The Hypoglycemic Phenotype Is Islet Cell Autonomous in Short-Chain Hydroxyacyl-CoA Dehydrogenase Deficient Mice
|
|
|
- Lisa O’Brien’
- 5 years ago
- Views:
Transcription
1 1672 Diabetes Volume 65, June 2016 Anders Molven, 1,2,3,4 Jennifer Hollister-Lock, 1 Jiang Hu, 1 Rachael Martinez, 1 Pål R. Njølstad, 3,5 Chong Wee Liew, 1,6 Gordon Weir, 1 and Rohit N. Kulkarni 1,7,8 The Hypoglycemic Phenotype Is Islet Cell Autonomous in Short-Chain Hydroxyacyl-CoA Dehydrogenase Deficient Mice Diabetes 2016;65: DOI: /db ISLET STUDIES Congenital hyperinsulinism of infancy (CHI) can be caused by inactivating mutations in the gene encoding short-chain 3-hydroxyacyl-CoA dehydrogenase (SCHAD), a ubiquitously expressed enzyme involved in fatty acid oxidation. The hypersecretion of insulin may be explained by a loss of interaction between SCHAD and glutamate dehydrogenase in the pancreatic b-cells. However, there is also a general accumulation of metabolites specific for the enzymatic defect in affected individuals. It remains to be explored whether hypoglycemia in SCHAD CHI can be uncoupled from the systemic effect on fatty acid oxidation. We therefore transplanted islets from global SCHAD knockout (SCHADKO) mice into mice with streptozotocin-induced diabetes. After transplantation, SCHADKO islet recipients exhibited significantly lower random and fasting blood glucose compared with mice transplanted with normal islets or nondiabetic, nontransplanted controls. Furthermore, intraperitoneal glucose tolerance was improved in animals receiving SCHADKO islets compared with those receiving normal islets. Graft b-cell proliferation and apoptosis rates were similar in the two transplantation groups. We conclude that hypoglycemia in SCHAD-CHI is islet cell autonomous. Persistent hyperinsulinemic hypoglycemia in neonates, often termed congenital hyperinsulinism of infancy (CHI), comprises a group of inherited disorders in which the hallmarks are persistent hypoglycemia and inappropriately elevated insulin secretion (1). Both diagnosis and treatment of CHI are complicated. Medications tend to be ineffective, and partial or subtotal surgical resection of the pancreas mayeventuallybeneededinmanypatients(1). Mutations in nine different genes can cause CHI (2). These genes are expressed in the pancreatic b-cell, where they are involved in the regulation of insulin secretion. About half of the CHI cases are channelopathies being caused by inactivating mutations in the ABCC8 and KCNJ11 genes that encode subunits of the b-cells K ATP channel (2,3). Most of the other genetic CHI etiologies can be classified as metabolopathies, as they directly involve dysregulated metabolic pathways due to mutated enzyme genes (2). A classic example of the latter category is CHI caused by recessive mutations in HADH (4,5). This gene encodes short-chain 3-hydroxyacyl-CoA dehydrogenase (SCHAD), a mitochondrial enzyme that catalyzes the third of the four steps in fatty acid oxidation. More than 20 patients with SCHAD deficiency have now been reported. Intriguingly, they exhibit leucine-sensitive, diazoxide-responsive CHI rather than a phenotype typical for fatty acid oxidation disorders (6). The mechanism behind SCHAD CHI remained enigmatic for many years until Li et al. (7) investigated a global knockout mouse model (SCHADKO) and demonstrated that SCHAD serves to inhibit the enzyme glutamate dehydrogenase (GDH), another protein directly implicated in CHI (8). 1 Islet Cell and Regenerative Biology, Joslin Diabetes Center, Harvard Stem Cell Institute, Harvard Medical School, Boston, MA 2 Gade Laboratory for Pathology, Department of Clinical Medicine, University of Bergen, Bergen, Norway 3 KG Jebsen Center for Diabetes Research, Department of Clinical Science, University of Bergen, Bergen, Norway 4 Department of Pathology, Haukeland University Hospital, Bergen, Norway 5 Department of Pediatrics, Haukeland University Hospital, Bergen, Norway 6 Department of Physiology and Biophysics, University of Illinois at Chicago, Chicago, IL 7 Department of Medicine, Brigham and Women s Hospital, Harvard Medical School, Boston, MA 8 Harvard Stem Cell Institute, Boston, MA Corresponding author: Anders Molven, anders.molven@uib.no. Received 26 October 2015 and accepted 4 March by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered.
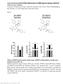
2 diabetes.diabetesjournals.org Molven and Associates 1673 However, HADH is ubiquitously expressed, suggesting that SCHAD deficiency is a metabolic disease with possible implications outside the context of the pancreatic b-cell. As such, in affected individuals there is abnormal accumulation of 3-hydroxybutyryl-carnitine in blood and 3-hydroxyglutaric acid in urine metabolites that are both directly attributable to the enzymatic defect (4 6). To explore whether the impact of global knockout of the SCHAD enzyme on glucose homeostasis is secondary only to defects in the islets of Langerhans, we transplanted SCHADKO islets into mice with streptozotocin (STZ)- induced diabetes. RESEARCH DESIGN AND METHODS Mouse Strains Islet donor mice were of the SCHADKO strain (mixed B6/129 background) reported by Li et al. (7). Mice were bred as Hadh +/2 heterozygotes and Hadh +/+ (wild type [WT]), and Hadh 2/2 (SCHADKO) offspring were used for the experiments. Genotyping was performed as previously described (7). Recipient animals were male ICR-SCID mice purchased from Taconic (Hudson, NY). Diabetes was induced 1 week before transplantation by injection of 180 mg/kg i.p. STZ (Sigma-Aldrich, St. Louis, MO), freshly dissolved in sodium citrate buffer (ph 4.5). Morning blood glucose was determined in tail vein blood and measured by a OneTouch Ultra glucometer (LifeScan, Milpitas, CA). Recipient mice were considered diabetic when their random fed blood glucose level exceeded 350 mg/dl on at least two consecutive days. The animals were kept on a 12-h light/dark cycle at 22 C and had ad libitum access to food and water. All animal experiments were approved by the Joslin Institutional Animal Care and Use Committee and were in accordance with National Institutes of Health guidelines. Isolation of Islets and Transplantation Islets were isolated from 10- to 12-week-old male and female SCHADKO and WT mice as previously described (9). Four hundred islet equivalents were transplanted under the left kidney capsule of STZ-induced diabetic mice (10). The islets were gently centrifuged into a capillary plastic tubing shortly before transplantation, and graft volume was calculated. Random fed blood glucose and body weight were monitored on a weekly basis until nephrectomy of the islet graft containing kidney in week 11. Recipient mice were considered cured of diabetes when the glucose level was,200 mg/dl for at least 2 consecutive weeks. The animals were killed after 3 consecutive days of hyperglycemia (.200 mg/dl) postnephrectomy. Glucose Tolerance Testing Donors We used a similar approach to that reported by Li et al. (7) and performed glucose tolerance testing by oral administration of 20% dextrose at a dose of 2 g/kg body wt 3 weeks before transplantation. Male WT and SCHADKO mice were fasted overnight (16 18 h) with water available ad libitum. The next morning blood glucose was measured before (time point 0) and 15, 30, 60, and 120 min after glucose administration. Recipients To avoid the stress of oral gavage in recipients that underwent surgery and consistent with the routine approach in our laboratory, we performed intraperitoneal glucose tolerance testing (IPGTT) 10 weeks posttransplantation. The mice were fasted overnight (16 18 h), and blood glucose was measured before (time point 0) and 15, 30, 60, 90, and 120 min after injection of glucose at a dose of 2 g/kg body wt i.p. At 0, 30, and 90 min, ml blood was collected for insulin measurement. Plasma insulin was measured by the Diabetes Research Center Specialized Assay Core at Joslin Diabetes Center using the rodent insulin ELISA kit (catalog no ) from Crystal Chem (Downers Grove, IL). Tissue Processing and Immunohistochemistry Upon nephrectomy, the recipient kidneys were immediately fixed in 4% paraformaldehyde solution for 2 h and then stored in PBS at 4 C until processing, paraffin embedding, and sectioning at 5 mm thickness. Four WT grafts and three from the SCHADKO group were recovered after final sectioning and insulin staining. Primary antibody against SCHAD (catalog no. ab154088, rabbit polyclonal, Abcam, Cambridge, MA; dilution 1:1,000) was applied overnightat4 Cfollowedbysecondaryantibody(catalogno ,JacksonImmunoResearch,WestGrove,PA; dilution 1:400) and signal amplification by the TSA Fluorescein System (catalog no. NEL701A; PerkinElmer, Boston, MA). Primary antibody against insulin (catalog no. ab7842, guinea pig polyclonal, Abcam; dilution 1:400) was then applied at room temperature for 1 h, followed by secondary antibody (catalog no , Jackson Immuno- Research; dilution 1:400). For glucagon and Ki-67 staining, mouse monoclonal antibodies from, respectively, Sigma- Aldrich (catalog no. G2654, 1:8,000) and BD Biosciences, San Jose, CA (catalog no , 1:50), were used. Secondary antibody was from Jackson ImmunoResearch (catalog no , 1:400). TUNEL staining was done with the ApopTag Fluorescein In Situ Apoptosis Detection kit (Merck Millipore, Darmstadt, Germany). Statistics Data are presented as mean 6 SEM. Statistical significance between groups was tested with unpaired two-tailed Student t test. RESULTS We confirmed that the donor (SCHADKO) mice exhibited the previously reported hypoglycemic phenotype (7) by measuring blood glucose levels after a 24-h fast and observing a significant difference between the WT and SCHADKO animals ( vs mg/dl) (Fig. 1A). Oral glucose tolerance testing on overnight-fasted animals also showed
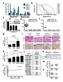
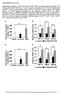
3 1674 Cell-Autonomous Hypoglycemia in SCHAD Deficiency Diabetes Volume 65, June 2016 Figure 1 Glucose metabolism in male donor mice at age 7 8 weeks, i.e., 3 weeks before transplantation. A: Blood glucose in WT (n = 16) and SCHADKO (n = 16) animals fasted for 24 h. B: Oral glucose tolerance in WT (blue line, n = 7) and SCHADKO (red line, n = 6) animals. C: Serum insulin levels after an overnight fast in WT (n = 7) and SCHADKO (n = 6) animals. *Statistical significance at P = ***Statistical significance at P = significantly lower blood glucose levels in the SCHADKO mice at 15 and 30 min (Fig. 1B). The glycemic response evaluated as area under the curve, using basal glucose level as baseline, appeared lower in the SCHADKO animals but did not reach statistical significance (WT 18, ,860 vs. SCHADKO 14, ,230; P =0.20).Again,thiswassimilar to the previous observation (7). Upon fasting, circulating insulin levels were low and not significantly different between the groups (Fig. 1C). The insulin-to-glucose ratio, however, was different between WT and SCHADKO ( vs ; P = 0.05). Next, we performed the transplantation experiment using 10- to 12-week-old SCHADKO animals or their WT littermates as islet donors and diabetic ICR-SCID mice as recipients. Mean transplanted islet volumes were mm 3 for WT grafts vs mm 3 for SCHADKO grafts (P = 0.52). Among 12 recipients transplanted with WT islets, 6 (50%) had exhibited stable normoglycemia 3 weeks after transplantation. The corresponding cure rate among SCHADKO recipients was six of eight (75%) transplanted animals. Both experimental groups showed a fall in blood glucose by 1 week after transplantation. Starting at 3 weeks, we observed a significant difference in random blood glucose levels between the groups at all but one time point, with the mice receiving SCHADKO islets always exhibiting lower levels (Fig. 2A). The average difference over the 3- to 11- week period between the two transplantation groups was 56 mg/dl. Random blood glucose values in the nontransplanted ICR-SCID control mice were generally between the values observed for the two transplanted groups. The small spikeinbloodglucoseobservedinweek10inbothtransplanted groups likely reflects a stress response as a consequence of the IPGTT being performed the day prior to the weekly measurement of random blood glucose. In the days after STZ injection, the mice lost a substantial part of their body weight due to the development of diabetes (not shown). However, after returning to normoglycemia upon islet transplantation, they rapidly regained weight and at week 8 had caught up to the body weights of nontransplanted control mice (Fig. 2C). The rate of weight gain was similar in the two transplanted groups. Five weeks after transplantation, blood glucose levels were measured in 24-h fasted mice. The animals receiving SCHADKO islets had significantly lower levels than both WT transplanted and nontransplanted mice ( vs and mg/dl, respectively) (Fig. 3A). IPGTT performed at week 10 posttransplantation in overnight-fasted animals revealed that mice transplanted with WT islets exhibited significantly impaired glucose tolerance compared with both SCHADKO recipients and nontransplanted controls (area under the curve 35, ,730 vs. 27, ,520 [P = 0.05] and 24, ,420 [P = 0.03], respectively) (Fig. 3B). The glucose tolerance was similar in the SCHADKO recipients and nontransplanted controls (P = 0.45), and the blood glucose levels were not significantly different at any of the time points (Fig. 3B). Circulating insulin levels were comparable at baseline (time 0) (Fig. 3C), and stimulation with glucose led to a similar secretory response at 30 and 90 mins in all three groups (Fig. 3C). To confirm that the effects on normalization of the blood glucose were indeed secondary to transplanted islets, we removed the recipient kidney in both transplantation groups in week 11. Shortly thereafter, all mice became hyperglycemic and started to lose weight (Fig. 2B and D). Four days postnephrectomy, random blood glucose levels were generally.350 mg/dl and the animals were killed. The recipient kidneys were sectioned and immunostained for insulin and SCHAD, confirming that
4 diabetes.diabetesjournals.org Molven and Associates 1675 Figure 2 Effect on random blood glucose (A and B) and body weight (C and D) of transplanting SCHADKO and WT islets into diabetic ICR-SCID mice. A: Measurements of random blood glucose over 11 weeks after transplantation. B: Measurements of random blood glucose after nephrectomy. C: Body weight changes over 11 weeks after transplantation. D: Measurements of body weight after nephrectomy. Time point 0 refers to the date of transplantation (A and C) or nephrectomy (B and D). The red and blue lines represent data from mice that received, respectively, SCHADKO (n =6)orWT(n = 6) islets and returned to normoglycemia. In panels A and C, the gray line shows measurements in a control group of age-matched, nontransplanted, and euglycemic ICR-SCID mice (n = 8). *Statistical significance at P = **Statistical significance at P = 0.01 comparing the SCHADKO and WT transplantation groups. SCHAD protein was not expressed in the islet grafts derived from SCHADKO animals (Fig. 4A). We observed no systematic difference between groups with regard to the size of recovered grafts or b-cells counted per section. The b-cell proliferation rate, as evaluated by double staining for insulin and Ki-67, was between 0 and 0.5%, with no differences between groups. No apopotic b-cells were detected in any of the grafts by the TUNEL assay. Finally, there was no obvious difference in b-cell to a-cell ratio between WT and SCHADKO transplants (Fig. 4B).
5 1676 Cell-Autonomous Hypoglycemia in SCHAD Deficiency Diabetes Volume 65, June 2016 Figure 3 Glucose metabolism in diabetic mice that received either normal (WT, n = 6; blue) or SCHADKO (n = 6; red) islets and in agematched, nontransplanted, and euglycemic control ICR-SCID mice (Ctr) (n = 8; gray). A: Fasting blood glucose (24 h). Measurements were done 5 weeks after transplantation. **Statistical significance at P = B: Intraperitoneal glucose tolerance at week 10 posttransplantation. *Statistical significance at P = 0.05 comparing the WT group with the controls. C: Serum insulin levels during IPGTT (0, 30, and 90 min after glucose injection). Percentages of glucagon-positive cells (fraction of all insulinor glucagon-positive cells) were 6.3, 9.5, 13.0, and 14.3% for WT grafts and 13.7, 15.1, and 17.4% for SCHADKO grafts. Single cells positive for both markers were not seen in any section. DISCUSSION The phenotype of CHI caused by SCHAD deficiency has been difficult to explain as a defect in fatty acid degradation. Initially, it was suggested that the protein acted as a coupling factor between fatty acid oxidation and glucose metabolism in the pancreatic b-cell and that the disease mechanism was K ATP channel independent and involved accumulation of certain metabolites (4,5,11). However, more recent studies have reported that SCHAD interacts with and inhibits the enzyme GDH (7,12), a finding consistent with the protein-sensitive hyperinsulinemic hypoglycemia noted in some SCHAD-deficient patients (13). However, it is well established that fatty acids and their metabolites can modulate insulin secretion (14 16), and newly discovered esters of hydroxy-fatty acids were described to have a surprising antidiabetes effect (17). Because some fatty acid related metabolites are clearly increased both in humans with SCHAD deficiency (4 6) and in the corresponding animal model (7), it is possible that systemic metabolic changes play a role in the persistent hyperinsulinemic hypoglycemia characteristic of the disease. We therefore directly addressed this issue by transplanting pancreatic islets from either SCHADKO or WT animals into STZ-induced diabetic recipients. The SCHADKO donor mice in our studies exhibited the phenotype reported previously (7), i.e., lower blood glucose levels in the fasted state and similar glycemic responses compared with WT mice (Fig. 1A and B). In contrast to Li et al. (7), we did not detect an increased basal insulin level in SCHADKO mice (Fig. 1C) but did observe an inappropriately elevated insulin-to-glucose ratio (7). Moreover, 3 weeks after transplantation the SCHADKO islet-transplanted mice presented a significantly lower random fed glucose level than mice transplanted with WT islets (Fig. 2A). Upon removal of the kidney that had received the graft, all animals in both transplantation groups rapidlylostweightandbecame overtly diabetic (Fig. 2B and D). Together, these results support the notion that the established euglycemia depended mostly on the functional islet graft. We also considered the possibility that a quantitative effect may explain the lower blood glucose levels in the SCHADKO group. Thus, although the average graft volume pretransplantation was ;10% smaller in the WT group comparedwiththeschadkogroup,thedifferencedid not reach statistical significance (P = 0.52). Evaluation of the grafts recovered after nephrectomy did not reveal significant differences in b-cell number, rate of b-cell proliferation or apoptosis, or a-cell to b-cell ratio between groups. These data suggest that it is unlikely that graft size/ composition or b-cell proliferation/survival directly contributes to the differences in blood glucose observed. Despite these observations, we cannot completely exclude the possibility that SCHADKO islets possess inherent biological properties, independent of the blood glucose lowering impact of the genetic defect, that make them functionally superior to WT islets in our transplantation model. We propose that SCHADKO islets alone are sufficient to recapitulate the hypoglycemic phenotype observed in the animal model of global SCHAD deficiency. Indeed, transplantation with SCHADKO islets was able to maintain glucose tolerance in the normal range in the recipients and was comparable with nontransplanted controls, in contrast to the attenuated glucose tolerance in animals receiving WT islets. Whether endogenous metabolites produced from the
6 diabetes.diabetesjournals.org Molven and Associates 1677 be addressed in more detail by studying mice with b-cell specific knockout of the Hadh gene. Acknowledgments. The SCHADKO mouse strain was kindly provided by Charles Stanley (Children s Hospital of Philadelphia) and Arnold W. Strauss (University of Cincinnati College of Medicine). The authors are grateful to Brooke Sullivan (Joslin Diabetes Center) for assistance with islet transplantation. Funding. The study was supported by a Fulbright fellowship to A.M. and by grants from the Novo Nordisk Foundation (NNF15OC ), Western Norway Regional Health Authority, KG Jebsen Foundation, and The Research Council of Norway (Norges Forskningsråd) (FRIMEDBIO ). P.R.N. was supported by a European Research Council Advanced Grant. C.W.L. was supported by a grant from the National Institutes of Health (K99-DK ). R.N.K. was supported by National Institutes of Health grants R01-DK and R01-DK Duality of Interest. No potential conflicts of interest relevant to this article were reported. Author Contributions. A.M., J.H.-L., J.H., and R.M. researched data. A.M., P.R.N., C.W.L., G.W., and R.N.K. conceived the project. A.M. and R.N.K. directed and supervised the study. A.M. wrote the manuscript. All authors reviewed and edited the manuscript. A.M. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Figure 4 Immunohistochemistry performed on islet grafts recovered after nephrectomy in the 11th week of the study. Representative sections from the WT and SCHADKO transplantation groups are shown. The dashed white lines indicate outline of the kidney parenchyma. Scale bars are 50 mm. A: Staining performed with fluorescent antibodies against SCHAD (green) and insulin (INS) (red). B: Staining performed with fluorescent antibodies against glucagon (GCG) (green) and insulin (red). SCHAD-deficient islets contribute to the phenotype warrants further investigation. Our study does not address a potential role of SCHAD in b-cells versus other cell types in the islet. In this regard, it is notable that insulin-independent glucagon suppression contributes to hypoglycemia in a mouse model of CHI caused by GDH mutation, suggesting that a signal generated via b-cell mitochondria and GDH specifically inhibits a-cells (18). As SCHAD inhibits GDH, impaired a-cell function may also be relevant for SCHAD deficiency, and of note, the first family described with the disease was regarded as glucagon deficient (19). The question of how SCHAD might affect glucagon secretion could References 1. Kapoor RR, James C, Hussain K. Advances in the diagnosis and management of hyperinsulinemic hypoglycemia. Nat Clin Pract Endocrinol Metab 2009;5: Rahman SA, Nessa A, Hussain K. Molecular mechanisms of congenital hyperinsulinism. J Mol Endocrinol 2015;54:R119 R Sandal T, Laborie LB, Brusgaard K, et al. The spectrum of ABCC8 mutations in Norwegian patients with congenital hyperinsulinism of infancy. Clin Genet 2009;75: Clayton PT, Eaton S, Aynsley-Green A, et al. Hyperinsulinism in short-chain L-3-hydroxyacyl-CoA dehydrogenase deficiency reveals the importance of betaoxidation in insulin secretion. J Clin Invest 2001;108: Molven A, Matre GE, Duran M, et al. Familial hyperinsulinemic hypoglycemia caused by a defect in the SCHAD enzyme of mitochondrial fatty acid oxidation. Diabetes 2004;53: Molven A, Helgeland G, Sandal T, Njølstad PR. The molecular genetics and pathophysiology of congenital hyperinsulinism caused by short-chain 3- hydroxyacyl-coa dehydrogenase deficiency. In Monogenic Hyperinsulinemic Hypoglycemia Disorders. Frontiers in Diabetes. 21. Stanley CA, De León DD, Eds. Basel, Switzerland, Karger, 2012, p Li C, Chen P, Palladino A, et al. Mechanism of hyperinsulinism in shortchain 3-hydroxyacyl-CoA dehydrogenase deficiency involves activation of glutamate dehydrogenase. J Biol Chem 2010;285: Palladino AA, Stanley CA. The hyperinsulinism/hyperammonemia syndrome. Rev Endocr Metab Disord 2010;11: Gotoh M, Ohzato H, Dono K, et al. Successful islet isolation from preserved rat pancreas following pancreatic ductal collagenase at the time of harvesting. Horm Metab Res Suppl 1990;25: Davalli AM, Ogawa Y, Scaglia L, et al. Function, mass, and replication of porcine and rat islets transplanted into diabetic nude mice. Diabetes 1995;44: Martens GA, Vervoort A, Van de Casteele M, et al. Specificity in beta cell expression of L-3-hydroxyacyl-CoA dehydrogenase, short chain, and potential role in down-regulating insulin release. J Biol Chem 2007;282: Filling C, Keller B, Hirschberg D, et al. Role of short-chain hydroxyacyl CoA dehydrogenases in SCHAD deficiency. Biochem Biophys Res Commun 2008;368: Heslegrave AJ, Kapoor RR, Eaton S, et al. Leucine-sensitive hyperinsulinaemic hypoglycaemia in patients with loss of function mutations in 3-Hydroxyacyl-CoA Dehydrogenase. Orphanet J Rare Dis 2012;7:25
7 1678 Cell-Autonomous Hypoglycemia in SCHAD Deficiency Diabetes Volume 65, June Graciano MF, Valle MM, Kowluru A, Curi R, Carpinelli AR. Regulation of insulin secretion and reactive oxygen species production by free fatty acids in pancreatic islets. Islets 2011;3: Las G, Mayorek N, Dickstein K, Bar-Tana J. Modulation of insulin secretion by fatty acyl analogs. Diabetes 2006;55: Nolan CJ, Madiraju MS, Delghingaro-Augusto V, Peyot ML, Prentki M. Fatty acid signaling in the beta-cell and insulin secretion. Diabetes 2006;55(Suppl. 2): S16 S Yore MM, Syed I, Moraes-Vieira PM, et al. Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cell 2014;159: Kibbey RG, Choi CS, Lee HY, et al. Mitochondrial GTP insensitivity contributes to hypoglycemia in hyperinsulinemia hyperammonemia by inhibiting glucagon release. Diabetes 2014;63: Vidnes J, Øyasaeter S. Glucagon deficiency causing severe neonatal hypoglycemia in a patient with normal insulin secretion. Pediatr Res 1977;11:
Overview. o Limitations o Normal regulation of blood glucose o Definition o Symptoms o Clinical forms o Pathophysiology o Treatment.
 Pål R. Njølstad MD PhD KG Jebsen Center for Diabetes Research University of Bergen, Norway Depertment of Pediatrics Haukeland University Hospital Broad Institute of Harvard & MIT Cambridge, MA, USA Hypoglycemia
Pål R. Njølstad MD PhD KG Jebsen Center for Diabetes Research University of Bergen, Norway Depertment of Pediatrics Haukeland University Hospital Broad Institute of Harvard & MIT Cambridge, MA, USA Hypoglycemia
Leucine-sensitive hyperinsulinaemic hypoglycaemia in patients with loss of function mutations in 3-Hydroxyacyl-CoA Dehydrogenase
 Heslegrave et al. Orphanet Journal of Rare Diseases 2012, 7:25 RESEARCH Open Access Leucine-sensitive hyperinsulinaemic hypoglycaemia in patients with loss of function mutations in 3-Hydroxyacyl-CoA Dehydrogenase
Heslegrave et al. Orphanet Journal of Rare Diseases 2012, 7:25 RESEARCH Open Access Leucine-sensitive hyperinsulinaemic hypoglycaemia in patients with loss of function mutations in 3-Hydroxyacyl-CoA Dehydrogenase
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets
 Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
Supplementary Table 1. Primer sequences for conventional RT-PCR on mouse islets Gene 5 Forward 3 5 Reverse 3.T. Product (bp) ( C) mnox1 GTTCTTGGGCTGCCTTGG GCTGGGGCGGCGG 60 300 mnoxa1 GCTTTGCCGCGTGC GGTTCGGGTCCTTTGTGC
SUPPLEMENTARY DATA. Supplementary experimental procedures
 Supplementary experimental procedures Glucose tolerance, insulin secretion and insulin tolerance tests Glucose tolerance test (GTT) and glucose stimulated insulin secretion (GSIS) was measured in over-night
Supplementary experimental procedures Glucose tolerance, insulin secretion and insulin tolerance tests Glucose tolerance test (GTT) and glucose stimulated insulin secretion (GSIS) was measured in over-night
Congenital hyperinsulinism
 SWISS SOCIETY OF NEONATOLOGY Winner of the Case of the Year Award 2012 Congenital hyperinsulinism FEBRUARY 2012 2 Morgillo D, Berger TM, Caduff JH, Barthlen W, Mohnike K, Mohnike W, Neonatal and Pediatric
SWISS SOCIETY OF NEONATOLOGY Winner of the Case of the Year Award 2012 Congenital hyperinsulinism FEBRUARY 2012 2 Morgillo D, Berger TM, Caduff JH, Barthlen W, Mohnike K, Mohnike W, Neonatal and Pediatric
Inhibition of DYRK1A stimulates human beta-cell proliferation
 Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Abbreviations: P- paraffin-embedded section; C, cryosection; Bio-SA, biotin-streptavidin-conjugated fluorescein amplification.
 Supplementary Table 1. Sequence of primers for real time PCR. Gene Forward primer Reverse primer S25 5 -GTG GTC CAC ACT ACT CTC TGA GTT TC-3 5 - GAC TTT CCG GCA TCC TTC TTC-3 Mafa cds 5 -CTT CAG CAA GGA
Supplementary Table 1. Sequence of primers for real time PCR. Gene Forward primer Reverse primer S25 5 -GTG GTC CAC ACT ACT CTC TGA GTT TC-3 5 - GAC TTT CCG GCA TCC TTC TTC-3 Mafa cds 5 -CTT CAG CAA GGA
Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Patient Demographics 5/12/2016
 Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Pediatric Endocrine Nursing Society May 14, 2016 Enyo Dzata, MSN, CRNP Congenital Hyperinsulinism Center Division
Beyond the Naked Eye: A Case Presentation on a Rare Form of Congenital Hyperinsulinism (HI) Pediatric Endocrine Nursing Society May 14, 2016 Enyo Dzata, MSN, CRNP Congenital Hyperinsulinism Center Division
Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches
 Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches during application of 500 µm Ca 2+ at the intracellular
Supplementary Figure 1: Steviol and stevioside potentiate TRPM5 in a cell-free environment. (a) TRPM5 currents are activated in inside-out patches during application of 500 µm Ca 2+ at the intracellular
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
Challenging diagnosis of congenital hyperinsulinism in two infants of diabetic mothers with rare pathogenic KCNJ11 and HNF4A gene variants
 Huerta-Saenz et al. International Journal of Pediatric Endocrinology (2018) 2018:5 https://doi.org/10.1186/s13633-018-0060-7 CASE REPORT Open Access Challenging diagnosis of congenital hyperinsulinism
Huerta-Saenz et al. International Journal of Pediatric Endocrinology (2018) 2018:5 https://doi.org/10.1186/s13633-018-0060-7 CASE REPORT Open Access Challenging diagnosis of congenital hyperinsulinism
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Remission in Non-Operated Patients with Diffuse Disease and Long-Term Conservative Treatment.
 5th Congenital Hyperinsulinism International Family Conference Milan, September 17-18 Remission in Non-Operated Patients with Diffuse Disease and Long-Term Conservative Treatment. PD Dr. Thomas Meissner
5th Congenital Hyperinsulinism International Family Conference Milan, September 17-18 Remission in Non-Operated Patients with Diffuse Disease and Long-Term Conservative Treatment. PD Dr. Thomas Meissner
Secondary Negative Effects of Isolation Enzyme (s) On Human Islets. A.N.Balamurugan
 Secondary Negative Effects of Isolation Enzyme (s) On Human Islets A.N.Balamurugan Human Islets Functional Mass Preservation 18-25 min 10 min 8-12 min 10-15 min 45-60 min pancreas in chamber 37º Sub-Optimal
Secondary Negative Effects of Isolation Enzyme (s) On Human Islets A.N.Balamurugan Human Islets Functional Mass Preservation 18-25 min 10 min 8-12 min 10-15 min 45-60 min pancreas in chamber 37º Sub-Optimal
Inflammation & Type 2 Diabetes Prof. Marc Y. Donath
 Inflammation & Type 2 Diabetes 1, MD Chief Endocrinology, Diabetes & Metabolism University Hospital Basel Petersgraben 4 CH-431 Basel, Switzerland MDonath@uhbs.ch Innate immunity as a sensor of metabolic
Inflammation & Type 2 Diabetes 1, MD Chief Endocrinology, Diabetes & Metabolism University Hospital Basel Petersgraben 4 CH-431 Basel, Switzerland MDonath@uhbs.ch Innate immunity as a sensor of metabolic
Advances in the diagnosis and management of hyperinsulinemic hypoglycemia
 Advances in the diagnosis and management of hyperinsulinemic hypoglycemia Ritika R Kapoor, Chela James and Khalid Hussain* SUMMARY Hyperinsulinemic hypoglycemia (HH) is a consequence of unregulated insulin
Advances in the diagnosis and management of hyperinsulinemic hypoglycemia Ritika R Kapoor, Chela James and Khalid Hussain* SUMMARY Hyperinsulinemic hypoglycemia (HH) is a consequence of unregulated insulin
Supplemental Information. b Cell Aging Markers Have Heterogeneous. Distribution and Are Induced by Insulin Resistance
 Cell Metabolism, Volume 25 Supplemental Information b Cell Aging Markers Have Heterogeneous Distribution and Are Induced by Insulin Resistance Cristina Aguayo-Mazzucato, Mark van Haaren, Magdalena Mruk,
Cell Metabolism, Volume 25 Supplemental Information b Cell Aging Markers Have Heterogeneous Distribution and Are Induced by Insulin Resistance Cristina Aguayo-Mazzucato, Mark van Haaren, Magdalena Mruk,
Surgery in Congenital Hyperinsulinismless. Winfried Barthlen
 Surgery in Congenital Hyperinsulinismless may be more Winfried Barthlen congenital hyperinsulinism - very rare (1:40.000) - uncontrolled insulin secretion - life threatening hypoglycemia symptoms - unconsciousness,
Surgery in Congenital Hyperinsulinismless may be more Winfried Barthlen congenital hyperinsulinism - very rare (1:40.000) - uncontrolled insulin secretion - life threatening hypoglycemia symptoms - unconsciousness,
RECENTLY, WE AND others described an unusual form
 0013-7227/01/$03.00/0 The Journal of Clinical Endocrinology & Metabolism 86(8):3724 3728 Printed in U.S.A. Copyright 2001 by The Endocrine Society Acute Insulin Responses to Leucine in Children with the
0013-7227/01/$03.00/0 The Journal of Clinical Endocrinology & Metabolism 86(8):3724 3728 Printed in U.S.A. Copyright 2001 by The Endocrine Society Acute Insulin Responses to Leucine in Children with the
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Mouse Models for Studying Human Islet Transplantation
 Mouse Models for Studying Human Islet Transplantation Ronald G. Gill, Joshua Beilke,, Nathan Kuhl,, Michelle Kerklo, and Mark M. Nicolls Barbara Davis Center for Childhood Diabetes University of Colorado
Mouse Models for Studying Human Islet Transplantation Ronald G. Gill, Joshua Beilke,, Nathan Kuhl,, Michelle Kerklo, and Mark M. Nicolls Barbara Davis Center for Childhood Diabetes University of Colorado
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO
 Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Diabetes: Definition Pathophysiology Treatment Goals. By Scott Magee, MD, FACE
 Diabetes: Definition Pathophysiology Treatment Goals By Scott Magee, MD, FACE Disclosures No disclosures to report Definition of Diabetes Mellitus Diabetes Mellitus comprises a group of disorders characterized
Diabetes: Definition Pathophysiology Treatment Goals By Scott Magee, MD, FACE Disclosures No disclosures to report Definition of Diabetes Mellitus Diabetes Mellitus comprises a group of disorders characterized
Strain Differences in the Diabetogenic Activity of Streptozotocin in Mice
 1110 Biol. Pharm. Bull. 29(6) 1110 1119 (2006) Vol. 29, No. 6 Strain Differences in the Diabetogenic Activity of Streptozotocin in Mice Koji HAYASHI, Rhyoji KOJIMA, and Mikio ITO* Laboratory of Analytical
1110 Biol. Pharm. Bull. 29(6) 1110 1119 (2006) Vol. 29, No. 6 Strain Differences in the Diabetogenic Activity of Streptozotocin in Mice Koji HAYASHI, Rhyoji KOJIMA, and Mikio ITO* Laboratory of Analytical
A novel case of compound heterozygous congenital hyperinsulinism without high insulin levels
 Brady et al. International Journal of Pediatric Endocrinology (2015) 2015:16 DOI 10.1186/s13633-015-0012-4 CASE REPORT Open Access A novel case of compound heterozygous congenital hyperinsulinism without
Brady et al. International Journal of Pediatric Endocrinology (2015) 2015:16 DOI 10.1186/s13633-015-0012-4 CASE REPORT Open Access A novel case of compound heterozygous congenital hyperinsulinism without
Use of Lanreotide (long acting Somatostatin analogue) in Congenital Hyperinsulinism (CHI)
 Use of Lanreotide (long acting Somatostatin analogue) in Congenital Hyperinsulinism (CHI) Dr Pratik Shah Clinical Research fellow in Hyperinsulinism Clinical Molecular Genetics Unit Institute of Child
Use of Lanreotide (long acting Somatostatin analogue) in Congenital Hyperinsulinism (CHI) Dr Pratik Shah Clinical Research fellow in Hyperinsulinism Clinical Molecular Genetics Unit Institute of Child
First-in-Class Treatment for Hyperinsulinemic Hypoglycemia. January 31, 2017
 358 First-in-Class Treatment for Hyperinsulinemic Hypoglycemia January 31, 2017 Forward-Looking Statement Certain statements contained herein including, but not limited to, expected licensing transactions,
358 First-in-Class Treatment for Hyperinsulinemic Hypoglycemia January 31, 2017 Forward-Looking Statement Certain statements contained herein including, but not limited to, expected licensing transactions,
Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus
 Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes
 Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes T Jourdan, G Godlewski, R Cinar, A Bertola, G Szanda, J Liu, J Tam, T Han, B Mukhopadhyay,
Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes T Jourdan, G Godlewski, R Cinar, A Bertola, G Szanda, J Liu, J Tam, T Han, B Mukhopadhyay,
Discussion & Conclusion
 Discussion & Conclusion 7. Discussion DPP-4 inhibitors augment the effects of incretin hormones by prolonging their half-life and represent a new therapeutic approach for the treatment of type 2 diabetes
Discussion & Conclusion 7. Discussion DPP-4 inhibitors augment the effects of incretin hormones by prolonging their half-life and represent a new therapeutic approach for the treatment of type 2 diabetes
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Future Therapies in the Treatment of Diabetes: Islet Transplantation
 Future Therapies in the Treatment of Diabetes: Islet Transplantation Michael R. Rickels, MD, MS Associate Professor of Medicine University of Pennsylvania Perelman School of Medicine Medical Director,
Future Therapies in the Treatment of Diabetes: Islet Transplantation Michael R. Rickels, MD, MS Associate Professor of Medicine University of Pennsylvania Perelman School of Medicine Medical Director,
Making Mature Human Islet Cells from Stem Cells to Model Disease and Treat Diabetes
 University of British Columbia Departments of Surgery and Cellular & Physiological Sciences Making Mature Human Islet Cells from Stem Cells to Model Disease and Treat Diabetes 2016 International Conference
University of British Columbia Departments of Surgery and Cellular & Physiological Sciences Making Mature Human Islet Cells from Stem Cells to Model Disease and Treat Diabetes 2016 International Conference
Therapeutic strategy to reduce Glucagon secretion
 Clinical focus on glucagon: α-cell as a companion of β-cell Therapeutic strategy to reduce Glucagon secretion Sunghwan Suh Dong-A University Conflict of interest disclosure None Committee of Scientific
Clinical focus on glucagon: α-cell as a companion of β-cell Therapeutic strategy to reduce Glucagon secretion Sunghwan Suh Dong-A University Conflict of interest disclosure None Committee of Scientific
Pancreatic Insulinoma Presenting. with Episodes of Hypoinsulinemic. Hypoglycemia in Elderly ---- A Case Report
 2008 19 432-436 Pancreatic Insulinoma Presenting with Episodes of Hypoinsulinemic Hypoglycemia in Elderly ---- A Case Report Chieh-Hsiang Lu 1, Shih-Che Hua 1, and Chung-Jung Wu 2,3 1 Division of Endocrinology
2008 19 432-436 Pancreatic Insulinoma Presenting with Episodes of Hypoinsulinemic Hypoglycemia in Elderly ---- A Case Report Chieh-Hsiang Lu 1, Shih-Che Hua 1, and Chung-Jung Wu 2,3 1 Division of Endocrinology
Glucose Homeostasis. Liver. Glucose. Muscle, Fat. Pancreatic Islet. Glucose utilization. Glucose production, storage Insulin Glucagon
 Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
Glucose Homeostasis Liver Glucose Glucose utilization Glucose production, storage Insulin Glucagon Muscle, Fat Pancreatic Islet Classification of Diabetes Type 1 diabetes Type 2 diabetes Other types of
Maturity-onset diabetes of the young (MODY) is a heterogeneous group
 Over the years, different forms of maturity-onset diabetes of the young (MODY) have been identified, with mutations in a number of different genes associated with a MODY-like phenotype. Depending on the
Over the years, different forms of maturity-onset diabetes of the young (MODY) have been identified, with mutations in a number of different genes associated with a MODY-like phenotype. Depending on the
Laboratory of Experimental Medicine, Brussels Free University, B-1070 Brussels', Belgium and Novo Nordisk A/S, DK-2880 Bagsvaerd, Denmark
 Vol. 43, No. 2, October 1997 Pages 233-240 CATONC AND SECRETORY EFFECTS OF 6-O-ACETYL-D-GLUCOSE N RAT PANCREATC SLETS Hassan JJAKL, Ole KRK and Willy J. MALASSE Laboratory of Experimental Medicine, Brussels
Vol. 43, No. 2, October 1997 Pages 233-240 CATONC AND SECRETORY EFFECTS OF 6-O-ACETYL-D-GLUCOSE N RAT PANCREATC SLETS Hassan JJAKL, Ole KRK and Willy J. MALASSE Laboratory of Experimental Medicine, Brussels
José Oberholzer, MD. Director Charles O. Strickler Transplant Center. Chief, Division of Transplantation
 José Oberholzer, MD Director Charles O. Strickler Transplant Center Chief, Division of Transplantation Professor of Surgery and Biomedical Engineering DISCLOSURES No consulting for, no payments, no stocks
José Oberholzer, MD Director Charles O. Strickler Transplant Center Chief, Division of Transplantation Professor of Surgery and Biomedical Engineering DISCLOSURES No consulting for, no payments, no stocks
Determination of serum insulin level by ELISA
 Practical course: Basic biochemical methods and ischemic heart models Determination of serum insulin level by ELISA A practical manual Tamas Csont, MD, PhD Supported by: HURO/0901/069/2.3.1 1 BACKGROUND
Practical course: Basic biochemical methods and ischemic heart models Determination of serum insulin level by ELISA A practical manual Tamas Csont, MD, PhD Supported by: HURO/0901/069/2.3.1 1 BACKGROUND
Transplantation of Microencapsulated Neonatal Porcine Islets in Patients with Type 1 Diabetes: Safety and Efficacy
 Transplantation of Microencapsulated Neonatal Porcine Islets in Patients with Type 1 Diabetes: Safety and Efficacy R.B Elliott, O Garkavenko, P Tan, N.N Skaletsky, A Guliev, B Draznin Auckland, New Zealand;
Transplantation of Microencapsulated Neonatal Porcine Islets in Patients with Type 1 Diabetes: Safety and Efficacy R.B Elliott, O Garkavenko, P Tan, N.N Skaletsky, A Guliev, B Draznin Auckland, New Zealand;
SHORT COMMUNICATION. J. C. Reubi & A. Perren & R. Rehmann & B. Waser & E. Christ & M. Callery & A. B. Goldfine & M. E. Patti
 Diabetologia (2010) 53:2641 2645 DOI 10.1007/s00125-010-1901-y SHORT COMMUNICATION Glucagon-like peptide-1 (GLP-1) receptors are not overexpressed in pancreatic islets from patients with severe hyperinsulinaemic
Diabetologia (2010) 53:2641 2645 DOI 10.1007/s00125-010-1901-y SHORT COMMUNICATION Glucagon-like peptide-1 (GLP-1) receptors are not overexpressed in pancreatic islets from patients with severe hyperinsulinaemic
Kidney. Heart. Lung. Sirt1. Gapdh. Mouse IgG DAPI. Rabbit IgG DAPI
 a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
a e Na V 1.5 Ad-LacZ Ad- 110KD b Scn5a/ (relative to Ad-LacZ) f 150 100 50 0 p = 0.65 Ad-LacZ Ad- c Heart Lung Kidney Spleen 110KD d fl/fl c -/- DAPI 20 µm Na v 1.5 250KD fl/fl Rabbit IgG DAPI fl/fl Mouse
THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals
 Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
The Effects of Exenatide on the Autoimmune Development of Diabetes. A Senior Honors Thesis
 The Effects of Exenatide on the Autoimmune Development of Diabetes A Senior Honors Thesis Presented in Partial Fulfillment of the Requirements for Graduation with Distinction in Microbiology in the Undergraduate
The Effects of Exenatide on the Autoimmune Development of Diabetes A Senior Honors Thesis Presented in Partial Fulfillment of the Requirements for Graduation with Distinction in Microbiology in the Undergraduate
A novel mutation of ABCC8 gene in a patient with diazoxide-unresponsive congenital hyperinsulinism
 Case report Park Korean JS, J et Pediatr al. Congenital 2016;59(Suppl hyperinsulinism 1):S116-120with a novel mutation of ABCC8 pissn 1738-1061 eissn 2092-7258 Korean J Pediatr A novel mutation of ABCC8
Case report Park Korean JS, J et Pediatr al. Congenital 2016;59(Suppl hyperinsulinism 1):S116-120with a novel mutation of ABCC8 pissn 1738-1061 eissn 2092-7258 Korean J Pediatr A novel mutation of ABCC8
Metabolic responses to leptin in obese db/db mice are strain dependent
 Am J Physiol Regulatory Integrative Comp Physiol 281: R115 R132, 2001. Metabolic responses to leptin in obese db/db mice are strain dependent RUTH B. S. HARRIS, TIFFANY D. MITCHELL, XIAOLANG YAN, JACOB
Am J Physiol Regulatory Integrative Comp Physiol 281: R115 R132, 2001. Metabolic responses to leptin in obese db/db mice are strain dependent RUTH B. S. HARRIS, TIFFANY D. MITCHELL, XIAOLANG YAN, JACOB
Supporting Information
 Supporting Information Soltani et al. 10.1073/pnas.1102715108 SI Experimental Procedures Evaluation of Mice and Drug Administration. IPGTT, insulin tolerance test, and glucagon tolerance test were performed
Supporting Information Soltani et al. 10.1073/pnas.1102715108 SI Experimental Procedures Evaluation of Mice and Drug Administration. IPGTT, insulin tolerance test, and glucagon tolerance test were performed
Cook Children s HI Center. Paul Thornton Medical Director Cook Children s Hyperinsulinism Center
 Cook Children s HI Center Paul Thornton Medical Director Cook Children s Hyperinsulinism Center Formed in Oct 2010 Cook Children s HI Center Mission: To provide excellence in medical care to patients with
Cook Children s HI Center Paul Thornton Medical Director Cook Children s Hyperinsulinism Center Formed in Oct 2010 Cook Children s HI Center Mission: To provide excellence in medical care to patients with
NIH Public Access Author Manuscript Diabetologia. Author manuscript; available in PMC 2014 February 01.
 NIH Public Access Author Manuscript Published in final edited form as: Diabetologia. 2013 February ; 56(2): 231 233. doi:10.1007/s00125-012-2788-6. Lipotoxicity impairs incretin signalling V. Poitout 1,2
NIH Public Access Author Manuscript Published in final edited form as: Diabetologia. 2013 February ; 56(2): 231 233. doi:10.1007/s00125-012-2788-6. Lipotoxicity impairs incretin signalling V. Poitout 1,2
Mechanism of hyperinsulinism in short-chain 3-hydroxyacyl-CoA dehydrogenase deficiency involves activation of glutamate dehydrogenase
 JBC Papers in Press. Published on July 29, 21 as Manuscript M11.123638 The latest version is at http://www.jbc.org/cgi/doi/1.174/jbc.m11.123638 Mechanism of hyperinsulinism in short-chain 3-hydroxyacyl-CoA
JBC Papers in Press. Published on July 29, 21 as Manuscript M11.123638 The latest version is at http://www.jbc.org/cgi/doi/1.174/jbc.m11.123638 Mechanism of hyperinsulinism in short-chain 3-hydroxyacyl-CoA
CARBOHYDRATE METABOLISM Disorders
 CARBOHYDRATE METABOLISM Disorders molecular formula C12H22O11 Major index which describes metabolism of carbohydrates, is a sugar level in blood. In healthy people it is 4,4-6,6 mmol/l (70-110 mg/dl)
CARBOHYDRATE METABOLISM Disorders molecular formula C12H22O11 Major index which describes metabolism of carbohydrates, is a sugar level in blood. In healthy people it is 4,4-6,6 mmol/l (70-110 mg/dl)
Journal Club WS 2012/13 Stefanie Nickl
 Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
BIOL212- Biochemistry of Disease. Metabolic Disorders: Diabetes
 BIOL212- Biochemistry of Disease Metabolic Disorders: Diabetes Diabetes mellitus is, after heart disease and cancer, the third leading cause of death in the west. Insulin is either not secreted in sufficient
BIOL212- Biochemistry of Disease Metabolic Disorders: Diabetes Diabetes mellitus is, after heart disease and cancer, the third leading cause of death in the west. Insulin is either not secreted in sufficient
Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 )
 770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
Functional characterization of rare variants of SCHAD, a protein involved in unregulated insulin secretion Henrikke Nilsen Hovland
 Functional characterization of rare variants of SCHAD, a protein involved in unregulated insulin secretion Henrikke Nilsen Hovland This thesis is submitted in partial fulfilment of the requirements for
Functional characterization of rare variants of SCHAD, a protein involved in unregulated insulin secretion Henrikke Nilsen Hovland This thesis is submitted in partial fulfilment of the requirements for
28 Regulation of Fasting and Post-
 28 Regulation of Fasting and Post- Prandial Glucose Metabolism Keywords: Type 2 Diabetes, endogenous glucose production, splanchnic glucose uptake, gluconeo-genesis, glycogenolysis, glucose effectiveness.
28 Regulation of Fasting and Post- Prandial Glucose Metabolism Keywords: Type 2 Diabetes, endogenous glucose production, splanchnic glucose uptake, gluconeo-genesis, glycogenolysis, glucose effectiveness.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Figure S1 (A) Comparison of body weight and blood glucose levels in male wild-type (WT) and Cre littermates. Mice were maintained on a normal chow
 Figure S1 (A) Comparison of body weight and blood glucose levels in male wild-type (WT) and Cre littermates. Mice were maintained on a normal chow diet (n = 7 per genotype). Body weight and fed blood glucose
Figure S1 (A) Comparison of body weight and blood glucose levels in male wild-type (WT) and Cre littermates. Mice were maintained on a normal chow diet (n = 7 per genotype). Body weight and fed blood glucose
β-cell Preservation and Regeneration After Islet Transplantation
 β-cell Preservation and Regeneration After Islet Transplantation Jyuhn-Huarng Juang, MD Division of Endocrinology and Metabolism, Department of Internal Medicine, Chang Gung University and Memorial Hospital,
β-cell Preservation and Regeneration After Islet Transplantation Jyuhn-Huarng Juang, MD Division of Endocrinology and Metabolism, Department of Internal Medicine, Chang Gung University and Memorial Hospital,
28/04/51. Introduction. Insulin signaling effects on memory and mood. Is accelerated brain aging a consequence of diabetes? chronic hyperglycemia
 Introduction Insulin signaling effects on memory and mood (Review) Diabetes mellitus is a chronic disease resulting from defects in insulin secretion, insulin action, or both Long-term diabetes Lawrence
Introduction Insulin signaling effects on memory and mood (Review) Diabetes mellitus is a chronic disease resulting from defects in insulin secretion, insulin action, or both Long-term diabetes Lawrence
Supplementary Figure 1
 Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
Modulating the Incretin System: A New Therapeutic Strategy for Type 2 Diabetes
 Modulating the Incretin System: A New Therapeutic Strategy for Type 2 Diabetes Geneva Clark Briggs, PharmD, BCPS Adjunct Professor at University of Appalachia College of Pharmacy Clinical Associate, Medical
Modulating the Incretin System: A New Therapeutic Strategy for Type 2 Diabetes Geneva Clark Briggs, PharmD, BCPS Adjunct Professor at University of Appalachia College of Pharmacy Clinical Associate, Medical
Complete Diabetes Mellitus Panel, Brochure
 Complete Diabetes Mellitus Panel, Brochure Interest in any of the products, request or order them at Bio-Connect Diagnostics. Bio-Connect Diagnostics B.V. T NL +31 (0)26 326 44 60 T BE +32 (0)2 502 12
Complete Diabetes Mellitus Panel, Brochure Interest in any of the products, request or order them at Bio-Connect Diagnostics. Bio-Connect Diagnostics B.V. T NL +31 (0)26 326 44 60 T BE +32 (0)2 502 12
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study
 Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
Supplementary Table 1. Antibodies and dilutions used in the immunohistochemical study Antigen Species Clone Source Dilution Insulin Guinea pig - Dako, Carpinteria, 1:200 Glucagon Rabbit - Dako, Carpinteria,
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Neonatal Diabetes. Objectives. Conflicts of Interest Disclosure. No conflicts of interest related to the content of this presentation
 Neonatal Diabetes Shannon Abernethy BSN, RN, CPN Pediatric Nurse Navigator Bon Secours Virginia Medical Group Pediatric Endocrinology and Diabetes Associates 1 Objectives Identify and define neonatal diabetes
Neonatal Diabetes Shannon Abernethy BSN, RN, CPN Pediatric Nurse Navigator Bon Secours Virginia Medical Group Pediatric Endocrinology and Diabetes Associates 1 Objectives Identify and define neonatal diabetes
Hypoglycemia in congenital hyperinsulinism
 How a normal body works: Our body is constantly at work. Our cells need a source of energy, and this source of energy is called glucose. The process is quite simple; think of it like an assembly line.
How a normal body works: Our body is constantly at work. Our cells need a source of energy, and this source of energy is called glucose. The process is quite simple; think of it like an assembly line.
PANCREATIC BETA CELLS PRODUCE AND SECRETE
 15 March, 2018 PANCREATIC BETA CELLS PRODUCE AND SECRETE Document Filetype: PDF 374.06 KB 0 PANCREATIC BETA CELLS PRODUCE AND SECRETE Among the oldest and cheapest drugs for diabetes are the drugs that
15 March, 2018 PANCREATIC BETA CELLS PRODUCE AND SECRETE Document Filetype: PDF 374.06 KB 0 PANCREATIC BETA CELLS PRODUCE AND SECRETE Among the oldest and cheapest drugs for diabetes are the drugs that
The Surgeon General s Call to Action and the
 Case of the Month Pamela Heaberlin, MS, RN, NNP-BC Section Editor 2.3 HOURS Continuing Education Congenital Hyperinsulinism Exclusive Human Milk and Breastfeeding Taryn M. Edwards, MSN, CRNP, NNP-BC ;
Case of the Month Pamela Heaberlin, MS, RN, NNP-BC Section Editor 2.3 HOURS Continuing Education Congenital Hyperinsulinism Exclusive Human Milk and Breastfeeding Taryn M. Edwards, MSN, CRNP, NNP-BC ;
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Distribution of Pancreatic Polypeptide in the Head of the Human Pancreas. By Andrew Kringas
 Distribution of Pancreatic Polypeptide in the Head of the Human Pancreas By Andrew Kringas Diabetes (Diabetes mellitus) A condition when the body doesn t properly respond to insulin Type 1 Type 2 Gestational
Distribution of Pancreatic Polypeptide in the Head of the Human Pancreas By Andrew Kringas Diabetes (Diabetes mellitus) A condition when the body doesn t properly respond to insulin Type 1 Type 2 Gestational
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the
 Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
Exenatide Treatment for 6 Months Improves Insulin Sensitivity in Adults With Type 1 Diabetes
 666 Diabetes Care Volume 37, March 2014 CLIN CARE/EDUCATION/NUTRITION/PSYCHOSOCIAL Exenatide Treatment for 6 Months Improves Insulin Sensitivity in Adults With Type 1 Diabetes Gayatri Sarkar, 1 May Alattar,
666 Diabetes Care Volume 37, March 2014 CLIN CARE/EDUCATION/NUTRITION/PSYCHOSOCIAL Exenatide Treatment for 6 Months Improves Insulin Sensitivity in Adults With Type 1 Diabetes Gayatri Sarkar, 1 May Alattar,
Janice Lazear, DNP, FNP-C, CDE DIAGNOSIS AND CLASSIFICATION OF DIABETES
 Janice Lazear, DNP, FNP-C, CDE DIAGNOSIS AND CLASSIFICATION OF DIABETES Objectives u At conclusion of the lecture the participant will be able to: 1. Differentiate between the classifications of diabetes
Janice Lazear, DNP, FNP-C, CDE DIAGNOSIS AND CLASSIFICATION OF DIABETES Objectives u At conclusion of the lecture the participant will be able to: 1. Differentiate between the classifications of diabetes
PANDER KO mice on high-fat diet are glucose intolerant yet resistant to fasting hyperglycemia and hyperinsulinemia
 FEBS Letters 585 (2011) 1345 1349 journal homepage: www.febsletters.org PANDER KO mice on high-fat diet are glucose intolerant yet resistant to fasting hyperglycemia and hyperinsulinemia Claudia E. Robert-Cooperman
FEBS Letters 585 (2011) 1345 1349 journal homepage: www.febsletters.org PANDER KO mice on high-fat diet are glucose intolerant yet resistant to fasting hyperglycemia and hyperinsulinemia Claudia E. Robert-Cooperman
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Expanded View Figures
 Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
23.1 Lipid Metabolism in Animals. Chapter 23. Micelles Lipid Metabolism in. Animals. Overview of Digestion Lipid Metabolism in
 Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Managing Congenital Hyperinsulinism in the Neonatal Period Vall d Hebron Hospital s Approach and Experience
 Managing Congenital Hyperinsulinism in the Alejandro Vargas Pieck Miquel Gussinyer Canadell María Clemente León Diego Yeste Fernández Universidad Autónoma de Barcelona Antonio Carrascosa Lezcano The first
Managing Congenital Hyperinsulinism in the Alejandro Vargas Pieck Miquel Gussinyer Canadell María Clemente León Diego Yeste Fernández Universidad Autónoma de Barcelona Antonio Carrascosa Lezcano The first
Multi-Parameter Apoptosis Assay Kit
 Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Multi-Parameter Apoptosis Assay Kit Catalog Number KA1335 5 x 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
ISLET STUDIES. Linda M. Nguyen, 1 Marina Pozzoli, 1 Thomas H. Hraha, 1 and Richard K.P. Benninger 1,2. Diabetes Volume 63, May
 Diabetes Volume 63, May 2014 1685 Linda M. Nguyen, 1 Marina Pozzoli, 1 Thomas H. Hraha, 1 and Richard K.P. Benninger 1,2 Decreasing Cx36 Gap Junction Coupling Compensates for Overactive K ATP Channels
Diabetes Volume 63, May 2014 1685 Linda M. Nguyen, 1 Marina Pozzoli, 1 Thomas H. Hraha, 1 and Richard K.P. Benninger 1,2 Decreasing Cx36 Gap Junction Coupling Compensates for Overactive K ATP Channels
THE IMPORTANCE OF GENETICS WHEN TREATING HYPERINSULINISM
 THE IMPORTANCE OF GENETICS WHEN TREATING HYPERINSULINISM HEATHER MCKNIGHT-MENCI, MSN, CRNP CHILDREN S HOSPITAL OF PHILADELPHIA WHAT IS HYPERINSULINISM? The leading cause of hypoglycemia in infants and
THE IMPORTANCE OF GENETICS WHEN TREATING HYPERINSULINISM HEATHER MCKNIGHT-MENCI, MSN, CRNP CHILDREN S HOSPITAL OF PHILADELPHIA WHAT IS HYPERINSULINISM? The leading cause of hypoglycemia in infants and
Supplementary Information. Induction of human pancreatic beta cell replication by inhibitors of dual specificity tyrosine regulated kinase
 Journal: Nature Medicine Supplementary Information Induction of human pancreatic beta cell replication by inhibitors of dual specificity tyrosine regulated kinase 1,2 Peng Wang PhD, 1,2 Juan-Carlos Alvarez-Perez
Journal: Nature Medicine Supplementary Information Induction of human pancreatic beta cell replication by inhibitors of dual specificity tyrosine regulated kinase 1,2 Peng Wang PhD, 1,2 Juan-Carlos Alvarez-Perez
Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro.
 Manuscript EMBO-2015-91058 Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. Holger A Russ, Audrey V Parent, Jennifer J Ringler, Thomas G Hennings, Gopika
Manuscript EMBO-2015-91058 Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. Holger A Russ, Audrey V Parent, Jennifer J Ringler, Thomas G Hennings, Gopika
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used in qpcr
 Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Diabetologia 9 Springer-Vcrlag 1993
 Diabetologia (1993) 36:305-309 Diabetologia 9 Springer-Vcrlag 1993 Preferential alteration of oxidative relative to total glycolysis in pancreatic islets of two rat models of inherited or acquired Type
Diabetologia (1993) 36:305-309 Diabetologia 9 Springer-Vcrlag 1993 Preferential alteration of oxidative relative to total glycolysis in pancreatic islets of two rat models of inherited or acquired Type
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Supplementary Figure 1.
 Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Gamma-aminobutyric acid (GABA) treatment blocks inflammatory pathways and promotes survival and proliferation of pancreatic beta cells
 Gamma-aminobutyric acid (GABA) treatment blocks inflammatory pathways and promotes survival and proliferation of pancreatic beta cells Gérald J. Prud homme, MD, FRCPC Keenan Research Centre for Biomedical
Gamma-aminobutyric acid (GABA) treatment blocks inflammatory pathways and promotes survival and proliferation of pancreatic beta cells Gérald J. Prud homme, MD, FRCPC Keenan Research Centre for Biomedical
Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells.
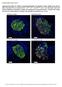 Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells. Pancreata from 16-weeks-old 6J +/+, 6J db/db, KS +/+ and KS db/db mice were harvested, fixed with
Supplementary Figure S1. Distinct compartmentalization of proinsulin in obese db/db mouse islet ß- cells. Pancreata from 16-weeks-old 6J +/+, 6J db/db, KS +/+ and KS db/db mice were harvested, fixed with
islets scored 1 week month months
 Supplementary Table 1. Sampling parameters for the morphometrical analyses Time (post- DT) Control mice (age-matched) α-cells mice pancreatic surface (mm 2 ) scored DT-treated mice islets scored mice pancreatic
Supplementary Table 1. Sampling parameters for the morphometrical analyses Time (post- DT) Control mice (age-matched) α-cells mice pancreatic surface (mm 2 ) scored DT-treated mice islets scored mice pancreatic
associated with serious complications, but reduce occurrences with preventive measures
 Wk 9. Management of Clients with Diabetes Mellitus 1. Diabetes Mellitus body s inability to metabolize carbohydrates, fats, proteins hyperglycemia associated with serious complications, but reduce occurrences
Wk 9. Management of Clients with Diabetes Mellitus 1. Diabetes Mellitus body s inability to metabolize carbohydrates, fats, proteins hyperglycemia associated with serious complications, but reduce occurrences
