p53 Controls Radiation-Induced Gastrointestinal Syndrome in Mice Independent of Apoptosis
|
|
|
- Gerard Holmes
- 5 years ago
- Views:
Transcription
1 p53 Controls Radiation-Induced Gastrointestinal Syndrome in Mice Independent of Apoptosis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Kirsch, D. G., P. M. Santiago, E. di Tomaso, J. M. Sullivan, W.-S. Hou, T. Dayton, L. B. Jeffords, et al. p53 Controls Radiation- Induced Gastrointestinal Syndrome in Mice Independent of Apoptosis. Science 327, no (January 28, 2010): American Association for the Advancement of Science (AAAS) Version Author's final manuscript Accessed Sun Jul 15 08:31:05 EDT 2018 Citable Link Terms of Use Detailed Terms Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use.
2 NIH Public Access Author Manuscript Published in final edited form as: Science January 29; 327(5965): doi: /science p53 Controls Radiation-Induced Gastrointestinal Syndrome in Mice Independent of Apoptosis David G. Kirsch 1,2,3, Philip M. Santiago 1, Emmanuelle di Tomasso 2, Julie M. Sullivan 3, Wu- Shiun Hou 1, Talya Dayton 1, Laura B. Jeffords 3, Pooja Sodha 1, Kim Mercer 1, Rhianna Cohen 1, Osamu Takeuchi 4, Stanley J. Korsmeyer 4,*, Roderick Bronson 5, Carla F. Kim 1, Kevin M. Haigis 1, Rakesh K. Jain 2, and Tyler Jacks 1,6 1 David H. Koch Institute for Integrative Cancer Research, Massachusetts Institute of Technology, Cambridge, MA USA 2 Department of Radiation Oncology, Massachusetts General Hospital and Harvard Medical School, Boston, MA USA 3 Departments of Radiation Oncology and Pharmacology & Cancer Biology, Duke University Medical Center, Durham NC USA 4 Dana Farber Cancer Institute, Boston MA USA 5 Tufts University School of Medicine and Veterinary Medicine, North Grafton, MA USA 6 Howard Hughes Medical Institute, Chevy Chase, MD USA Abstract Acute exposure to ionizing radiation can cause lethal damage to the gastrointestinal (GI) tract, a condition called the GI syndrome. Whether the target cells mediating the GI syndrome are derived from the epithelium or endothelium, and whether the target cells die by apoptosis or other mechanisms, are controversial issues. Studying mouse models, we found that selective deletion of the pro-apoptotic genes Bak1 and Bax from the GI epithelium or from endothelial cells did not protect mice from developing the GI syndrome after subtotal body gamma irradiation. In contrast, selective deletion of p53 from the GI epithelium, but not endothelial cells, sensitized irradiated mice to the GI syndrome. Transgenic mice overexpressing p53 in all tissues were protected from the GI syndrome after irradiation. These results suggest that the GI syndrome is caused by death of GI epithelial cells by a mechanism that is regulated by p53 but independent of apoptosis. Concern over the potential exposure of civilians to radiation has increased interest in understanding the mechanisms underlying radiation injury. Acute Radiation Syndrome (also called radiation sickness ) occurs when the body is exposed to a high dose of penetrating radiation in a short period of time. The constellation of symptoms that define Acute Radiation Syndrome include effects related to bone marrow toxicity (hematopoietic syndrome) and gastrointestinal toxicity (GI syndrome). The hematopoietic syndrome can occur after wholebody irradiation (WBI) at a dose of 6 Gy. The resultant reduction in lymphocytes and platelets leads to infection or hemorrhage, respectively, and death can occur within 30 days of exposure To whom correspondence should be addressed. tjacks@mit.edu. * deceased DGK: Departments of Radiation Oncology and Pharmacology & Cancer Biology, Duke University Medical Center, Durham NC USA W-SH: Genomics Research Center, Academia Sinica, Taipei 11529, Taiwan CFK: Department of Genetics, Harvard Medical School and Stem Cell Program, Children s Hospital, Boston MA USA KMH: Massachusetts General Hospital Cancer Center and Harvard Medical School, Charlestown, MA USA
3 Kirsch et al. Page 2 (1). In certain situations, death from the hematopoietic syndrome can be prevented (e.g., by shielding part of the bone marrow or by a subsequent bone marrow transplant) (2-4). At higher doses of radiation, death occurs within 10 days from damage to the GI tract (3,4). In contrast to the hematopoietic syndrome, no medical countermeasures are approved to prevent or treat the GI syndrome. Despite decades of study, the critical cellular targets of radiation-induced toxicity in the GI tract and the molecular mechanism of cell death underlying the GI syndrome remain controversial (5). For example, the role of p53 in the GI syndrome is unclear. Radiation activates p53 in the GI epithelium (6), which induces PUMA to kill cells via the intrinsic pathway of apoptosis in 4 hours (7-9). Deletion of either p53 or PUMA blocks radiationinduced apoptosis of most intestinal epithelial cells. Because death after WBI is delayed in mice lacking PUMA, p53-mediated apoptosis has been implicated in regulating the GI syndrome (9). Other research groups have reported that the absence of p53 does not alter the GI syndrome and proposed that it is instead regulated by radiation-induced apoptosis of endothelial cells (10). Although radiation-induced endothelial cell apoptosis can proceed in the absence of p53 or PUMA (9,11), the intrinsic pathway is still believed to be involved because Bax / and Bak1 / mice have reduced endothelial cell apoptosis following irradiation (12,13). Others have implicated p53 in protecting mice from the GI syndrome (14). Although radiationinduced apoptosis is suppressed in p53 null mice (6), other modes of cell death, such as mitotic catastrophe, can still occur (15). For example, 24 hours after radiation at doses of 8 Gy or more, reproductive or clonogenic cell death (hereafter referred to as mitotic death ), occurs in the small intestine (16). The molecular mechanism of this form of death remains unclear, but at this dose of radiation, a cell cycle arrest is followed by proliferation where some cells may undergo an aberrant mitosis and die by mitotic catastrophe (15,17,18). To investigate the cellular target and the molecular mechanism of cell death in the acute radiation syndrome, we have utilized the Cre-loxP system (19). Mice in which Cre is expressed under the control of a tissue-specific promoter can selectively delete DNA flanked by loxp sites, which is termed a floxed (FL) allele, in a cell type-specific manner. Therefore, we utilized mice (VillinCre) that express Cre specifically throughout the GI epithelium under the control of the villin promoter (20) and mice (Tie2Cre) in which Cre is expressed specifically in endothelial cells and the hematopoietic system under the control of the Tie2 promoter (21). To investigate the role of the intrinsic pathway of apoptosis in the acute radiation syndrome, VillinCre or Tie2Cre mice were crossed with mice carrying floxed alleles for Bax (22). In some settings, BAK can mediate BAX-independent apoptosis via the intrinsic pathway (23). Therefore, experiments were carried out on a Bak1 null genetic background (24). To determine if Bax was deleted in the hematopoietic system in Tie2Cre; Bak1 / ; Bax FL/ mice, we subjected cell lysates from the spleen, thymus, and lymph nodes to Western blot analysis. In these tissues, the levels of BAX were significantly reduced when compared to Tie2Cre; Bak1 / ; Bax FL/+ littermate controls (Fig. 1A). Deleting the intrinsic pathway of apoptosis from the hematopoietic system in Tie2Cre; Bak1 / ; Bax FL/ mice protected the bone marrow (fig. S1) and improved survival of the mice after exposure to 12.5 Gy WBI (Fig. 1B). Therefore, the intrinsic pathway of apoptosis is required for death from the hematopoietic syndrome. Because Cre is also expressed in endothelial cells in Tie2Cre mice, it is possible that protection of the vascular niche (25) within the bone marrow contributed to increased survival from WBI. We next examined the role of the intrinsic pathway of apoptosis in the GI syndrome. For these experiments, the bone marrow of the front legs was shielded for subtotal-body irradiation (SBI)
4 Kirsch et al. Page 3 to avoid the hematopoietic syndrome (fig. S2). Initial experiments were performed with mice in which either Bak1 or Bax was deleted. No difference in survival from the GI syndrome was observed for Bak1 / mice vs. Bak1 +/+ littermate controls or between Bax / mice vs. Bax +/+ littermate controls after SBI (fig. S3). Because Bak1 / ; Bax / mice are generally not viable (23), we studied mice in which Bak1 and Bax were deleted from either endothelial cells or GI epithelial cells. To determine whether Bax was deleted from endothelial cells in Tie2Cre; Bak1 / ; Bax FL/ mice, endothelial cells were isolated from lungs by fluorescent activated cell sorting (FACS). PECAM+, SCA-1+ cells were collected (24) and analyzed by Western blot for BAX (Fig. 2A). In these cells from Tie2Cre; Bak1 / ; Bax FL/ mice, BAX was not detected. Within the villi, apoptosis of mesenchymal cells, which includes endothelial cells and lymphocytes, occurs 4 hours after doses of radiation that cause the GI syndrome (10,26). Therefore, we harvested the small intestine of mice 4 hours after Gy SBI and assessed mesenchymal cell apoptosis within the villi by TUNEL staining. Deleting the intrinsic pathway of apoptosis in Tie2Cre; Bak1 / ; Bax FL/ mice significantly reduced mesenchymal cell apoptosis compared to Tie2Cre; Bak / ; Bax FL/+ mice that retained a wild-type Bax allele (Fig. 2 B and C), but did not improve survival from the GI syndrome after SBI (Fig. 2D and fig. S4). These results suggest that mesenchymal cell apoptosis within the intervillus region of the intestine does not initiate the GI syndrome. We also evaluated the role of the intrinsic pathway of apoptosis in mediating the GI syndrome in GI epithelial cells. To determine whether BAX was deleted in the GI epithelium of VillinCre; Bak1 / ; Bax FL/ mice, we isolated epithelial cells from the small intestine and analyzed them for BAX expression by Western blot (24). In VillinCre; Bak1 / ; Bax FL/ mice, BAX was deleted from the GI epithelium (Fig. 3A). Although deleting the intrinsic pathway of apoptosis in GI epithelium reduced apoptosis in the crypt epithelium 5-fold after irradiation (Fig. 3 B and C), this did not improve survival after SBI (Fig. 3D and fig. S5). Taken together, these results suggest that survival from the GI syndrome is not determined by apoptosis of either endothelial cells or GI epithelial cells. At radiation doses that can cause the GI syndrome, mitotic death occurs within the small intestine (16). Although the molecular mechanism of this type of cell death is not completely understood, it may involve progression through an aberrant mitosis with damaged DNA (27). Therefore, we looked for evidence of aberrant mitosis in the crypts of the small intestine after irradiation. In unirradiated mice, almost all cells undergoing mitosis showed chromosomes that aligned symmetrically (Fig 4A and fig. S6). 24 hours after 13.4 Gy SBI, most of the mitotic cells in the crypts were aberrant because chromosomes aligned in an asymmetrical fashion with lagging chromosomes or in micronuclei (Fig 4A and fig. S6). Because p53 is required in the small intestine for optimal cell cycle arrest after radiation to prevent inappropriate entry into mitosis (18), we generated mice in which p53 was deleted in the GI epithelium (VillinCre; p53 FL/ ) to investigate whether cell cycle checkpoints in GI epithelial cells regulate the GI syndrome. Indeed, the VillinCre; p53 FL/ mice were more sensitive to SBI than VillinCre; p53 FL/+ littermates (Fig. 4B), even though we did not observe a difference in surviving crypts at 90 hours after SBI (fig. S7). These results point to cells within the GI crypt epithelium as the primary cellular target of radiation mediating the GI syndrome. Moreover, as reported by others (14), we also found mice lacking the cyclin-dependent kinase inhibitor p21 to be sensitive to the GI syndrome (fig. S8). To investigate whether deletion of the intrinsic pathway of apoptosis could suppress sensitivity of the GI syndrome that results from loss of p53, we generated mice (VillinCre; p53 FL/ ; Bak1 / ; Bax FL/ ) in which p53 and the intrinsic pathway of apoptosis were deleted from GI epithelial cells (24). Despite deletion of Bak1 and Bax, mice remained sensitive to the GI syndrome when p53 was deleted in GI epithelial cells (Fig 4C).
5 Kirsch et al. Page 4 These results are consistent with a model where progression through p53-regulated cell cycle checkpoints after radiation leads to reproductive or mitotic death of GI epithelial cells independent of apoptosis to cause the GI syndrome (15,16,18). To test this model in a gain-offunction experiment, we studied super p53 tg b mice, which contain two extra copies of the wild-type p53 gene (28). Following irradiation, super p53 mice exhibit an enhanced DNA damage response with elevated levels of p53-dependent apoptosis and increased induction of the cyclin-dependent kinase inhibitor, p21 (28). Consistent with a model where p53-dependent cell cycle arrest regulates the GI syndrome, we observed that p53 tg b mice are protected from the GI syndrome after SBI (Fig 4D). We also generated mice in which p53 was deleted from endothelial cells. These Tie2Cre; p53 FL/ mice were not more sensitive to the acute GI syndrome after SBI (fig. S9). Surprisingly, all of these mice died from delayed radiation effects within 2 months (fig. S9). Therefore, p53 may not only protect the GI epithelium from acute injury (14), but p53 may also protect endothelial cells from late effects from radiation. These results have potential implications for efforts to develop medical countermeasures against radiation. Drugs that block the intrinsic pathway of apoptosis may be effective for the hematopoietic syndrome, but not the GI syndrome. Indeed, the p53 inhibitor, pifithrin-α, rescues mice from the lethal effects of the hematopoietic syndrome, but does not protect mice from the GI syndrome (14). Because complete inhibition of p53 may promote the GI syndrome (Fig. 4B), drugs for the acute radiation syndrome that block apoptosis should not compromise p53 function (29). This study may also have important implications for cancer therapy. Inhibitors of p53 function have been proposed as a way to limit the acute toxicity of radiation therapy and increase tumor response without increasing the risk of radiation-induced cancers (30-32). For many patients, acute toxicity is not dose limiting in the clinic. Instead, the risk of late effects from radiation therapy, which can be secondary to vascular injury (33), frequently limits the radiation dose that can safely be delivered to treat a cancer. Our finding that deleting p53 from endothelial cells sensitizes mice to late effects of radiation (fig. S9) raises the possibility of increased late effects when p53 inhibitors are combined with radiation therapy. Supplementary Material Acknowledgments Refer to Web version on PubMed Central for supplementary material. 34. We thank M. Serrano for the super p53 mice; A.Berns for the p53 FL mice; P. Crawford and J. Gordon for advice; E. Travis for providing a model jig; J. Down, C. Pope, and K. Doppke for performing dosimetry measurements; and K. Held for critically reading the manuscript. This work is funded by the Howard Hughes Medical Institute (TJ), partially by Cancer Center Support (core) grant P30-CA14051 from the NCI (TJ), NCI grant KO8 CA (DGK), pilot project from the Dana Farber Cancer Center for Medical Countermeasures Against Radiation U19-AI06775 (DGK), RC1-AI (DGK), the American Society of Clinical Oncology Young Investigator Award (DGK), and NCI grant P01 CA80124 (RKJ). TJ is the David H. Koch Professor of Biology and a Daniel K. Ludwig Scholar. References and Notes 1. Mettler FA, Voelz GL. N. Engl. J. Med 2002;346:1554. [PubMed: ] 2. van Bekkum DW, Schotman E. Int. J. Radiat. Biol 1974;25: Terry NHA, Travis EL. Int. J. Radiat. Onc. Biol. Phys 1989;17: Mason KA, Withers HR, McBride WH, Davis CA, Smathers JB. Radiation Res 1989;117:480. [PubMed: ] 5. Brown JM. Int. J. Radiat. Onc. Bio. Phys 2008;70:799.
6 Kirsch et al. Page 5 6. Merritt AJ, et al. Cancer Res 1994;54:614. [PubMed: ] 7. Potten CS. Nature 1977;269:518. [PubMed: ] 8. Jeffers JR, et al. Cancer Cell 2003;4:321. [PubMed: ] 9. Qiu W, et al. Cell Stem Cell 2008;2:576. [PubMed: ] 10. Paris F, et al. Science 2001;293:293. [PubMed: ] 11. Santana P, et al. Cell 1996;86:189. [PubMed: ] 12. Garcia-Barros M, et al. Science 2003;300:1155. [PubMed: ] 13. Rotolo J, et al. Int. J. Radiat. Onc. Bio. Phys 2008;70: Komarova EA, et al. Oncogene 2004;23:3265. [PubMed: ] 15. Merritt AJ, Allen TD, Potten CS, Hickman JA. Oncogene 1997;14:2759. [PubMed: ] 16. Hendry JH, Potten CS. Int. J. Radiat. Biol 1982;42: Lesher J, Lesher S. Radiation Res 1974;57:148. [PubMed: ] 18. Potten CS, Owen G, Roberts SA. Int. J. Radiat. Biol 1990;57:185. [PubMed: ] 19. Branda CS, Dymecki SM. Dev. Cell 2004;6:7. [PubMed: ] 20. El Marjou F, et al. Genesis 2004;39:186. [PubMed: ] 21. Koni PA, et al. J. Exp. Med 2001;193:741. [PubMed: ] 22. Takeuchi O, et al. Proc. Natl. Acad. Sci. U.S.A 2005;102: [PubMed: ] 23. Lindsten T, et al. Mol. Cell 2000;6:1389. [PubMed: ] 24. Table describing genotypes used in this study, methods, and text are in Supporting Online Material 25. Salter AB, et al. Blood 2009;113:2104. [PubMed: ] 26. Crawford PA, Gordon JI. PNAS 2005;102: [PubMed: ] 27. Kroemer G, et al. Cell Death Differ 2005;12:1463. [PubMed: ] 28. Garcia-Cao I, et al. EMBO J 2002;21:6225. [PubMed: ] 29. Burdelya LG, et al. Science 2008;320:226. [PubMed: ] 30. Komarov PG, et al. Science 1999;285:1733. [PubMed: ] 31. Christophorou MA, Ringshausen I, Finch AJ, Swigart LB, Evan GI. Nature 2006;443:214. [PubMed: ] 32. Burdelya LG, et al. Cancer Res 2006;66:9356. [PubMed: ] 33. Coderre JA, et al. Radiation Res 2006;166:495. [PubMed: ]
7 Kirsch et al. Page 6 Fig. 1. Deletion of the intrinsic pathway of apoptosis in the hematopoietic system protects mice from death after whole-body gamma irradiation (WBI). A. Western blot for BAX and tubulin in spleen, lymph node, and thymus from Tie2Cre; Bak1 / ; Bax FL/ mice (FL/ ) demonstrates deletion of Bax in the hematopoietic system. Tie2Cre; Bak1 / ; Bax FL/+ mice (FL/+) retain Bax and serve as a control. B. Kaplan-Meier survival analysis of Tie2Cre; Bak1 / ; Bax FL/+ mice and Tie2Cre; Bak1 / ; Bax FL/ litter mates after 12.5 Gy WBI. By log-rank comparison, p =
8 Kirsch et al. Page 7 Fig. 2. Deletion of the intrinsic pathway of apoptosis from endothelial cells does not protect mice from subtotal-body gamma irradiation (SBI). A. Western blot for BAX and Vinculin from lung endothelial cells isolated from mice that contained no Cre ( ), VillinCre (Villin), or Tie2Cre (Tie2) and the indicated Bax genotype. B. Mesenchymal cell (endothelial cell and lymphocyte) apoptosis is decreased in Tie2Cre; Bak1 / ; Bax FL/ mice. TUNEL (brown) and MECA-32 (pink) immunohistochemistry identify apoptotic mesenchymal cells within the villi of Tie2Cre; Bak1 / ; Bax FL/+ mice. Scale bar, 100 μm. C. Quantification of mesenchymal cell apoptosis in the villi of the distal small intestine. The mean number of mesenchymal cells undergoing apoptosis in a total of at least 87 villi from 3 mice is shown; *p < 0.01 by two-tailed t test. Error bars indicate SEM. D. Kaplan-Meier survival analysis of Tie2Cre; Bak1 / ; Bax FL/+ mice and Tie2Cre; Bak1 / ; Bax FL/ litter mates after Gy SBI. By log-rank comparison, p =NS.
9 Kirsch et al. Page 8 Fig. 3. Deletion of the intrinsic pathway of apoptosis from GI epithelial cells does not protect mice from subtotal-body gamma irradiation (SBI). A. Western blot for BAX and Vinculin from GI epithelial cells isolated from mice without Cre ( ), with VillinCre (VL), or Tie2Cre (T2), and the indicated Bax genotype. B. Hematoxylin and eosin stained sections of distal small intestine 4 hours after Gy SBI demonstrates suppression of apoptosis within the crypt epithelium in VillinCre; Bak1 / ; Bax FL/ mice. A green asterisk labels cells with characteristic apoptotic morphology. Scale bar, 100 μm. C. Quantification of GI epithelial cell apoptosis in the crypts of the distal small intestine. The mean number of apoptotic cells in a total of at least 100 crypts in 3 mice is shown. Error bars indicate SEM. *p < 0.01, by two-tailed t test. Similar results were observed in the proximal small intestine. D. Kaplan-Meier survival analysis of VillinCre; Bak1 / ; Bax FL/+ and VillinCre; Bak1 / ; Bax FL/ mice after Gy SBI. By log-rank comparison, p = NS.
10 Kirsch et al. Page 9 Fig. 4. Regulation of the GI syndrome by p53 in epithelial cells is independent of apoptosis. A. Quantification of normal (NL) and aberrant (AB) mitoses in GI crypt epithelial cells of the proximal and distal small intestine from wild-type mice without ( ) or 24 hours after (+) 13.4 Gy SBI with ionizing radiation. The mean number of mitoses per crypt in a total of at least 115 crypts from 6 mice is shown. Error bars indicate SEM. *p < 0.04 for aberrant mitoses, twosided t test and **p < for normal mitoses, two-sided t test. B. Kaplan-Meier survival analysis of VillinCre; p53 FL/+ and VillinCre; p53 FL/ mice after Gy SBI with gamma irradiation. p = by log-rank test. C. Kaplan-Meier survival analysis of at least 9 mice of each of the indicated genotype after 15 Gy SBI with gamma irradiation. p = between p53 FL/ and p53 FL/+ mice and p = NS between Bax FL/ and Bax FL/+ mice by log-rank test. D. Kaplan-Meier survival analysis of super p53 tg b mice and C57/BL6 p53 +/+ littermate controls after 13.4 Gy SBI with ionizing radiation. p = 0.02 by log-rank test.
NIH Public Access Author Manuscript Int J Radiat Oncol Biol Phys. Author manuscript; available in PMC 2010 September 15.
 NIH Public Access Author Manuscript Published in final edited form as: Int J Radiat Oncol Biol Phys. 2010 March 15; 76(4): 973 977. doi:10.1016/j.ijrobp.2009.11.038. Imaging Primary Lung Cancers in Mice
NIH Public Access Author Manuscript Published in final edited form as: Int J Radiat Oncol Biol Phys. 2010 March 15; 76(4): 973 977. doi:10.1016/j.ijrobp.2009.11.038. Imaging Primary Lung Cancers in Mice
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice
 Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
Role of p53 in regulating tissue response to radiation by mechanisms independent of apoptosis
 Review Article Role of p53 in regulating tissue response to radiation by mechanisms independent of apoptosis Chang-Lung Lee 1, Jordan M. Blum 2, David G. Kirsch 1,2 1 Department of Radiation Oncology,
Review Article Role of p53 in regulating tissue response to radiation by mechanisms independent of apoptosis Chang-Lung Lee 1, Jordan M. Blum 2, David G. Kirsch 1,2 1 Department of Radiation Oncology,
BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information)
 BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information) David Dankort, David P. Curley, Robert A. Cartlidge, Betsy Nelson, Anthony N. Karnezis,
BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information) David Dankort, David P. Curley, Robert A. Cartlidge, Betsy Nelson, Anthony N. Karnezis,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2607 Figure S1 Elf5 loss promotes EMT in mammary epithelium while Elf5 overexpression inhibits TGFβ induced EMT. (a, c) Different confocal slices through the Z stack image. (b, d) 3D rendering
DOI: 10.1038/ncb2607 Figure S1 Elf5 loss promotes EMT in mammary epithelium while Elf5 overexpression inhibits TGFβ induced EMT. (a, c) Different confocal slices through the Z stack image. (b, d) 3D rendering
UNC-Duke Biology Course for Residents Fall Cell Cycle Effects of Radiation
 UNC-Duke Biology Course for Residents Fall 2018 1 Cell Cycle: Sequence of changes in a cell starting with the moment the cell is created by cell division, continuing through the doubling of the DNA and
UNC-Duke Biology Course for Residents Fall 2018 1 Cell Cycle: Sequence of changes in a cell starting with the moment the cell is created by cell division, continuing through the doubling of the DNA and
UNC-Duke Biology Course for Residents Fall
 UNC-Duke Biology Course for Residents Fall 2018 1 UNC-Duke Biology Course for Residents Fall 2018 2 UNC-Duke Biology Course for Residents Fall 2018 3 UNC-Duke Biology Course for Residents Fall 2018 4 UNC-Duke
UNC-Duke Biology Course for Residents Fall 2018 1 UNC-Duke Biology Course for Residents Fall 2018 2 UNC-Duke Biology Course for Residents Fall 2018 3 UNC-Duke Biology Course for Residents Fall 2018 4 UNC-Duke
Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al
 Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al Suppl. Fig. 1 Tissue DN C Proteins kd TSC1-17 TSC 1 loxp bp -48-285 ctin PEMs Neutrophils
Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al Suppl. Fig. 1 Tissue DN C Proteins kd TSC1-17 TSC 1 loxp bp -48-285 ctin PEMs Neutrophils
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Early cell death (FGF) B No RunX transcription factor produced Yes No differentiation
 Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Radiation Oncology. Initial Certification Qualifying (Computer-based) Examination: Study Guide for Radiation and Cancer Biology
 Radiation Oncology Initial Certification Qualifying (Computer-based) Examination: Study Guide for Radiation and Cancer Biology This exam tests your knowledge of the principles of cancer and radiation biology
Radiation Oncology Initial Certification Qualifying (Computer-based) Examination: Study Guide for Radiation and Cancer Biology This exam tests your knowledge of the principles of cancer and radiation biology
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Image-Guided Radiotherapy Targets Macromolecules. through Altering the Tumor Microenvironment
 Supporting Information Image-Guided Radiotherapy Targets Macromolecules through ltering the Tumor Microenvironment uthors: Oliver K. ppelbe,, Qingbei Zhang,, harles. Pelizzari, Ralph R. Weichselbaum,,
Supporting Information Image-Guided Radiotherapy Targets Macromolecules through ltering the Tumor Microenvironment uthors: Oliver K. ppelbe,, Qingbei Zhang,, harles. Pelizzari, Ralph R. Weichselbaum,,
Cancer. The fundamental defect is. unregulated cell division. Properties of Cancerous Cells. Causes of Cancer. Altered growth and proliferation
 Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Bone Marrow Pop. (% Total) Mature Pool (Absolute %) Immature Pool (Absolute %) A10 EC Control A10 EC Control A10 EC Control
 Bone Marrow Pop. (% Total) Mature Pool (Asolute %) Immature Pool (Asolute %) A10 EC A10 EC A10 EC Myeloid 50.7 57.5 37.5 46.2 13.2 11.3 Erythroid 38.3 23.2 33.3 16.8 9.3 6.3 Lymphocytes 13.8 19.0 - - -
Bone Marrow Pop. (% Total) Mature Pool (Asolute %) Immature Pool (Asolute %) A10 EC A10 EC A10 EC Myeloid 50.7 57.5 37.5 46.2 13.2 11.3 Erythroid 38.3 23.2 33.3 16.8 9.3 6.3 Lymphocytes 13.8 19.0 - - -
Supplementary Figure 1. Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Nature Immunology: doi: /ni.
 Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
Supplementary Figure 1 Efficiency of Mll4 deletion and its effect on T cell populations in the periphery. Expression of Mll4 floxed alleles (16-19) in naive CD4 + T cells isolated from lymph nodes and
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse
 Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Supplemental figure legends Supplemental Figure 1. Intracranial transduction of a modified ptomo lentiviral vector in the mouse hippocampus targets GFAP-positive but not NeuN-positive cells. (A) Stereotaxic
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
AP VP DLP H&E. p-akt DLP
 A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
A B AP VP DLP H&E AP AP VP DLP p-akt wild-type prostate PTEN-null prostate Supplementary Fig. 1. Targeted deletion of PTEN in prostate epithelium resulted in HG-PIN in all three lobes. (A) The anatomy
fl/+ KRas;Atg5 fl/+ KRas;Atg5 fl/fl KRas;Atg5 fl/fl KRas;Atg5 Supplementary Figure 1. Gene set enrichment analyses. (a) (b)
 KRas;At KRas;At KRas;At KRas;At a b Supplementary Figure 1. Gene set enrichment analyses. (a) GO gene sets (MSigDB v3. c5) enriched in KRas;Atg5 fl/+ as compared to KRas;Atg5 fl/fl tumors using gene set
KRas;At KRas;At KRas;At KRas;At a b Supplementary Figure 1. Gene set enrichment analyses. (a) GO gene sets (MSigDB v3. c5) enriched in KRas;Atg5 fl/+ as compared to KRas;Atg5 fl/fl tumors using gene set
Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic
 Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic B cells from WT Nfat2 +/+, TCL1 Nfat2 +/+ and TCL1 Nfat2
Supplementary Figure 1: Expression of NFAT proteins in Nfat2-deleted B cells (a+b) Protein expression of NFAT2 (a) and NFAT1 (b) in isolated splenic B cells from WT Nfat2 +/+, TCL1 Nfat2 +/+ and TCL1 Nfat2
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence
 Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Supplemental Table 1. Primers used for RT-PCR analysis of inflammatory cytokines Gene Primer Sequence IL-1α Forward primer 5 -CAAGATGGCCAAAGTTCGTGAC-3' Reverse primer 5 -GTCTCATGAAGTGAGCCATAGC-3 IL-1β
Inhibition of DYRK1A stimulates human beta-cell proliferation
 Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Atg5 flox/flox ; CAG-Cre, 19M brain heart lung. spleen stomach colon. Takamura_Fig. S1
 Takamura_Fig. S1 brain heart lung spleen stomach colon kidney SM Supplemental Figure 1 Histological findings of tg5 flox/flox ;CG-Cre mouse tissues. H&E staining of the brain, heart, lung, spleen, stomach,
Takamura_Fig. S1 brain heart lung spleen stomach colon kidney SM Supplemental Figure 1 Histological findings of tg5 flox/flox ;CG-Cre mouse tissues. H&E staining of the brain, heart, lung, spleen, stomach,
SOPten flox/flox (KO) Pten flox/flox (WT) flox allele 6.0 kb. Pten. Actin. ! allele 2.3 kb. Supplementary Figure S1. Yanagi, et al.
 s1 A Pten flox/flox () SOPten flox/flox () flox allele 6. kb B Pten flox/flox () SOPten flox/flox () Pten Actin! allele 2.3 kb Supplementary Figure S1. Yanagi, et al. A B BrdU BrdU positive cells ( ) 3
s1 A Pten flox/flox () SOPten flox/flox () flox allele 6. kb B Pten flox/flox () SOPten flox/flox () Pten Actin! allele 2.3 kb Supplementary Figure S1. Yanagi, et al. A B BrdU BrdU positive cells ( ) 3
Supplementary Fig. 1: ATM is phosphorylated in HER2 breast cancer cell lines. (A) ATM is phosphorylated in SKBR3 cells depending on ATM and HER2
 Supplementary Fig. 1: ATM is phosphorylated in HER2 breast cancer cell lines. (A) ATM is phosphorylated in SKBR3 cells depending on ATM and HER2 activity. Upper panel: Representative histograms for FACS
Supplementary Fig. 1: ATM is phosphorylated in HER2 breast cancer cell lines. (A) ATM is phosphorylated in SKBR3 cells depending on ATM and HER2 activity. Upper panel: Representative histograms for FACS
Genomic Instability Induced by Ionizing Radiation
 Genomic Instability Induced by Ionizing Radiation Christian Streffer Universitätsklinikum Essen, 45122 Essen, Germany INTRODUCTION In contrast to general assumptions it has frequently been shown that DNA
Genomic Instability Induced by Ionizing Radiation Christian Streffer Universitätsklinikum Essen, 45122 Essen, Germany INTRODUCTION In contrast to general assumptions it has frequently been shown that DNA
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
stability and tumor suppression
 Supplementary information The stress kinase MKK7 couples oncogenic stress to p53 stability and tumor suppression Daniel Schramek 1, Athanassios Kotsinas 2, Arabella Meixner 1, Teiji Wada 1, Ulrich Elling
Supplementary information The stress kinase MKK7 couples oncogenic stress to p53 stability and tumor suppression Daniel Schramek 1, Athanassios Kotsinas 2, Arabella Meixner 1, Teiji Wada 1, Ulrich Elling
The Role of Rac Signaling in The Perivascular Niche
 The Role of Rac Signaling in The Perivascular Niche Felicia Ciuculescu Diaspora and Higher Education and Research Perspectives in Personalized Medicine- from Concept to Clinical Application Center for
The Role of Rac Signaling in The Perivascular Niche Felicia Ciuculescu Diaspora and Higher Education and Research Perspectives in Personalized Medicine- from Concept to Clinical Application Center for
Cancer. The fundamental defect is. unregulated cell division. Properties of Cancerous Cells. Causes of Cancer. Altered growth and proliferation
 Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Differential effects of oncogenic K-Ras and N-Ras on proliferation, differentiation, and tumor progression in the colon
 SUPPLEMENTARY INFORMATION Differential effects of oncogenic K-Ras and N-Ras on proliferation, differentiation, and tumor progression in the colon Kevin M. Haigis, Krystle Kendall, Yufang Wang, Ann Cheung,
SUPPLEMENTARY INFORMATION Differential effects of oncogenic K-Ras and N-Ras on proliferation, differentiation, and tumor progression in the colon Kevin M. Haigis, Krystle Kendall, Yufang Wang, Ann Cheung,
Supplemental Data Tamoxifen administration to Vil-Scap- mice.
 Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
Supplemental Data FIGURE S1. Tamoxifen administration to Vil-Scap - mice. In the experiments shown in Fig. 1 to Fig. 5, tamoxifen (2 mg per dose) was dissolved in corn oil and administered by orogastric
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
SUPPLEMENTARY INFORMATION
 a. Smo+/+ b. Smo+/+ 5.63 5.48 c. Lin- d. e. 6 5 4 3 Ter119 Mac B T Sca1 Smo+/+ 25 15 2 o BMT 2 1 5 * Supplementary Figure 1: Deletion of Smoothened does not alter the frequency of hematopoietic lineages
a. Smo+/+ b. Smo+/+ 5.63 5.48 c. Lin- d. e. 6 5 4 3 Ter119 Mac B T Sca1 Smo+/+ 25 15 2 o BMT 2 1 5 * Supplementary Figure 1: Deletion of Smoothened does not alter the frequency of hematopoietic lineages
An extra copy of p53 suppresses development of spontaneous Kras-driven but not radiationinduced
 An extra copy of p53 suppresses development of spontaneous Kras-driven but not radiationinduced cancer Everett J. Moding,, Chang-Lung Lee, David G. Kirsch JCI Insight. 2016;1(10):e86698. https://doi.org/10.1172/jci.insight.86698.
An extra copy of p53 suppresses development of spontaneous Kras-driven but not radiationinduced cancer Everett J. Moding,, Chang-Lung Lee, David G. Kirsch JCI Insight. 2016;1(10):e86698. https://doi.org/10.1172/jci.insight.86698.
293T cells were transfected with indicated expression vectors and the whole-cell extracts were subjected
 SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Formation of a complex between Slo1 and CRL4A CRBN E3 ligase. (a) HEK 293T cells were transfected with indicated expression vectors and the whole-cell
Supplementary Figure 1. Successful excision of genes from WBM lysates and
 Supplementary Information: Supplementary Figure 1. Successful excision of genes from WBM lysates and survival of mice with different genotypes. (a) The proper excision of Pten, p110α, p110α and p110δ was
Supplementary Information: Supplementary Figure 1. Successful excision of genes from WBM lysates and survival of mice with different genotypes. (a) The proper excision of Pten, p110α, p110α and p110δ was
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Chronic variable stress activates hematopoietic stem cells
 SUPPLEMENTARY INFORMATION Chronic variable stress activates hematopoietic stem cells Timo Heidt *, Hendrik B. Sager *, Gabriel Courties, Partha Dutta, Yoshiko Iwamoto, Alex Zaltsman, Constantin von zur
SUPPLEMENTARY INFORMATION Chronic variable stress activates hematopoietic stem cells Timo Heidt *, Hendrik B. Sager *, Gabriel Courties, Partha Dutta, Yoshiko Iwamoto, Alex Zaltsman, Constantin von zur
A20 (TNFAIP3) deficiency in myeloid cells triggers erosive polyarthritis resembling rheumatoid arthritis
 A20 (TNFAIP3) deficiency in myeloid cells triggers erosive polyarthritis resembling rheumatoid arthritis Mourad Matmati 1,2 *, Peggy Jacques 3 *, Jonathan Maelfait 1,2, Eveline Verheugen 3, Mirjam Kool
A20 (TNFAIP3) deficiency in myeloid cells triggers erosive polyarthritis resembling rheumatoid arthritis Mourad Matmati 1,2 *, Peggy Jacques 3 *, Jonathan Maelfait 1,2, Eveline Verheugen 3, Mirjam Kool
Cell Death & Renewal (part 2)
 17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
17 Cell Death & Renewal (part 2) Programmed Cell Death A major signaling pathway that promotes cell survival is initiated by the enzyme PI 3-kinase, which phosphorylates PIP2 to form PIP3, which activates
Supplementary Material
 Supplementary Material Summary: The supplementary information includes 1 table (Table S1) and 4 figures (Figure S1 to S4). Supplementary Figure Legends Figure S1 RTL-bearing nude mouse model. (A) Tumor
Supplementary Material Summary: The supplementary information includes 1 table (Table S1) and 4 figures (Figure S1 to S4). Supplementary Figure Legends Figure S1 RTL-bearing nude mouse model. (A) Tumor
Transcription factor Foxp3 and its protein partners form a complex regulatory network
 Supplementary figures Resource Paper Transcription factor Foxp3 and its protein partners form a complex regulatory network Dipayan Rudra 1, Paul deroos 1, Ashutosh Chaudhry 1, Rachel Niec 1, Aaron Arvey
Supplementary figures Resource Paper Transcription factor Foxp3 and its protein partners form a complex regulatory network Dipayan Rudra 1, Paul deroos 1, Ashutosh Chaudhry 1, Rachel Niec 1, Aaron Arvey
Radiation-induced induced Genomic Instability and Bystander Effects: implications for radiation leukaemogenesis
 Radiation-induced induced Genomic Instability and Bystander Effects: implications for radiation leukaemogenesis University of Dundee Medical School Eric G Wright Professor of Experimental Haematology The
Radiation-induced induced Genomic Instability and Bystander Effects: implications for radiation leukaemogenesis University of Dundee Medical School Eric G Wright Professor of Experimental Haematology The
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Amelio et al., http://www.jcb.org/cgi/content/full/jcb.201203134/dc1 Figure S1. mir-24 regulates proliferation and by itself induces
CELL CYCLE REGULATION AND CANCER. Cellular Reproduction II
 CELL CYCLE REGULATION AND CANCER Cellular Reproduction II THE CELL CYCLE Interphase G1- gap phase 1- cell grows and develops S- DNA synthesis phase- cell replicates each chromosome G2- gap phase 2- cell
CELL CYCLE REGULATION AND CANCER Cellular Reproduction II THE CELL CYCLE Interphase G1- gap phase 1- cell grows and develops S- DNA synthesis phase- cell replicates each chromosome G2- gap phase 2- cell
Supplementary Information
 Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
Assessment of the biological effects of strange Radiation 1
 Annales de la Fondation Louis de Broglie, Volume 31 no 4, 2006 463 Assessment of the biological effects of strange Radiation 1 E.A. Pryakhin, G.A. Tryapitsina a, L.I. Urutskoyev b, A.V. Akleyev c a Chalyabinsk
Annales de la Fondation Louis de Broglie, Volume 31 no 4, 2006 463 Assessment of the biological effects of strange Radiation 1 E.A. Pryakhin, G.A. Tryapitsina a, L.I. Urutskoyev b, A.V. Akleyev c a Chalyabinsk
T cell development October 28, Dan Stetson
 T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung
 Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung immortalized broncho-epithelial cells (AALE cells) expressing
Supplementary Figure 1. A. Bar graph representing the expression levels of the 19 indicated genes in the microarrays analyses comparing human lung immortalized broncho-epithelial cells (AALE cells) expressing
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Supplemental Table 1 Molecular Profile of the SCLC Cell Line Panel
 Supplemental Table 1 Molecular Profile of the SCLC Cell Line Panel p53 RB Myc Cell Line Mutation A Mutation A Amplification B COR-L103 C p.y234c p.d584e L-Myc NCI-H526 p.s33_splice None N-Myc NCI-H1048
Supplemental Table 1 Molecular Profile of the SCLC Cell Line Panel p53 RB Myc Cell Line Mutation A Mutation A Amplification B COR-L103 C p.y234c p.d584e L-Myc NCI-H526 p.s33_splice None N-Myc NCI-H1048
IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia
 Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Supplementary. presence of the. (c) mrna expression. Error. in naive or
 Figure 1. (a) Naive CD4 + T cells were activated in the presence of the indicated cytokines for 3 days. Enpp2 mrna expression was measured by qrt-pcrhr, infected with (b, c) Naive CD4 + T cells were activated
Figure 1. (a) Naive CD4 + T cells were activated in the presence of the indicated cytokines for 3 days. Enpp2 mrna expression was measured by qrt-pcrhr, infected with (b, c) Naive CD4 + T cells were activated
Supporting Information
 Supporting Information Chan et al. 1.173/pnas.9654916 A Patient B Xenograft C * remaining feature of normal lymph node * * * D lymphocytes Infiltrating transitional carcinoma cells E Enlarged axillary
Supporting Information Chan et al. 1.173/pnas.9654916 A Patient B Xenograft C * remaining feature of normal lymph node * * * D lymphocytes Infiltrating transitional carcinoma cells E Enlarged axillary
Balancing intestinal and systemic inflammation through cell type-specific expression of
 Supplementary Information Balancing intestinal and systemic inflammation through cell type-specific expression of the aryl hydrocarbon receptor repressor Olga Brandstätter 1,2,6, Oliver Schanz 1,6, Julia
Supplementary Information Balancing intestinal and systemic inflammation through cell type-specific expression of the aryl hydrocarbon receptor repressor Olga Brandstätter 1,2,6, Oliver Schanz 1,6, Julia
Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A.
 Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A. Upper part, three-primer PCR strategy at the Mcm3 locus yielding
Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A. Upper part, three-primer PCR strategy at the Mcm3 locus yielding
Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors
 SUPPLEMENTARY INFORMATION Articles https://doi.org/1.138/s41591-18-135-2 In the format provided by the authors and unedited. Sequestration of T cells in bone marrow in the setting of glioblastoma and other
SUPPLEMENTARY INFORMATION Articles https://doi.org/1.138/s41591-18-135-2 In the format provided by the authors and unedited. Sequestration of T cells in bone marrow in the setting of glioblastoma and other
 a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
LDR and its potential application in clinics
 LDR and its potential application in clinics Lu Cai Departments of Medicine & Radiation Oncology University it of Louisville School of Medicine LDR effects Hormesis Adaptive response Bystander effects
LDR and its potential application in clinics Lu Cai Departments of Medicine & Radiation Oncology University it of Louisville School of Medicine LDR effects Hormesis Adaptive response Bystander effects
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Clinical Studies supporting the use of Antioxidants during Chemotherapy and Radiation
 Clinical Studies supporting the use of Antioxidants during Chemotherapy and Radiation Poly-MVA ORAC Analysis As you are reading the 5 Clinical Studies below, pay close attention to the list of Antioxidants
Clinical Studies supporting the use of Antioxidants during Chemotherapy and Radiation Poly-MVA ORAC Analysis As you are reading the 5 Clinical Studies below, pay close attention to the list of Antioxidants
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supporting Information
 Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supporting Information Franco et al. 10.1073/pnas.1015557108 SI Materials and Methods Drug Administration. PD352901 was dissolved in 0.5% (wt/vol) hydroxyl-propyl-methylcellulose, 0.2% (vol/vol) Tween
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
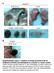 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
islets scored 1 week month months
 Supplementary Table 1. Sampling parameters for the morphometrical analyses Time (post- DT) Control mice (age-matched) α-cells mice pancreatic surface (mm 2 ) scored DT-treated mice islets scored mice pancreatic
Supplementary Table 1. Sampling parameters for the morphometrical analyses Time (post- DT) Control mice (age-matched) α-cells mice pancreatic surface (mm 2 ) scored DT-treated mice islets scored mice pancreatic
Supplemental Material
 Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
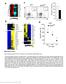 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figures
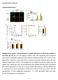 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure S1: Defective heterochromatin repair in HGPS progeroid cells
 Supplementary Figure S1: Defective heterochromatin repair in HGPS progeroid cells Immunofluorescence staining of H3K9me3 and 53BP1 in PH and HGADFN003 (HG003) cells at 24 h after γ-irradiation. Scale bar,
Supplementary Figure S1: Defective heterochromatin repair in HGPS progeroid cells Immunofluorescence staining of H3K9me3 and 53BP1 in PH and HGADFN003 (HG003) cells at 24 h after γ-irradiation. Scale bar,
Pharmacologic inhibition of histone demethylation as a therapy for pediatric brainstem glioma
 Supplementary information for: Pharmacologic inhibition of histone demethylation as a therapy for pediatric brainstem glioma Rintaro Hashizume 1, Noemi Andor 2, Yuichiro Ihara 2, Robin Lerner 2, Haiyun
Supplementary information for: Pharmacologic inhibition of histone demethylation as a therapy for pediatric brainstem glioma Rintaro Hashizume 1, Noemi Andor 2, Yuichiro Ihara 2, Robin Lerner 2, Haiyun
Basics of Radiation Biology
 Basics of Radiation Biology Sally A. Amundson Columbia University Center for Radiological Research http://www.cmcr.columbia.edu/ Overview Radiation damage to cells DNA Effects of radiation damage on cells
Basics of Radiation Biology Sally A. Amundson Columbia University Center for Radiological Research http://www.cmcr.columbia.edu/ Overview Radiation damage to cells DNA Effects of radiation damage on cells
Basics of Radiation Biology
 Basics of Radiation Biology Sally A. Amundson Columbia University Center for Radiological Research http://www.cmcr.columbia.edu/ Overview Radiation damage to cells DNA Effects of radiation damage on cells
Basics of Radiation Biology Sally A. Amundson Columbia University Center for Radiological Research http://www.cmcr.columbia.edu/ Overview Radiation damage to cells DNA Effects of radiation damage on cells
ToxStrategies, Inc. and Summit Toxicology
 Deborah Proctor Chad Thompson, Mark Harris, Mina Suh, Laurie Haws, Chris Kirman and Sean Hays ToxStrategies, Inc. and Summit Toxicology November 2012 Research Project funded by the Cr 6 Panel of the American
Deborah Proctor Chad Thompson, Mark Harris, Mina Suh, Laurie Haws, Chris Kirman and Sean Hays ToxStrategies, Inc. and Summit Toxicology November 2012 Research Project funded by the Cr 6 Panel of the American
Supplemental Information. Increased 4E-BP1 Expression Protects. against Diet-Induced Obesity and Insulin. Resistance in Male Mice
 Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Early Embryonic Development
 Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Supplementary Figure 1
 Combination index (CI) Supplementary Figure 1 2. 1.5 1. Ishikawa AN3CA Nou-1 Hec-18.5...2.4.6.8 1. Fraction affected (Fa) Supplementary Figure 1. The synergistic effect of PARP inhibitor and PI3K inhibitor
Combination index (CI) Supplementary Figure 1 2. 1.5 1. Ishikawa AN3CA Nou-1 Hec-18.5...2.4.6.8 1. Fraction affected (Fa) Supplementary Figure 1. The synergistic effect of PARP inhibitor and PI3K inhibitor
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/8/339/339ra69/dc1 Supplementary Materials for The caspase-8 inhibitor emricasan combines with the SMAC mimetic birinapant to induce necroptosis and
www.sciencetranslationalmedicine.org/cgi/content/full/8/339/339ra69/dc1 Supplementary Materials for The caspase-8 inhibitor emricasan combines with the SMAC mimetic birinapant to induce necroptosis and
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell. Tumor type
 Fig S1 A B Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell Tumor type -/- (n = 46) Q/- (n = 76) Q/Q (n = 31) Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0
Fig S1 A B Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell Tumor type -/- (n = 46) Q/- (n = 76) Q/Q (n = 31) Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0
Radiation biology. Dr. István Voszka. Department of Biophysics and Radiation Biology. Grotthus (1815) - Draper (1845)
 Radiation biology Dr. István Voszka Department of Biophysics and Radiation Biology Wilhelm Conrad Röntgen Antoine Henri Becquerel 1845-1923 1852-1908 1895 x-radiation 1896 - radioactivity Grotthus (1815)
Radiation biology Dr. István Voszka Department of Biophysics and Radiation Biology Wilhelm Conrad Röntgen Antoine Henri Becquerel 1845-1923 1852-1908 1895 x-radiation 1896 - radioactivity Grotthus (1815)
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was
 Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
SUPPLEMENTARY INFORMATION. Rett Syndrome Mutation MeCP2 T158A Disrupts DNA Binding, Protein Stability and ERP Responses
 SUPPLEMENTARY INFORMATION Rett Syndrome Mutation T158A Disrupts DNA Binding, Protein Stability and ERP Responses Darren Goffin, Megan Allen, Le Zhang, Maria Amorim, I-Ting Judy Wang, Arith-Ruth S. Reyes,
SUPPLEMENTARY INFORMATION Rett Syndrome Mutation T158A Disrupts DNA Binding, Protein Stability and ERP Responses Darren Goffin, Megan Allen, Le Zhang, Maria Amorim, I-Ting Judy Wang, Arith-Ruth S. Reyes,
Supplementary Materials for. c-abl Activation Plays a Role in α-synucleinopathy Induced Neurodegeneration
 Supplementary Materials for c-abl Activation Plays a Role in α-synucleinopathy Induced Neurodegeneration Saurav Brahmachari, Preston Ge, Su Hyun Lee, Donghoon Kim, Senthilkumar S. Karuppagounder, Manoj
Supplementary Materials for c-abl Activation Plays a Role in α-synucleinopathy Induced Neurodegeneration Saurav Brahmachari, Preston Ge, Su Hyun Lee, Donghoon Kim, Senthilkumar S. Karuppagounder, Manoj
Activation of a PML/TP53 axis underlies acute promyelocytic. leukemia cure
 Activation of a PML/TP53 ais underlies acute promyelocytic leukemia cure Julien Ablain, Kim Rice, Hassane Soilihi, Aurélien de Reynies, Saverio Minucci & Hugues de Thé a PLZF-RARA vs PLZF-RARA pathway
Activation of a PML/TP53 ais underlies acute promyelocytic leukemia cure Julien Ablain, Kim Rice, Hassane Soilihi, Aurélien de Reynies, Saverio Minucci & Hugues de Thé a PLZF-RARA vs PLZF-RARA pathway
Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation
 Research article Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation Teresa G. Tessner, Filipe Muhale, Terrence E. Riehl,
Research article Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation Teresa G. Tessner, Filipe Muhale, Terrence E. Riehl,
Modelling of Biological Processes
 Modelling of Biological Processes WHAT HAPPENS AFTER EARLY MOLECULAR DAMAGE? Stephen McMahon Queen s University, Belfast, Northern Ireland 3 rd August 2016 1 Do we need biology? The Linear-quadratic relationship
Modelling of Biological Processes WHAT HAPPENS AFTER EARLY MOLECULAR DAMAGE? Stephen McMahon Queen s University, Belfast, Northern Ireland 3 rd August 2016 1 Do we need biology? The Linear-quadratic relationship
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
TITLE: Reduction of Radiation- or Chemotherapy-Induced Toxicity by Specific Expression of Anti-Apoptotic Molecules in Normal Cells
 AD Award Number: DAMD17-01-1-0304 TITLE: Reduction of Radiation- or Chemotherapy-Induced Toxicity by Specific Expression of Anti-Apoptotic Molecules in Normal Cells PRINCIPAL INVESTIGATOR: Naoto T. Ueno,
AD Award Number: DAMD17-01-1-0304 TITLE: Reduction of Radiation- or Chemotherapy-Induced Toxicity by Specific Expression of Anti-Apoptotic Molecules in Normal Cells PRINCIPAL INVESTIGATOR: Naoto T. Ueno,
Infect MCF-7 cells carrying dcas9-vp64 + psm2-p65-hsf1 with SAM library or vector. Introduce AKT reporter
 Infect MCF-7 cells carrying dcas9-vp64 + psm2-p65-hsf1 with SM library or vector Introduce reporter Grow cells in presence of puromycin for 5 days Vector control SM library fewer surviving cells More surviving
Infect MCF-7 cells carrying dcas9-vp64 + psm2-p65-hsf1 with SM library or vector Introduce reporter Grow cells in presence of puromycin for 5 days Vector control SM library fewer surviving cells More surviving
