucose plus six oxygen six
Name: Date: Hour: Cell Energy (Cellular Respiration) Biology Final Review 14-15 Answer Key 1. What is the equation for cellular respiration in both words and formulas? Words: Glucose plus six oxygen six
Ch. 6: A Tour of the Cell
 Ch. 6: A Tour of the Cell 1. Compare the 2 Types of Cells PROKARYOTES BOTH EUKARYOTES Domain: Domain: Relative Size & Complexity: Relative Size & Complexity: No DNA in No Examples: Has Has Examples: 2.
Ch. 6: A Tour of the Cell 1. Compare the 2 Types of Cells PROKARYOTES BOTH EUKARYOTES Domain: Domain: Relative Size & Complexity: Relative Size & Complexity: No DNA in No Examples: Has Has Examples: 2.
Week 2. Macromolecules
 Week 2 In living organisms, smaller molecules are often attached to each other to make larger molecules. These smaller molecules are sometimes called monomers, and the larger molecules made from these
Week 2 In living organisms, smaller molecules are often attached to each other to make larger molecules. These smaller molecules are sometimes called monomers, and the larger molecules made from these
