Criteria for Annotation of Plant MicroRNAs
|
|
|
- Georgia Hill
- 6 years ago
- Views:
Transcription
1 The Plant Cell, Vol. 20: , December 2008, ª 2008 American Society of Plant Biologists Criteria for Annotation of Plant MicroRNAs Blake C. Meyers, a,1 Michael J. Axtell, b,1 Bonnie Bartel, c David P. Bartel, d David Baulcombe, e John L. Bowman, f Xiaofeng Cao, g James C. Carrington, h Xuemei Chen, i Pamela J. Green, a Sam Griffiths-Jones, j Steven E. Jacobsen, k Allison C. Mallory, l Robert A. Martienssen, m R. Scott Poethig, n Yijun Qi, o Herve Vaucheret, l Olivier Voinnet, p Yuichiro Watanabe, q Detlef Weigel, r and Jian-Kang Zhu i a Department of Plant and Soil Sciences, Delaware Biotechnology Institute, University of Delaware, Newark, Delaware b Department of Biology and Huck Institutes of the Life Sciences, Pennsylvania State University, University Park, Pennsylvania c Department of Biochemistry and Cell Biology, Rice University, Houston, Texas d Howard Hughes Medical Institute, Department of Biology, Massachusetts Institute of Technology, Whitehead Institute, Cambridge, Massachusetts e Plant Sciences Department, Cambridge University, Cambridge CB2 3EA, United Kingdom f School of Biological Sciences, Monash University, Melbourne VIC 3800, Australia g Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, Beijing , China h Center for Genome Research and Biocomputing, Department of Botany and Plant Pathology, Oregon State University, Corvallis, Oregon i Department of Botany and Plant Sciences, Institute of Integrative Genome Biology, University of California, Riverside, California j Faculty of Life Sciences, University of Manchester, Manchester M13 9PT, United Kingdom k Howard Hughes Medical Institute, Department of Molecular, Cell, and Developmental Biology, University of California, Los Angeles, California l Laboratoire de Biologie Cellulaire, Institut Jean-Pierre Bourgin, Institut National de la Recherche Agronomique, Versailles Cedex, France m Cold Spring Harbor Laboratory, Cold Spring Harbor, New York n Department of Biology, University of Pennsylvania, Philadelphia, Pennsylvania o National Institute of Biological Sciences, Zhongguancun Life Science Park, Beijing , China p Institut de Biologie Moleculaire des Plantes du Centre National de la Recherche Scientifique 12, Strasbourg Cedex, France q Department of Life Sciences, University of Tokyo, Meguro, Tokyo , Japan r Department of Molecular Biology, Max Planck Institute for Developmental Biology, D Tübingen, Germany MicroRNAs (mirnas) are ;21 nucleotide noncoding RNAs produced by Dicer-catalyzed excision from stem-loop precursors. Many plant mirnas play critical roles in development, nutrient homeostasis, abiotic stress responses, and pathogen responses via interactions with specific target mrnas. mirnas are not the only Dicer-derived small RNAs produced by plants: A substantial amount of the total small RNA abundance and an overwhelming amount of small RNA sequence diversity is contributed by distinct classes of 21- to 24-nucleotide short interfering RNAs. This fact, coupled with the rapidly increasing rate of plant small RNA discovery, demands an increased rigor in mirna annotations. Herein, we update the specific criteria required for the annotation of plant mirnas, including experimental and computational data, as well as refinements to standard nomenclature. 1 Address correspondence to meyers@dbi.udel. edu or mja18@psu.edu. It has now been more than 5 years since the original guidelines for microrna (mirna) annotation in plants and animals were spelled out by a group of leading laboratories in the small RNA field (Ambros et al., 2003). To date,.180 genes encoding mirnas have been annotated in Arabidopsis thaliana, and.1000 have been annotated among all plants, as listed in mirbase, the home of mirna data (Griffiths-Jones et al., 2008). During this time, a variety of methods and approaches have been adopted by labs working on diverse plant species, and this has led to occasional differences in the criteria and quality of data used to annotate mirnas. The original guidelines stated that a small RNA could be designated as a mirna if it fulfilled a set of expression and biogenesis criteria (Ambros et al., 2003). The expression criteria included the identification of the small RNA by cloning and/or detection by hybridization, whereas the biogenesis criteria included a precursor transcript predicted to fold into a characteristic hairpin structure, phylogenetic conservation of the mirna sequence and precursor secondary structure, and increased accumulation of a precursor when Dicer activity is
2 December reduced (Ambros et al., 2003). However, in the past 5 years our understanding of small RNAs has grown, and the tools for analyzing them have improved. Many articles reporting new mirnas in plants have assimilated the insights of recent years, but some have not. We believe that it is time to revise the minimal criteria for plant mirna annotation with the goal of maintaining the highest quality data in the mirna registry. Herein, we refine the criteria for annotating mirnas of plants. In addition to slightly different characteristics of plant versus animal mirnas, the reason for this emphasis is that plants have relatively large and complex small RNA populations within which mirnas are often a minority. By contrast, most of the small RNAs in the soma of vertebrates and flies are mirnas. The more complex pool of plant small RNAs is largely due to the plant-specific RNA POLYMERASE IV/RNA POLYMER- ASE V (PolIV/PolV) dependent short interfering RNAs (sirnas) as well as secondary sirnas, some of which are trans-acting. These diverse, endogenous sirna populations make rigorous annotation of mirnas more challenging. The enormous numbers of small RNAs being cataloged using nextgeneration DNA sequencing technologies demand a renewed uniformity in plant mirna annotation to avoid misleading and inaccurate conflation of different classes of small RNAs. PRIMARY CRITERION: PRECISE EXCISION FROM THE STEM OF A STEM-LOOP PRECURSOR We submit that the fundamental defining feature of plant mirnas is the precise excision of an ;21-nucleotide mirna/ mirna* duplex from the stem of a singlestranded, stem-loop precursor. This duplex is an intermediate of mirna biogenesis that is present after cleavage of the MIRNA stem-loop but before the mature mirna enters the silencing complex. Stem-loop precursors are predicted using genomic DNA or known ESTs/transcripts as the input for RNA secondary structure prediction software. Plant MIRNA stem-loops are more variable in size and structural features than those of animals, but confidently identified instances share the following characteristics: (1) The mirna and mirna* are derived from opposite stem-arms such that they form a duplex with two nucleotide, 3# overhangs; (2) base-pairing between the mirna and the other arm of the hairpin, which includes the mirna*, is extensive such that there are typically four or fewer mismatched mirna bases; and (3) asymmetric bulges are minimal in size (one or two bases) and frequency (typically one or less), especially within the mirna/ mirna* duplex. Small RNA-producing stem-loops that slightly violate one of these criteria could still be annotated as MIRNAs, provided that there is exceptional evidence of precise mirna/mirna* excision; but in general, those that violate these characteristics should not be classified as MIRNAs. Biogenesis from a stem-loop excludes endogenous sirnas, as they generally arise from long, perfectly double-stranded RNA. The requirement for precision in biogenesis also excludes small RNAs that derive from arbitrary positions within otherwise acceptable stem-loop precursors as well as excluding randomly degraded mrnas that have fortuitous overlap with predicted stem-loops. However, the requirement for precision does not imply that all small RNAs expressed from a stem-loop must be either the precise mirna or precise mirna*. Deep sequencing data clearly show that there are often, if not always, low frequency positional and length variants from all MIRNA stem-loops. In addition, some MIRNA precursors give rise to two or more distinct mirna/mirna* duplexes from different positions. The point at which the level of precision that defines a mirna gives way to non-mirna imprecision is somewhat subjective. However, as a general rule of thumb, stem-loops in which more than ;25% of observed small RNA abundance does not correspond to one (or more; see multifunctional stem loops below) distinct mirna/mirna* duplexes should be considered too imprecise to qualify as MIRNAs. Although ancillary criteria can enhance a mirna annotation (see below), conclusive evidence of precise biogenesis from a qualifying stem-loop is the sole criterion that is both necessary and sufficient for mirna annotation. The primary criterion is most readily satisfied by the sequencing of cdnas derived from small RNA samples, coupled with analysis of the putative precursors of the small RNAs. In most cases, complete nuclear genome sequences are required to find all possible origins for sequenced small RNAs; however, species for which there is especially good EST coverage are also amenable to mirna analysis. Isolation of only one or two small RNAs matching a predicted stem-loop does not suffice for a confident annotation; this is because cases of low coverage could miss small RNAs deriving from heterogeneous positions or from the opposite genomic strand, which indicate non-mirna origins. Ideally, sequences representing both the mirna and mirna* would be used to satisfy the primary criterion. In the absence of mirna* confirmation, a clear dominance of a specific small RNA sequence (the mirna) from one arm of a predicted stem-loop is required. In these mirna*-deficient cases, annotation is best supported by isolation and sequencing of the candidate mirna from multiple, independent libraries. It should be noted that a very low abundance of just one or two sequencing reads of a putative mirna, followed by detection via RNA gel blots, does not satisfy the primary criterion: Detection of a discrete small RNA by blot hybridization would be unexpected for a spurious decay product of a larger precursor, but a low sequencing depth cannot discriminate between sirnas and mirnas nor eliminate the possibility of heterogeneous processing. In such cases of low-depth sequencing, more extensive blot analysis with multiple probes would be needed to rule out small RNA accumulation from other positions within the putative MIRNA locus and to rule out small RNA accumulation from the opposite genomic strand. Similarly, annotation of mirnas based solely on sequencing AGOassociated small RNAs (for instance, from immunoprecipitations) may be problematic because the 5# nucleotide specificity characteristic of many AGO proteins will prevent observation of the total population of small RNAs deriving from candidate stem-loops.
3 3188 The Plant Cell ANCILLARY CRITERIA Other characteristics can be used to bolster plant mirna annotations. However, these criteria are unnecessary and/or insufficient for mirna annotation. This should not be taken to mean that these features are unworthy of study; on the contrary, investigation of the following aspects of mirnas is critical for understanding the biological role and evolution of these molecules. Additionally, satisfaction of one or more of these ancillary criteria can significantly increase the confidence of a mirna annotation. However, because they are individually either unnecessary, insufficient, or both, the following criteria are ancillary for the strict purpose of mirna annotation: Conservation Conservation of mirnas, assessed using either bioinformatics or direct experimentation, is a powerful indicator of their functional relevance and ancient origin. Preservation across lineages of a predicted stem-loop secondary structure along with the embedded mirna sequence provides especially strong evidence in favor of a mirna annotation. Nonetheless, many bona fide plant mirnas lack readily detected homologs outside of the founding species. Thus, demonstration of conservation is not necessary for annotation of mirnas. However, in contrast with the other ancillary criteria, clear evidence of conservation of both the stem-loop secondary structure and the mature mirna sequence is by itself sufficient for confident annotation of orthologous mirnas, provided that the precise stem-loop biogenesis criterion was experimentally satisfied in at least one species. In this respect, our guidelines retain the original mirna criteria as described by Ambros et al. (2003). Targets Many currently known plant mirnas mediate the regulation of specific target mrnas by one or more molecular mechanisms, including target cleavage (Llave et al., 2002), translational repression (Aukerman and Sakai, 2003; Chen, 2004), and pairing to non-protein-coding RNAs without cleavage (Axtell et al., 2006; Franco-Zorrilla et al., 2007). The biological roles of mirnas seem to be restricted to target regulation or processing; however, it does not follow that identification of a target is necessary for mirna annotation. Many less-conserved plant mirnas have predicted targets that have proven difficult to confirm, while others appear to have no targets at all. Some of these may truly be target-less mirnas that are evolutionarily transient, whereas others may have undiscovered targets that are not amenable to identification using current computational and experimental techniques. In either case, determining the function of a mirna is not required for its annotation, just as protein-coding genes without known function can also be annotated. Small RNA-directed target regulation is also not sufficient for mirna annotation. For example, trans-acting sirnas also direct cleavage of target mrnas, while heterochromatic sirnas repress targets at the transcriptional level. DCL1 Dependence All plants have multiple DICER-LIKE (DCL) genes. Among these, DCL1 appears to be largely specialized for mirna production, whereas the others are specialized for the production of various sirnas. In Arabidopsis and rice, hypomorphic dcl1 mutations impact the accumulation of most, but not all, mirnas. However, because null dcl1 alleles are embryo lethal (at least in Arabidopsis), it is not possible to determine the exact dependence of every mirna on DCL1. Because of the presence of several mirnas whose accumulation depends upon DCL4 (Rajagopalan et al., 2006), DCL1 dependency is not necessary for mirna annotation. In addition, the observation of DCL1 dependence for accumulation of a small RNA is by itself insufficient to warrant annotation as a mirna: Certain sirnas, including many secondary sirnas and some natural antisense sirnas also require DCL1 for their accumulation but do not derive from precisely processed stemloops. A strict requirement for evidence of relatively precise excision from qualifying stem-loops obviates the need for genetic analysis of DCL dependencies and enables mirnas to be annotated in those species in which dcl1 mutants are unavailable. RDR and PolIV/PolV Independence Plants also have one or more RNA- DEPENDENT RNA POLYMERASES (RDRs) thought to produce the dsrna molecules from which many sirnas derive. Thus, RDR dependencies are characteristic of many sirnas. By contrast, mirnas are not excised from RDR-derived dsrnas. However, while RDR dependency defines many sirnas, RDR independence is not restricted to mirnas. For example, RDRindependent stem-loops will not satisfy the primary criterion if they are heterogenously processed. Many sirnas also depend on PolIV/PolV. But, as for RDR independence, PolIV/PolV independence is insufficient for mirna annotation. RDR and PolIV/PolV dependence also does not necessarily preclude mirna annotation: For instance, it is theoretically possible that RDR and/or PolIV/PolV-dependent sirnas might impinge upon MIRNA transcription, thus causing an indirect genetic dependency. In summary, testing of RDR and PolIV/PolV dependencies is neither necessary nor sufficient for mirna annotation. As with dcl1 mutants, there are many species in which mutants of functionally characterized RDRs and/or the PolIV/PolV subunits are unavailable. Thus, this ancillary criterion is similarly limited in applicability. REPEATS AND STRUCTURAL RNAS Endogenous sirnas derived from the PolIV/ PolV pathway preferentially accumulate from repetitive regions, including tandem repeats and transposon-derived sequences. Because the diversity of PolIV/ PolV-dependent small RNAs is much greater than that of mirnas, special care should be taken when annotating MIRNA loci that overlap with repetitive sequences. In these cases, it becomes particularly
4 December important to satisfy the primary criterion and to rule out the possibility of sirna production from the locus of interest. This is not to say that repetitive regions in principle cannot harbor MIRNA loci. However, the burden of proof must be high, indeed, when describing MIRNA loci within repetitive regions to avoid misannotation of sirnas as mirnas. Randomly sized fragments of non-dcldependent RNAs usually, if not always, contaminate libraries of small RNAs. Almost all of these are from the most abundant classes of cellular RNAs: rrnas and trnas. Thus, any small RNAs that match the sense strand of mature rrnas or trnas should be excluded a priori from consideration as mirnas. In well-annotated genomes, such as Arabidopsis, excluding rrnas and trnas is trivial. However, in other species, care should be taken to assemble and identify rrna and trna sequences prior to annotation of novel mirnas. Other classes of less abundant noncoding RNAs, such as small nuclear and small nucleolar RNAs, can also cause contamination issues. ASSIGNING mirnas TO DISTINCT FAMILIES An additional complexity of mirna annotation is the frequent presence within a genome of paralogous MIRNA loci producing identical or nearly identical mature mirnas. Logically, these identical or nearly identical mirnas have been grouped together into families. In a sequenced genome, once a single locus has been confirmed to produce a mirna by the primary criterion, the initial identification of other potential family members is trivial; assessment of the potential stem-loop precursors at each site where perfect or near-perfect matches to the mature mirna sequence occur yields a list of presumed paralogs that can be annotated as mirnas. For species with unsequenced genomes, identifying candidate mirna family members requires cdna sequences or other experimental data. Candidate family members identified in this way may have variable degrees of experimental support. For instance, both the initially confirmed MIRNA locus and a subsequently identified paralog could produce identical mirna and mirna* species, obscuring whether mirna production derives from the initially identified locus, the paralog, or both. In some cases, sequenced mirna* species or other rare mirna variants provide locusspecific signatures proving expression from a specific paralog. Optimal annotation of paralogs would ideally include information on whether the annotation is based on ambiguous or unambiguous experimental data, or upon similarity alone. The protocol of submission of novel mirnas to the mirbase registry (Griffiths- Jones et al., 2008) after the acceptance of a manuscript has worked well to ensure an orderly assignment of mirna numbers; we do not propose any changes to this protocol. Loci giving rise to identical or similar mirnas are assigned the same number with sequential alphabetical suffixes (i.e., MIR172a, MIR172b, etc.). mirbase assigns three-letter prefixes based upon the genus and species from which the MIRNA is derived. When two or more mirna-producing stem-loops are arrayed in tandem on a single precursor, each stem loop is treated as a distinct locus (for instance, Zea mays MIR156b and MIR156c; Chuck et al., 2007). However, there are some MIRNA genes that encode similar mature mirna sequences that for historical reasons have been assigned distinct identifiers (for instance, the mir156/157, mir165/166, and mir170/171 families). The designation of mirna names should take into consideration the number of mismatches compared with other named mirnas, with from zero to two being typical, but up to four being acceptable, provided that the mature mirnas derive from the same arm of the stem-loop in all cases. In addition, sequence-related mirnas with different targets can be classified into different families at the author s discretion. For example, the sequence-related but functionally distinct Arabidopsis mir159 and mir319 families (which regulate MYB- and TCP-family targets, respectively; Palatnik et al., 2007) sometimes have been placed in separate families. MULTIFUNCTIONAL STEM-LOOPS There are several cases for which multiple mirnas accumulate from the same precursor; these include cases where mirna and mirna* species accumulate to approximately equal levels (for instance, mir832; Rajagopalan et al., 2006), cases where overlapping but distinct mirna species are produced from the same arm of a stem-loop (for instance, mir161; Allen et al., 2004), and cases where multiple mirna/mirna* duplexes are sequentially excised (for instance, mir163; Kurihara and Watanabe, 2004). In such cases, the naming procedure for mirnas becomes a bit more complicated. When there are two mirna species that accumulate in approximately equal proportions and derive from the same initial duplex, the mirna/mirna* nomenclature is omitted in favor of the suffixes 5p and 3p, which designate the mirna species arising from the 5# and 3# arms, respectively, of the stem-loop. The distinct mirna species resulting from overlapping or sequential processing should be distinguished by numerical suffixes (e.g., mir161.1 and mir161.2). In some cases, the numerical suffixes indicating the locus of origin must be combined with letter designations for locus-specific variants (for an example, see the rice MIR444d locus; Lu et al., 2008). Finally, recent data suggest that some mirna*s could indeed be competent to direct target cleavage in plants (German et al., 2008), suggesting that revisions to the 5p/3p system of nomenclature might be in store for these previously annotated mirna loci. CONCLUSIONS A problem in providing a definitive list of criteria for mirna annotation is that this list is likely to evolve over time, much as standards have evolved over the last 5 years. Our intent with this communication is to delineate criteria that we believe uniquely characterize mirnas and to provide guidelines for using these criteria to discriminate mirnas from other small RNAs. One goal is to head off the future annotation of mirnas that have a high
5 3190 The Plant Cell likelihood of being sirnas; many sequences of such uncertain provenance are likely to be identified for a broad range of plant species now that next-generation sequencing of small RNAs has become commonplace. With the application of these renewed criteria, we hope to maintain a mirna registry of the highest possible quality even at the risk of excluding some possibly valid entries that will require more support before inclusion. Authors must be self-policing in their annotations, but it also falls to reviewers to diligently flag questionable claims of mirnas. With so many articles in so many journals, it may be difficult to effectively review each and every mirna manuscript, but we hope that these criteria will be helpful to both authors and reviewers of articles describing new mirnas. REFERENCES Allen, E., Xie, Z., Gustafson, A.M., Sung, G.H., Spatafora, J.W., and Carrington, J.C. (2004). Evolution of microrna genes by inverted duplication of target gene sequences in Arabidopsis thaliana. Nat. Genet. 36: Ambros, V., et al. (2003). A uniform system for microrna annotation. RNA 9: Aukerman, M.J., and Sakai, H. (2003). Regulation of flowering time and floral organ identity by a microrna and its APETALA2-like target genes. Plant Cell 15: Axtell, M.J., Jan, C., Rajagopalan, R., and Bartel, D.P. (2006). A two-hit trigger for sirna biogenesis in plants. Cell 127: Chen, X. (2004). A microrna as a translational repressor of APETALA2 in Arabidopsis flower development. Science 303: Chuck, G., Cigan, A.M., Saeteurn, K., and Hake, S. (2007). The heterochronic maize mutant Corngrass1 results from overexpression of a tandem microrna. Nat. Genet. 39: Franco-Zorrilla, J.M., Valli, A., Todesco, M., Mateos, I., Puga, M.I., Rubio-Somoza, I., Leyva, A., Weigel, D., Garcia, J.A., and Paz- Ares, J. (2007). Target mimicry provides a new mechanism for regulation of microrna activity. Nat. Genet. 39: German, M.A., et al. (2008). Global identification of microrna-target RNA pairs by parallel analysis of RNA ends. Nat. Biotechnol. 26: Griffiths-Jones, S., Saini, H.K., van Dongen, S., and Enright, A.J. (2008). mirbase: Tools for microrna genomics. Nucleic Acids Res. 36: D154 D158. Kurihara, Y., and Watanabe, Y. (2004). Arabidopsis micro-rna biogenesis through Dicerlike 1 protein functions. Proc. Natl. Acad. Sci. USA 101: Llave, C., Xie, Z., Kasschau, K.D., and Carrington, J.C. (2002). Cleavage of Scarecrow-like mrna targets directed by a class of Arabidopsis mirna. Science 297: Lu, C., et al. (2008). Genome-wide analysis for discovery of rice micrornas reveals natural antisense micrornas (nat-mirnas). Proc. Natl. Acad. Sci. USA 105: Palatnik, J.F., Wollmann, H., Schommer, C., Schwab, R., Boisbouvier, J., Rodriguez, R., Warthmann, N., Allen, E., Dezulian, T., Huson, D., Carrington, J.C., and Weigel, D. (2007). Sequence and expression differences underlie functional specialization of Arabidopsis micrornas mir159 and mir319. Dev. Cell 13: Rajagopalan, R., Vaucheret, H., Trejo, J., and Bartel, D.P. (2006). A diverse and evolutionarily fluid set of micrornas in Arabidopsis thaliana. Genes Dev. 20:
6 Criteria for Annotation of Plant MicroRNAs Blake C. Meyers, Michael J. Axtell, Bonnie Bartel, David P. Bartel, David Baulcombe, John L. Bowman, Xiaofeng Cao, James C. Carrington, Xuemei Chen, Pamela J. Green, Sam Griffiths-Jones, Steven E. Jacobsen, Allison C. Mallory, Robert A. Martienssen, R. Scott Poethig, Yijun Qi, Herve Vaucheret, Olivier Voinnet, Yuichiro Watanabe, Detlef Weigel and Jian-Kang Zhu Plant Cell 2008;20; ; originally published online December 12, 2008; DOI /tpc This information is current as of June 18, 2018 References Permissions etocs CiteTrack Alerts Subscription Information This article cites 14 articles, 7 of which can be accessed free at: /content/20/12/3186.full.html#ref-list-1 Sign up for etocs at: Sign up for CiteTrack Alerts at: Subscription Information for The Plant Cell and Plant Physiology is available at: American Society of Plant Biologists ADVANCING THE SCIENCE OF PLANT BIOLOGY
he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003)
 MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
Identification of mirnas in Eucalyptus globulus Plant by Computational Methods
 International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 2 Issue 5 May 2013 PP.70-74 Identification of mirnas in Eucalyptus globulus Plant by Computational
International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 2 Issue 5 May 2013 PP.70-74 Identification of mirnas in Eucalyptus globulus Plant by Computational
Bi 8 Lecture 17. interference. Ellen Rothenberg 1 March 2016
 Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Supplemental Figure 1. Small RNA size distribution from different soybean tissues.
 Supplemental Figure 1. Small RNA size distribution from different soybean tissues. The size of small RNAs was plotted versus frequency (percentage) among total sequences (A, C, E and G) or distinct sequences
Supplemental Figure 1. Small RNA size distribution from different soybean tissues. The size of small RNAs was plotted versus frequency (percentage) among total sequences (A, C, E and G) or distinct sequences
ANALYSIS OF SOYBEAN SMALL RNA POPULATIONS HLAING HLAING WIN THESIS
 ANALYSIS OF SOYBEAN SMALL RNA POPULATIONS BY HLAING HLAING WIN THESIS Submitted in partial fulfillment of the requirements for the degree of Master of Science in Bioinformatics in the Graduate College
ANALYSIS OF SOYBEAN SMALL RNA POPULATIONS BY HLAING HLAING WIN THESIS Submitted in partial fulfillment of the requirements for the degree of Master of Science in Bioinformatics in the Graduate College
MicroRNAs and Their Regulatory Roles in Plants
 Annu. Rev. lant Biol. 2006. 57:19 53 The Annual Review of lant Biology is online at plant.annualreviews.org doi: 10.1146/ annurev.arplant.57.032905.105218 opyright c 2006 by Annual Reviews. All rights
Annu. Rev. lant Biol. 2006. 57:19 53 The Annual Review of lant Biology is online at plant.annualreviews.org doi: 10.1146/ annurev.arplant.57.032905.105218 opyright c 2006 by Annual Reviews. All rights
Circular RNAs (circrnas) act a stable mirna sponges
 Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Cloning and Characterization of MicroRNAs from Rice W
 The Plant Cell, Vol. 17, 1397 1411, May 2005, www.plantcell.org ª 2005 American Society of Plant Biologists Cloning and Characterization of MicroRNAs from Rice W Ramanjulu Sunkar, Thomas Girke, Pradeep
The Plant Cell, Vol. 17, 1397 1411, May 2005, www.plantcell.org ª 2005 American Society of Plant Biologists Cloning and Characterization of MicroRNAs from Rice W Ramanjulu Sunkar, Thomas Girke, Pradeep
Phenomena first observed in petunia
 Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
High AU content: a signature of upregulated mirna in cardiac diseases
 https://helda.helsinki.fi High AU content: a signature of upregulated mirna in cardiac diseases Gupta, Richa 2010-09-20 Gupta, R, Soni, N, Patnaik, P, Sood, I, Singh, R, Rawal, K & Rani, V 2010, ' High
https://helda.helsinki.fi High AU content: a signature of upregulated mirna in cardiac diseases Gupta, Richa 2010-09-20 Gupta, R, Soni, N, Patnaik, P, Sood, I, Singh, R, Rawal, K & Rani, V 2010, ' High
COMPUTATIONAL ANALYSIS OF SMALL RNAs IN MAIZE MUTANTS WITH DEFECTS IN DEVELOPMENT AND PARAMUTATION. Reza Hammond
 COMPUTATIONAL ANALYSIS OF SMALL RNAs IN MAIZE MUTANTS WITH DEFECTS IN DEVELOPMENT AND PARAMUTATION by Reza Hammond A thesis submitted to the Faculty of the University of Delaware in partial fulfillment
COMPUTATIONAL ANALYSIS OF SMALL RNAs IN MAIZE MUTANTS WITH DEFECTS IN DEVELOPMENT AND PARAMUTATION by Reza Hammond A thesis submitted to the Faculty of the University of Delaware in partial fulfillment
Widespread Long Noncoding RNAs as Endogenous Target Mimics for MicroRNAs in Plants 1[W]
![Widespread Long Noncoding RNAs as Endogenous Target Mimics for MicroRNAs in Plants 1[W] Widespread Long Noncoding RNAs as Endogenous Target Mimics for MicroRNAs in Plants 1[W]](/thumbs/96/127589351.jpg) Widespread Long Noncoding RNAs as Endogenous Target Mimics for MicroRNAs in Plants 1[W] Hua-Jun Wu 2, Zhi-Min Wang 2, Meng Wang, and Xiu-Jie Wang* State Key Laboratory of Plant Genomics, Institute of Genetics
Widespread Long Noncoding RNAs as Endogenous Target Mimics for MicroRNAs in Plants 1[W] Hua-Jun Wu 2, Zhi-Min Wang 2, Meng Wang, and Xiu-Jie Wang* State Key Laboratory of Plant Genomics, Institute of Genetics
In silico identification of mirnas and their targets from the expressed sequence tags of Raphanus sativus
 www.bioinformation.net Hypothesis Volume 8(2) In silico identification of mirnas and their targets from the expressed sequence tags of Raphanus sativus Charuvaka Muvva 1 *, Lata Tewari 2, Kasoju Aruna
www.bioinformation.net Hypothesis Volume 8(2) In silico identification of mirnas and their targets from the expressed sequence tags of Raphanus sativus Charuvaka Muvva 1 *, Lata Tewari 2, Kasoju Aruna
Improved annotation of C. elegans micrornas by deep sequencing reveals structures associated with processing by Drosha and Dicer
 BIOINFORMATICS Improved annotation of C. elegans micrornas by deep sequencing reveals structures associated with processing by Drosha and Dicer M. BRYAN WARF, 1 W. EVAN JOHNSON, 2 and BRENDA L. BASS 1
BIOINFORMATICS Improved annotation of C. elegans micrornas by deep sequencing reveals structures associated with processing by Drosha and Dicer M. BRYAN WARF, 1 W. EVAN JOHNSON, 2 and BRENDA L. BASS 1
MicroRNA in Cancer Karen Dybkær 2013
 MicroRNA in Cancer Karen Dybkær RNA Ribonucleic acid Types -Coding: messenger RNA (mrna) coding for proteins -Non-coding regulating protein formation Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
MicroRNA in Cancer Karen Dybkær RNA Ribonucleic acid Types -Coding: messenger RNA (mrna) coding for proteins -Non-coding regulating protein formation Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
IDENTIFICATION OF IN SILICO MIRNAS IN FOUR PLANT SPECIES FROM FABACEAE FAMILY
 Original scientific paper 10.7251/AGRENG1803122A UDC633:34+582.736.3]:577.2 IDENTIFICATION OF IN SILICO MIRNAS IN FOUR PLANT SPECIES FROM FABACEAE FAMILY Bihter AVSAR 1*, Danial ESMAEILI ALIABADI 2 1 Sabanci
Original scientific paper 10.7251/AGRENG1803122A UDC633:34+582.736.3]:577.2 IDENTIFICATION OF IN SILICO MIRNAS IN FOUR PLANT SPECIES FROM FABACEAE FAMILY Bihter AVSAR 1*, Danial ESMAEILI ALIABADI 2 1 Sabanci
2009 LANDES BIOSCIENCE. DO NOT DISTRIBUTE.
 [Epigenetics 4:2, 1-6; 16 February 2009]; 2009 Landes Bioscience Research Paper Determining the conservation of DNA methylation in Arabidopsis This manuscript has been published online, prior to printing.once
[Epigenetics 4:2, 1-6; 16 February 2009]; 2009 Landes Bioscience Research Paper Determining the conservation of DNA methylation in Arabidopsis This manuscript has been published online, prior to printing.once
Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins.
 Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins. The RNA transcribed from a complex transcription unit
Alternative RNA processing: Two examples of complex eukaryotic transcription units and the effect of mutations on expression of the encoded proteins. The RNA transcribed from a complex transcription unit
Eukaryotic small RNA Small RNAseq data analysis for mirna identification
 Eukaryotic small RNA Small RNAseq data analysis for mirna identification P. Bardou, C. Gaspin, S. Maman, J. Mariette, O. Rué, M. Zytnicki INRA Sigenae Toulouse INRA MIA Toulouse GenoToul Bioinfo INRA MaIAGE
Eukaryotic small RNA Small RNAseq data analysis for mirna identification P. Bardou, C. Gaspin, S. Maman, J. Mariette, O. Rué, M. Zytnicki INRA Sigenae Toulouse INRA MIA Toulouse GenoToul Bioinfo INRA MaIAGE
The Cornucopia of Small RNAs in Plant Genomes
 Rice (2008) 1:52 62 DOI 10.1007/s12284-008-9008-5 REVIEW The Cornucopia of Small RNAs in Plant Genomes Stacey A. Simon & Jixian Zhai & Jia Zeng & Blake C. Meyers Received: 10 June 2008 /Accepted: 3 July
Rice (2008) 1:52 62 DOI 10.1007/s12284-008-9008-5 REVIEW The Cornucopia of Small RNAs in Plant Genomes Stacey A. Simon & Jixian Zhai & Jia Zeng & Blake C. Meyers Received: 10 June 2008 /Accepted: 3 July
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Identification of novel and candidate mirnas in rice by high throughput sequencing. and J-K Zhu,
 Identification of novel and candidate mirnas in rice by high throughput sequencing Ramanjulu Sunkar 1,4*, Xuefeng Zhou 2* Yun Zheng 2, Weixiong Zhang 2, and Jian-Kang Zhu, 3,4 1 Department of Biochemistry
Identification of novel and candidate mirnas in rice by high throughput sequencing Ramanjulu Sunkar 1,4*, Xuefeng Zhou 2* Yun Zheng 2, Weixiong Zhang 2, and Jian-Kang Zhu, 3,4 1 Department of Biochemistry
sirna count per 50 kb small RNAs matching the direct strand Repeat length (bp) per 50 kb repeats in the chromosome
 Qi et al. 26-3-2564C Qi et al., Figure S1 sirna count per 5 kb small RNAs matching the direct strand sirna count per 5 kb small RNAs matching the complementary strand Repeat length (bp) per 5 kb repeats
Qi et al. 26-3-2564C Qi et al., Figure S1 sirna count per 5 kb small RNAs matching the direct strand sirna count per 5 kb small RNAs matching the complementary strand Repeat length (bp) per 5 kb repeats
MicroRNA-mediated incoherent feedforward motifs are robust
 The Second International Symposium on Optimization and Systems Biology (OSB 8) Lijiang, China, October 31 November 3, 8 Copyright 8 ORSC & APORC, pp. 62 67 MicroRNA-mediated incoherent feedforward motifs
The Second International Symposium on Optimization and Systems Biology (OSB 8) Lijiang, China, October 31 November 3, 8 Copyright 8 ORSC & APORC, pp. 62 67 MicroRNA-mediated incoherent feedforward motifs
RNA-seq Introduction
 RNA-seq Introduction DNA is the same in all cells but which RNAs that is present is different in all cells There is a wide variety of different functional RNAs Which RNAs (and sometimes then translated
RNA-seq Introduction DNA is the same in all cells but which RNAs that is present is different in all cells There is a wide variety of different functional RNAs Which RNAs (and sometimes then translated
Supplementary information for: Human micrornas co-silence in well-separated groups and have different essentialities
 Supplementary information for: Human micrornas co-silence in well-separated groups and have different essentialities Gábor Boross,2, Katalin Orosz,2 and Illés J. Farkas 2, Department of Biological Physics,
Supplementary information for: Human micrornas co-silence in well-separated groups and have different essentialities Gábor Boross,2, Katalin Orosz,2 and Illés J. Farkas 2, Department of Biological Physics,
MicroRNA and Male Infertility: A Potential for Diagnosis
 Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
MicroRNAs, RNA Modifications, RNA Editing. Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM
 MicroRNAs, RNA Modifications, RNA Editing Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM Expanding world of RNAs mrna, messenger RNA (~20,000) trna, transfer
MicroRNAs, RNA Modifications, RNA Editing Bora E. Baysal MD, PhD Oncology for Scientists Lecture Tue, Oct 17, 2017, 3:30 PM - 5:00 PM Expanding world of RNAs mrna, messenger RNA (~20,000) trna, transfer
micrornas (mirna) and Biomarkers
 micrornas (mirna) and Biomarkers Small RNAs Make Big Splash mirnas & Genome Function Biomarkers in Cancer Future Prospects Javed Khan M.D. National Cancer Institute EORTC-NCI-ASCO November 2007 The Human
micrornas (mirna) and Biomarkers Small RNAs Make Big Splash mirnas & Genome Function Biomarkers in Cancer Future Prospects Javed Khan M.D. National Cancer Institute EORTC-NCI-ASCO November 2007 The Human
Cross species analysis of genomics data. Computational Prediction of mirnas and their targets
 02-716 Cross species analysis of genomics data Computational Prediction of mirnas and their targets Outline Introduction Brief history mirna Biogenesis Why Computational Methods? Computational Methods
02-716 Cross species analysis of genomics data Computational Prediction of mirnas and their targets Outline Introduction Brief history mirna Biogenesis Why Computational Methods? Computational Methods
Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Chapter 18 Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development and is responsible for differences
Chapter 18 Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development and is responsible for differences
CONTRACTING ORGANIZATION: Cold Spring Harbor Laboratory Cold Spring Harbor, NY 11724
 AD Award Number: W81XWH-06-1-0249 TITLE: Detection of genes modifying sensitivity to proteasome inhibitors using a shrna Library in Breast Cancer PRINCIPAL INVESTIGATOR: Gregory J. Hannon, Ph.D. CONTRACTING
AD Award Number: W81XWH-06-1-0249 TITLE: Detection of genes modifying sensitivity to proteasome inhibitors using a shrna Library in Breast Cancer PRINCIPAL INVESTIGATOR: Gregory J. Hannon, Ph.D. CONTRACTING
Novel and Stress-Regulated MicroRNAs and Other Small RNAs from Arabidopsis W
 The Plant Cell, Vol. 16, 2001 2019, August 2004, www.plantcell.org ª 2004 American Society of Plant Biologists Novel and Stress-Regulated MicroRNAs and Other Small RNAs from Arabidopsis W Ramanjulu Sunkar
The Plant Cell, Vol. 16, 2001 2019, August 2004, www.plantcell.org ª 2004 American Society of Plant Biologists Novel and Stress-Regulated MicroRNAs and Other Small RNAs from Arabidopsis W Ramanjulu Sunkar
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Genetics and Genomics in Medicine Chapter 6 Questions
 Genetics and Genomics in Medicine Chapter 6 Questions Multiple Choice Questions Question 6.1 With respect to the interconversion between open and condensed chromatin shown below: Which of the directions
Genetics and Genomics in Medicine Chapter 6 Questions Multiple Choice Questions Question 6.1 With respect to the interconversion between open and condensed chromatin shown below: Which of the directions
Small RNAs and Cross-Kingdom RNAi in Plant-Microbial Interaction Hailing Jin
 Small RNAs and Cross-Kingdom RNAi in Plant-Microbial Interaction Hailing Jin Department of Plant Pathology & Microbiology Institute for Integrative Genome Biology micrornas (mirnas) Small RNAs PolII PolIV
Small RNAs and Cross-Kingdom RNAi in Plant-Microbial Interaction Hailing Jin Department of Plant Pathology & Microbiology Institute for Integrative Genome Biology micrornas (mirnas) Small RNAs PolII PolIV
Transcriptome-wide analysis of microrna expression in the malaria mosquito Anopheles gambiae
 Biryukova et al. BMC Genomics 2014, 15:557 RESEARCH ARTICLE Open Access Transcriptome-wide analysis of microrna expression in the malaria mosquito Anopheles gambiae Inna Biryukova 1*, Tao Ye 2 and Elena
Biryukova et al. BMC Genomics 2014, 15:557 RESEARCH ARTICLE Open Access Transcriptome-wide analysis of microrna expression in the malaria mosquito Anopheles gambiae Inna Biryukova 1*, Tao Ye 2 and Elena
V16: involvement of micrornas in GRNs
 What are micrornas? V16: involvement of micrornas in GRNs How can one identify micrornas? What is the function of micrornas? Elisa Izaurralde, MPI Tübingen Huntzinger, Izaurralde, Nat. Rev. Genet. 12,
What are micrornas? V16: involvement of micrornas in GRNs How can one identify micrornas? What is the function of micrornas? Elisa Izaurralde, MPI Tübingen Huntzinger, Izaurralde, Nat. Rev. Genet. 12,
mirna Dr. S Hosseini-Asl
 mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
Genome-Wide Profiling and Analysis of Arabidopsis sirnas
 Genome-Wide Profiling and Analysis of Arabidopsis sirnas PLoS BIOLOGY Kristin D. Kasschau 1,2, Noah Fahlgren 1,2,3, Elisabeth J. Chapman 1,2,3, Christopher M. Sullivan 1,2, Jason S. Cumbie 1,2, Scott A.
Genome-Wide Profiling and Analysis of Arabidopsis sirnas PLoS BIOLOGY Kristin D. Kasschau 1,2, Noah Fahlgren 1,2,3, Elisabeth J. Chapman 1,2,3, Christopher M. Sullivan 1,2, Jason S. Cumbie 1,2, Scott A.
Characterization and comparison of flower bud micrornas from yellow-horn species
 Characterization and comparison of flower bud micrornas from yellow-horn species Y. Ao 1,2 1 Key Laboratory for Silviculture and Conservation, Ministry of Education, Beijing Forestry University, Beijing,
Characterization and comparison of flower bud micrornas from yellow-horn species Y. Ao 1,2 1 Key Laboratory for Silviculture and Conservation, Ministry of Education, Beijing Forestry University, Beijing,
CRS4 Seminar series. Inferring the functional role of micrornas from gene expression data CRS4. Biomedicine. Bioinformatics. Paolo Uva July 11, 2012
 CRS4 Seminar series Inferring the functional role of micrornas from gene expression data CRS4 Biomedicine Bioinformatics Paolo Uva July 11, 2012 Partners Pharmaceutical company Fondazione San Raffaele,
CRS4 Seminar series Inferring the functional role of micrornas from gene expression data CRS4 Biomedicine Bioinformatics Paolo Uva July 11, 2012 Partners Pharmaceutical company Fondazione San Raffaele,
The Biology and Genetics of Cells and Organisms The Biology of Cancer
 The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
The Biology and Genetics of Cells and Organisms The Biology of Cancer Mendel and Genetics How many distinct genes are present in the genomes of mammals? - 21,000 for human. - Genetic information is carried
High-resolution identification and abundance profiling of cassava (Manihot esculenta Crantz) micrornas
 Khatabi et al. BMC Genomics (216) 17:85 DOI 1.1186/s12864-16-2391-1 RESEARCH ARTICLE High-resolution identification and abundance profiling of cassava (Manihot esculenta Crantz) micrornas Behnam Khatabi
Khatabi et al. BMC Genomics (216) 17:85 DOI 1.1186/s12864-16-2391-1 RESEARCH ARTICLE High-resolution identification and abundance profiling of cassava (Manihot esculenta Crantz) micrornas Behnam Khatabi
Research Article Base Composition Characteristics of Mammalian mirnas
 Journal of Nucleic Acids Volume 2013, Article ID 951570, 6 pages http://dx.doi.org/10.1155/2013/951570 Research Article Base Composition Characteristics of Mammalian mirnas Bin Wang Department of Chemistry,
Journal of Nucleic Acids Volume 2013, Article ID 951570, 6 pages http://dx.doi.org/10.1155/2013/951570 Research Article Base Composition Characteristics of Mammalian mirnas Bin Wang Department of Chemistry,
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION. Ana M. Martinez
 NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University
 Role of Chemical lexposure in Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University CNV Discovery Reference Genetic
Role of Chemical lexposure in Generating Spontaneous Copy Number Variants (CNVs) Jennifer Freeman Assistant Professor of Toxicology School of Health Sciences Purdue University CNV Discovery Reference Genetic
Cloning and characterization of small RNAs from Medicago truncatula reveals four novel legume-specific microrna families
 Research Cloning and characterization of small RNAs from Blackwell Oxford, NPH New 0028-646X 1469-8137 2915 10.1111/j.1469-8137.2009.02915.x June 085??? 98??? Original XXThe Phytologist Authors UK Article
Research Cloning and characterization of small RNAs from Blackwell Oxford, NPH New 0028-646X 1469-8137 2915 10.1111/j.1469-8137.2009.02915.x June 085??? 98??? Original XXThe Phytologist Authors UK Article
Small RNAs and how to analyze them using sequencing
 Small RNAs and how to analyze them using sequencing Jakub Orzechowski Westholm (1) Long- term bioinforma=cs support, Science For Life Laboratory Stockholm (2) Department of Biophysics and Biochemistry,
Small RNAs and how to analyze them using sequencing Jakub Orzechowski Westholm (1) Long- term bioinforma=cs support, Science For Life Laboratory Stockholm (2) Department of Biophysics and Biochemistry,
RECAP (1)! In eukaryotes, large primary transcripts are processed to smaller, mature mrnas.! What was first evidence for this precursorproduct
 RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
Santosh Patnaik, MD, PhD! Assistant Member! Department of Thoracic Surgery! Roswell Park Cancer Institute!
 Santosh Patnaik, MD, PhD Assistant Member Department of Thoracic Surgery Roswell Park Cancer Institute MicroRNA biology, techniques and applications History Biogenesis Nomenclature Tissue specificity Mechanisms
Santosh Patnaik, MD, PhD Assistant Member Department of Thoracic Surgery Roswell Park Cancer Institute MicroRNA biology, techniques and applications History Biogenesis Nomenclature Tissue specificity Mechanisms
MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting sirnas
 RESOURCE/METHODOLOGY MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting sirnas Jixian Zhai, 1,2 Dong-Hoon Jeong, 1,2 Emanuele De Paoli, 1,2,7
RESOURCE/METHODOLOGY MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting sirnas Jixian Zhai, 1,2 Dong-Hoon Jeong, 1,2 Emanuele De Paoli, 1,2,7
38 Int'l Conf. Bioinformatics and Computational Biology BIOCOMP'16
 38 Int'l Conf. Bioinformatics and Computational Biology BIOCOMP'16 PGAR: ASD Candidate Gene Prioritization System Using Expression Patterns Steven Cogill and Liangjiang Wang Department of Genetics and
38 Int'l Conf. Bioinformatics and Computational Biology BIOCOMP'16 PGAR: ASD Candidate Gene Prioritization System Using Expression Patterns Steven Cogill and Liangjiang Wang Department of Genetics and
Table S1. Relative abundance of AGO1/4 proteins in different organs. Table S2. Summary of smrna datasets from various samples.
 Supplementary files Table S1. Relative abundance of AGO1/4 proteins in different organs. Table S2. Summary of smrna datasets from various samples. Table S3. Specificity of AGO1- and AGO4-preferred 24-nt
Supplementary files Table S1. Relative abundance of AGO1/4 proteins in different organs. Table S2. Summary of smrna datasets from various samples. Table S3. Specificity of AGO1- and AGO4-preferred 24-nt
Small RNAs and how to analyze them using sequencing
 Small RNAs and how to analyze them using sequencing RNA-seq Course November 8th 2017 Marc Friedländer ComputaAonal RNA Biology Group SciLifeLab / Stockholm University Special thanks to Jakub Westholm for
Small RNAs and how to analyze them using sequencing RNA-seq Course November 8th 2017 Marc Friedländer ComputaAonal RNA Biology Group SciLifeLab / Stockholm University Special thanks to Jakub Westholm for
1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics
 1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics to gain mechanistic insight! 4. Return to step 2, as
1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics to gain mechanistic insight! 4. Return to step 2, as
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing
 University of Pennsylvania ScholarlyCommons Departmental Papers (Biology) Department of Biology 1-15-2010 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor
University of Pennsylvania ScholarlyCommons Departmental Papers (Biology) Department of Biology 1-15-2010 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor
MicroRNA expression profiling and functional analysis in prostate cancer. Marco Folini s.c. Ricerca Traslazionale DOSL
 MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
MicroRNA expression profiling and functional analysis in prostate cancer Marco Folini s.c. Ricerca Traslazionale DOSL What are micrornas? For almost three decades, the alteration of protein-coding genes
genomics for systems biology / ISB2020 RNA sequencing (RNA-seq)
 RNA sequencing (RNA-seq) Module Outline MO 13-Mar-2017 RNA sequencing: Introduction 1 WE 15-Mar-2017 RNA sequencing: Introduction 2 MO 20-Mar-2017 Paper: PMID 25954002: Human genomics. The human transcriptome
RNA sequencing (RNA-seq) Module Outline MO 13-Mar-2017 RNA sequencing: Introduction 1 WE 15-Mar-2017 RNA sequencing: Introduction 2 MO 20-Mar-2017 Paper: PMID 25954002: Human genomics. The human transcriptome
Regulation of Gene Expression in Eukaryotes
 Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Chapter 2. Investigation into mir-346 Regulation of the nachr α5 Subunit
 15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design
 Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design Crick's "Central Dogma"? DNA makes RNA makes protein Crick's "Central Dogma"? DNA makes
Alan Weiner BIOCHEM 530 Friday, MEB 248 October 23, 2015 RNA structure, the ribosome, structure-based drug design Crick's "Central Dogma"? DNA makes RNA makes protein Crick's "Central Dogma"? DNA makes
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
microrna Presented for: Presented by: Date:
 microrna Presented for: Presented by: Date: 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions
microrna Presented for: Presented by: Date: 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions
Breast cancer. Risk factors you cannot change include: Treatment Plan Selection. Inferring Transcriptional Module from Breast Cancer Profile Data
 Breast cancer Inferring Transcriptional Module from Breast Cancer Profile Data Breast Cancer and Targeted Therapy Microarray Profile Data Inferring Transcriptional Module Methods CSC 177 Data Warehousing
Breast cancer Inferring Transcriptional Module from Breast Cancer Profile Data Breast Cancer and Targeted Therapy Microarray Profile Data Inferring Transcriptional Module Methods CSC 177 Data Warehousing
Genomics 98 (2011) Contents lists available at ScienceDirect. Genomics. journal homepage:
 Genomics 98 (2011) 46 Contents lists available at ScienceDirect Genomics journal homepage: www.elsevier.com/locate/ygeno Global analysis of gene-level microrna expression in Arabidopsis using deep sequencing
Genomics 98 (2011) 46 Contents lists available at ScienceDirect Genomics journal homepage: www.elsevier.com/locate/ygeno Global analysis of gene-level microrna expression in Arabidopsis using deep sequencing
Identification of target genes of micrornas in retinoic acid-induced neuronal differentiation*
 Pure Appl. Chem., Vol. 77, No. 1, pp. 313 318, 2005. DOI: 10.1351/pac200577010313 2005 IUPAC Identification of target genes of micrornas in retinoic acid-induced neuronal differentiation* Hiroaki Kawasaki
Pure Appl. Chem., Vol. 77, No. 1, pp. 313 318, 2005. DOI: 10.1351/pac200577010313 2005 IUPAC Identification of target genes of micrornas in retinoic acid-induced neuronal differentiation* Hiroaki Kawasaki
Novel RNAs along the Pathway of Gene Expression. (or, The Expanding Universe of Small RNAs)
 Novel RNAs along the Pathway of Gene Expression (or, The Expanding Universe of Small RNAs) Central Dogma DNA RNA Protein replication transcription translation Central Dogma DNA RNA Spliced RNA Protein
Novel RNAs along the Pathway of Gene Expression (or, The Expanding Universe of Small RNAs) Central Dogma DNA RNA Protein replication transcription translation Central Dogma DNA RNA Spliced RNA Protein
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Human Genome: Mapping, Sequencing Techniques, Diseases
 Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
Identification of mirna from Porphyra yezoensis by High-Throughput Sequencing and Bioinformatics Analysis
 Identification of mirna from Porphyra yezoensis by High-Throughput Sequencing and Bioinformatics Analysis Chengwei Liang 1., Xiaowen Zhang 2., Jian Zou 2, Dong Xu 2, Feng Su 1, Naihao Ye 2 * 1 Qingdao
Identification of mirna from Porphyra yezoensis by High-Throughput Sequencing and Bioinformatics Analysis Chengwei Liang 1., Xiaowen Zhang 2., Jian Zou 2, Dong Xu 2, Feng Su 1, Naihao Ye 2 * 1 Qingdao
Small RNA and PARE sequencing in flower bud reveal the involvement of srnas in endodormancy release of Japanese pear (Pyrus pyrifolia 'Kosui')
 Bai et al. BMC Genomics (2016) 17:230 DOI 10.1186/s12864-016-2514-8 RESEARCH ARTICLE Open Access Small RNA and PARE sequencing in flower bud reveal the involvement of srnas in endodormancy release of Japanese
Bai et al. BMC Genomics (2016) 17:230 DOI 10.1186/s12864-016-2514-8 RESEARCH ARTICLE Open Access Small RNA and PARE sequencing in flower bud reveal the involvement of srnas in endodormancy release of Japanese
Mature microrna identification via the use of a Naive Bayes classifier
 Mature microrna identification via the use of a Naive Bayes classifier Master Thesis Gkirtzou Katerina Computer Science Department University of Crete 13/03/2009 Gkirtzou K. (CSD UOC) Mature microrna identification
Mature microrna identification via the use of a Naive Bayes classifier Master Thesis Gkirtzou Katerina Computer Science Department University of Crete 13/03/2009 Gkirtzou K. (CSD UOC) Mature microrna identification
Repressive Transcription
 Repressive Transcription The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Guenther, M. G., and R. A.
Repressive Transcription The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Guenther, M. G., and R. A.
Gynophore mirna analysis at different developmental stages in Arachis duranensis
 Gynophore mirna analysis at different developmental stages in Arachis duranensis Y. Shen 1,2 *, Y.H. Liu 1 *, X.J. Zhang 3, Q. Sha 1 and Z.D. Chen 1 1 Institute of Industrial Crops, Jiangsu Academy of
Gynophore mirna analysis at different developmental stages in Arachis duranensis Y. Shen 1,2 *, Y.H. Liu 1 *, X.J. Zhang 3, Q. Sha 1 and Z.D. Chen 1 1 Institute of Industrial Crops, Jiangsu Academy of
Intrinsic cellular defenses against virus infection
 Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
SC-L-H shared(37) Specific (1)
 A. Brain (total 2) Tissue-specific (2) Brain-heart shared (8) Specific (2) CNS-heart shared(68) Specific () Heart (total 4) Tissue-specific () B-L-H shared (37) specific () B. Brain (total 2) Tissue-specific
A. Brain (total 2) Tissue-specific (2) Brain-heart shared (8) Specific (2) CNS-heart shared(68) Specific () Heart (total 4) Tissue-specific () B-L-H shared (37) specific () B. Brain (total 2) Tissue-specific
Prediction of micrornas and their targets
 Prediction of micrornas and their targets Introduction Brief history mirna Biogenesis Computational Methods Mature and precursor mirna prediction mirna target gene prediction Summary micrornas? RNA can
Prediction of micrornas and their targets Introduction Brief history mirna Biogenesis Computational Methods Mature and precursor mirna prediction mirna target gene prediction Summary micrornas? RNA can
Deep Annotation of Populus trichocarpa MicroRNAs from Diverse Tissue Sets
 Deep Annotation of Populus trichocarpa MicroRNAs from Diverse Tissue Sets The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation
Deep Annotation of Populus trichocarpa MicroRNAs from Diverse Tissue Sets The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation
Analysis of Massively Parallel Sequencing Data Application of Illumina Sequencing to the Genetics of Human Cancers
 Analysis of Massively Parallel Sequencing Data Application of Illumina Sequencing to the Genetics of Human Cancers Gordon Blackshields Senior Bioinformatician Source BioScience 1 To Cancer Genetics Studies
Analysis of Massively Parallel Sequencing Data Application of Illumina Sequencing to the Genetics of Human Cancers Gordon Blackshields Senior Bioinformatician Source BioScience 1 To Cancer Genetics Studies
Computational identification and analysis of novel sugarcane micrornas
 Thiebaut et al. BMC Genomics 2012, 13:290 RESEARCH ARTICLE Open Access Computational identification and analysis of novel sugarcane micrornas Flávia Thiebaut 1, Clícia Grativol 1, Mariana Carnavale-Bottino
Thiebaut et al. BMC Genomics 2012, 13:290 RESEARCH ARTICLE Open Access Computational identification and analysis of novel sugarcane micrornas Flávia Thiebaut 1, Clícia Grativol 1, Mariana Carnavale-Bottino
RNA- seq Introduc1on. Promises and pi7alls
 RNA- seq Introduc1on Promises and pi7alls DNA is the same in all cells but which RNAs that is present is different in all cells There is a wide variety of different func1onal RNAs Which RNAs (and some1mes
RNA- seq Introduc1on Promises and pi7alls DNA is the same in all cells but which RNAs that is present is different in all cells There is a wide variety of different func1onal RNAs Which RNAs (and some1mes
RNA interference (RNAi)
 RN interference (RNi) Natasha aplen ene Silencing Section Office of Science and Technology Partnerships Office of the Director enter for ancer Research National ancer Institute ncaplen@mail.nih.gov Plants
RN interference (RNi) Natasha aplen ene Silencing Section Office of Science and Technology Partnerships Office of the Director enter for ancer Research National ancer Institute ncaplen@mail.nih.gov Plants
Identification and Characterization of MicroRNAs from Barley (Hordeum vulgare L.) by High-Throughput Sequencing
 Int. J. Mol. Sci. 2012, 13, 2973-2984; doi:10.3390/ijms13032973 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Identification and Characterization
Int. J. Mol. Sci. 2012, 13, 2973-2984; doi:10.3390/ijms13032973 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Identification and Characterization
CONTRACTING ORGANIZATION: Cold Spring Harbor Laboratory Cold Spring Harbor, NY 11724
 AD Award Number: W81XWH-05-1-0256 TITLE: Dicer in Mammary Tumor Stem Cell Maintenance PRINCIPAL INVESTIGATOR: Elizabeth P. Murchison CONTRACTING ORGANIZATION: Cold Spring Harbor Laboratory Cold Spring
AD Award Number: W81XWH-05-1-0256 TITLE: Dicer in Mammary Tumor Stem Cell Maintenance PRINCIPAL INVESTIGATOR: Elizabeth P. Murchison CONTRACTING ORGANIZATION: Cold Spring Harbor Laboratory Cold Spring
On the Reproducibility of TCGA Ovarian Cancer MicroRNA Profiles
 On the Reproducibility of TCGA Ovarian Cancer MicroRNA Profiles Ying-Wooi Wan 1,2,4, Claire M. Mach 2,3, Genevera I. Allen 1,7,8, Matthew L. Anderson 2,4,5 *, Zhandong Liu 1,5,6,7 * 1 Departments of Pediatrics
On the Reproducibility of TCGA Ovarian Cancer MicroRNA Profiles Ying-Wooi Wan 1,2,4, Claire M. Mach 2,3, Genevera I. Allen 1,7,8, Matthew L. Anderson 2,4,5 *, Zhandong Liu 1,5,6,7 * 1 Departments of Pediatrics
RECAP (1)! In eukaryotes, large primary transcripts are processed to smaller, mature mrnas.! What was first evidence for this precursorproduct
 RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
RECAP (1) In eukaryotes, large primary transcripts are processed to smaller, mature mrnas. What was first evidence for this precursorproduct relationship? DNA Observation: Nuclear RNA pool consists of
Ch. 18 Regulation of Gene Expression
 Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Differential expression of mirnas from the pri-mir-17-92a locus.
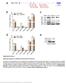 Supplementary Figure 1 Differential expression of mirnas from the pri-mir-17-92a locus. (a) The mir-17-92a expression unit in the third intron of the host mir-17hg transcript. (b,c) Impact of knockdown
Supplementary Figure 1 Differential expression of mirnas from the pri-mir-17-92a locus. (a) The mir-17-92a expression unit in the third intron of the host mir-17hg transcript. (b,c) Impact of knockdown
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing
 Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing Matthew W. Endres 1, Brian D. Gregory 2,3, Zhihuan Gao 1, Amy Wahba Foreman
Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing Matthew W. Endres 1, Brian D. Gregory 2,3, Zhihuan Gao 1, Amy Wahba Foreman
Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN
 Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN Institute for Brain Disorders and Neural Regeneration F.M. Kirby Program in Neural
Epigenetic Principles and Mechanisms Underlying Nervous System Function in Health and Disease Mark F. Mehler MD, FAAN Institute for Brain Disorders and Neural Regeneration F.M. Kirby Program in Neural
Eukaryotic Gene Regulation
 Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
Eukaryotic Gene Regulation Chapter 19: Control of Eukaryotic Genome The BIG Questions How are genes turned on & off in eukaryotes? How do cells with the same genes differentiate to perform completely different,
RNA- seq Introduc1on. Promises and pi7alls
 RNA- seq Introduc1on Promises and pi7alls RNA gives informa1on on which genes that are expressed How DNA get transcribed to RNA (and some1mes then translated to proteins) varies between e. g. - Tissues
RNA- seq Introduc1on Promises and pi7alls RNA gives informa1on on which genes that are expressed How DNA get transcribed to RNA (and some1mes then translated to proteins) varies between e. g. - Tissues
RNA Processing in Eukaryotes *
 OpenStax-CNX module: m44532 1 RNA Processing in Eukaryotes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you
OpenStax-CNX module: m44532 1 RNA Processing in Eukaryotes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you
Bioinformation by Biomedical Informatics Publishing Group
 Predicted RNA secondary structures for the conserved regions in dengue virus Pallavi Somvanshi*, Prahlad Kishore Seth Bioinformatics Centre, Biotech Park, Sector G, Jankipuram, Lucknow 226021, Uttar Pradesh,
Predicted RNA secondary structures for the conserved regions in dengue virus Pallavi Somvanshi*, Prahlad Kishore Seth Bioinformatics Centre, Biotech Park, Sector G, Jankipuram, Lucknow 226021, Uttar Pradesh,
In silico exploration of mirna from EST data of avocado and predicting its cross-kingdom effects on human
 2017; 6(9): 543-548 ISSN (E): 2277-7695 ISSN (P): 2349-8242 NAAS Rating 2017: 5.03 TPI 2017; 6(9): 543-548 2017 TPI www.thepharmajournal.com Received: 17-07-2017 Accepted: 18-08-2017 Drushti H. Bhatt Neha
2017; 6(9): 543-548 ISSN (E): 2277-7695 ISSN (P): 2349-8242 NAAS Rating 2017: 5.03 TPI 2017; 6(9): 543-548 2017 TPI www.thepharmajournal.com Received: 17-07-2017 Accepted: 18-08-2017 Drushti H. Bhatt Neha
Marta Puerto Plasencia. microrna sponges
 Marta Puerto Plasencia microrna sponges Introduction microrna CircularRNA Publications Conclusions The most well-studied regions in the human genome belong to proteincoding genes. Coding exons are 1.5%
Marta Puerto Plasencia microrna sponges Introduction microrna CircularRNA Publications Conclusions The most well-studied regions in the human genome belong to proteincoding genes. Coding exons are 1.5%
Alternative Splicing and Genomic Stability
 Alternative Splicing and Genomic Stability Kevin Cahill cahill@unm.edu http://dna.phys.unm.edu/ Abstract In a cell that uses alternative splicing, the total length of all the exons is far less than in
Alternative Splicing and Genomic Stability Kevin Cahill cahill@unm.edu http://dna.phys.unm.edu/ Abstract In a cell that uses alternative splicing, the total length of all the exons is far less than in
Genome-wide analysis of mirnas in Carya cathayensis
 Sun et al. BMC Plant Biology (2017) 17:228 DOI 10.1186/s12870-017-1180-6 RESEARCH ARTICLE Open Access Genome-wide analysis of mirnas in Carya cathayensis Zhi-chao Sun 1, Liang-sheng Zhang 1,2* and Zheng-jia
Sun et al. BMC Plant Biology (2017) 17:228 DOI 10.1186/s12870-017-1180-6 RESEARCH ARTICLE Open Access Genome-wide analysis of mirnas in Carya cathayensis Zhi-chao Sun 1, Liang-sheng Zhang 1,2* and Zheng-jia
omiras: MicroRNA regulation of gene expression
 omiras: MicroRNA regulation of gene expression Sören Müller, Goethe University of Frankfurt am Main Molecular Bioinformatics Group, Institute of Computer Science Plant Molecular Biology Group, Institute
omiras: MicroRNA regulation of gene expression Sören Müller, Goethe University of Frankfurt am Main Molecular Bioinformatics Group, Institute of Computer Science Plant Molecular Biology Group, Institute
