Energy-Efficient Extraction of Fuel and Chemical Feedstocks from Algae. (Supporting Information)
|
|
|
- Maryann Jackson
- 6 years ago
- Views:
Transcription
1 Energy-Efficient Extraction of Fuel and Chemical Feedstocks from Algae (Supporting Information) Rodrigo E. Teixeira Department of Chemical and Materials Engineering University of Alabama in Huntsville Huntsville, AL
2 Supplementary Figures Mitochondria Vacuole Starch grains Eukaryotic algae Lipid vesicle Nucleus Lipid vesicle a b c Lysis Extraction Ionic liquid phase Chloroform phase Chloroplasts Ionic liquid Ionic liquid hydrolysate Mass spectra H H H H H n H Cell wall polysaccharides Ionic liquid, 5-10% water C, < 50 min H H H H H Monomers C12-20 fatty acids sterols glycerophospholipids glycerolipids Supplementary Figure 1: Experimental schematic and summary of results. (a) Algae water suspensions initially mixed and dissolved in ionic liquids at temperatures of 100 to 140 C and atmospheric pressure. The reaction scheme resulting in oligomerization of cell wall polysaccharides is shown. (b) Created cell-free mixture of algae in ionic liquid (hydrolysate). Some free-floating chloroplasts are seen and the background changes to light-green due to released chlorophyll. (c) Chloroform-extracted hydrolysate after centrifugation. The chloroform phase contained a wide range of lipids matching the profiles expected from algae.
3 Supplementary Figure 2: Micrographs of Chlorella pyrenoidosa and Scenedesmus quadricauda. (a) C. pyrenoidosa and (b) S. quadricauda micrographs after (from left to right) 1 hour in boiling water, sonication for 30 min, direct sample of batch, reaction in [BMIM]Cl at 100 to 130 C (run #7 and #13), reaction in [BMIM]Cl at 90 to 100 C (run #4). Lower-right bar measures 10 μm.
4 Supplementary Figure 3: Micrographs of Chlorococcum hypnosporum and Chlorella vulgaris. (a) C. hypnosporum and (b) C. vulgaris micrographs after (from left to right) sonication for 30 min, direct sample of batch, reaction in [BMIM]Cl at 110 C (run #12 and #9). Lower-right bar measures 10 μm.
5 Supplementary Figure 4: Micrographs of Chlamydomonas moewusii during lysis. (a) Direct sample of batch. (b) After 5 min in [BMIM]Cl at 120 C, and (c) 10 min latter (run #10). Lower-right bar measures 10 μm.
6 Electronic Supplementary Material (ESI) for Green Chemistry Supplementary Figure 5: Hydrophobic ionic liquids. Ionic liquids formed by the cation 1butyl-3-methylimidazolium paired with the anions (a) hexafluorophosphate and (b) bis(trifluoromethylsulfonyl)imide imparted hydrophobic characters. These ionic liquids did not dissolve or lyse algae (Chlorella pyrenoidosa shown). Instead, after evaporation of water, clustered cell masses were formed.
7 Supplementary Text Cell wall toughness. The toughness of semi-crystalline cell wall microfibril networks giving structural support to algae cell walls was estimated from strain measurements by other researchers of bacterial cellulose sheets grown from Gluconacetobacter xylinus 1. Both materials are of microbial origin and were assumed to be of roughly similar degree of polymerization, crystallinity and structure. Previous measurements of the engineering stress on a tensile test machine were inadequate due to difficulties in assessing the local strain and the effective cross-sectional area, which likely changed during deformation. Instead, stress-strain (σ-ε) curves were determined micromechanically, using a Raman spectrometer microscope to measure molecular deformation. By measuring the strain-calibrated displacement of the 1095 cm -1 band, a Young s modulus, E, of 41 GPa was determined for the network. Interestingly, assuming random fiber orientations, this value corresponds to a single fibril modulus of 114 GPa, similar to the stiffness of high-grade Kevlar (124 GPa). Since the micromechanical stress-strain relationship was approximately linear, the toughness (γ) could be estimated as: γ = ε f 0 σ dε 1 2 ε 2 f E = 1 2 (0.026)2 ( ) = 14 MPa or ~ 9 kj/kg assuming a cellulose density of 1.5 Mg/m 3. For comparison, the toughness of graphite (carbon) fibers are ~ 12 kj/kg ( 2 ). Algae surface-to-mass ratio. From Northcote et al. 3 the radius of Chlorella pyrenoidosa is ~ 1.8 μm, which was adopted as a reasonable size for other algae. Assuming a spheroidal shape and a density of ~ 1 kg/m 3, the cell surface-to-volume and surface-to-mass ratios are: S V = 3 r = m m 2 /kg. Note that the mass basis assumes a well-hydrated cell.
8 Energy balance calculation details. To facilitate calculation of the dissolution and hydrolysis enthalpies, cellulose was adopted as a representative polysaccharide. The dissolution enthalpy of cellulose in 1-butyl-3-methylimidazolium chloride at T = 373 K was calculated from: ( ) $ Δh m i = RT ln x L L i γ i T m T 1 i. Here, Δh i m and T i m are the melting enthalpy and melting temperature of pure cellulose, respectively. However, the melting temperature is unknown, so the decomposition temperature (773 K) was adopted, giving an upper-bound estimate for the dissolution enthalpy. The mole fraction of dissolved cellulose at equilibrium with its solid form is x i L, which was measured experimentally to be 25% by weight 4. The activity coefficient between cellulose and ionic liquid is γ i L. The value γ i L = was obtained from calculations combining quantum mechanical computation of surface charge-density in a conductor, electrostatic interaction energy with a dielectric continuum and screening charge probability distributions 5. This gives a dissolution enthalpy of < 24 kj/kg. Assuming that cell wall polysaccharides constitute ~ 14% of dry biomass 3, dissolution consumes at most ~ 3 kj/kg. The measured enthalpy change for the hydrolysis of cellulose oligomers in aqueous buffer was measured at ~ -4 kj/kg of cellulose 6, or -560 J/kg of biomass. It was assumed that an approach temperature of 5 K is feasible and no fouling occurs in the plate heat exchanger (counter-current geometry). The reactor and ionic liquid recycle were treated as adiabatic except for heat loss through the lipid output stream, calculated straightforwardly using the heat capacity of vegetable oil (1.67 kj/kg-k). The separation of
9 solutes from ionic liquid and its recycle was assumed to consume 45% of the total process energy, following what is typically observed at chemical processing plants. The fraction of the total process energy supplied as work, or electricity, was taken to be the same as conventional algae extraction, or ~ 26% 7. Supplementary Table 1: Energy balance for algae extraction process. Quantity Unit Basis Reference Lipid content 0.18 w/w Lardon et al. (2009) Carbohydrate content 0.50 w/w Lardon et al. (2009) Protein content 0.28 w/w Lardon et al. (2009) Cell wall content 0.14 w/w Northcote et al. (1958) Input cell concentration 0.10 w/w Heat ex. approach temperature 5 K Heat ex. heat loss 209 kj/kg Dry biomass Polysaccharide dissolution enthalpy 24 kj/kg Polysaccharide Polysaccharide hydrolysis enthalpy -4 kj/kg Polysaccharide Karim et al. (1989) Heat loss via lipid stream 125 kj/kg Lipid Reactor net enthalpy change 33 kj/kg Dry biomass Separation contribution 0.45 Total separation energy 198 kj/kg Dry biomass Total heat 0.3 MJ/kg Dry biomass Total electricity 0.1 MJ/kg Dry biomass Supplementary Table 1 Legend: The energy balance for the steady state process was calculated from contributions by the heat exchanger, reactor, and ionic liquid chromatography (separation) steps. Well-to-Station net energy gain calculation details. The WTS model started from values calculated by Lardon et al. for all scenarios 7. Please refer to the original article for a description of the assumptions. In the present work, all values were normalized to the basis of 1 kg of dry algae biomass. The energy requirements for wet extraction related to pressure homogenization. Here, both the values from Lardon, which were extrapolations form dry extractions, and actual reported cases of algae processing 8, 9 were taken into account to derive a range from 4 to 9 MJ/
10 kg. The partition of this total energy into heat and electricity followed the dry extraction case. Even though algae compositions varied significantly between normal and low N conditions, their impact on the ionic liquid based process energy was negligible. Material flows from the original study were beyond the present scope. Produced energy was estimated as extracted lipid plus oilcake (mostly sugars and proteins) assuming 100% energy extraction of all components. Supplementary References 1.$ Y.-C. Hsieh, H. Yano, M. Nogi and S. Eichhorn, Cellulose, 2008, 15, $ S. Chand, Journal of Materials Science, 2000, 35, $ D. Northcote, K. Goulding and R. Horne, Biochemical Journal, 1958, 70, $ R. Swatloski, S. Spear, J. Holbrey and R. Rogers, J. Am. Chem. Soc., 2002, 124, $ J. Kahlen, K. Masuch and K. Leonhard, Green Chemistry, 2010, $ N. Karim and S. Kidokoro, Thermochimica Acta, 2004, 412, $ L. Lardon, A. Helias, B. Sialve, J. Steyer and. Bernard, Environmental Science & Technology, 2009, 43, $ H. Greenwell, L. Laurens, R. Shields, R. Lovitt and K. Flynn, Journal of the Royal Society Interface, 2010, 7, $ L. Xu, D. Brilman, J. Withag, G. Brem and S. Kersten, Bioresource Technology, 2011, 102,
Utilizing Biosolids As Packaging or Building Material
 Utilizing Biosolids As Packaging or Building Material Methods and equipment necessary to produce value added products from biosolids for packaging and building material applications. John F. Hunt, P.E.
Utilizing Biosolids As Packaging or Building Material Methods and equipment necessary to produce value added products from biosolids for packaging and building material applications. John F. Hunt, P.E.
Bio 12 Chapter 2 Test Review
 Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
EH1008 Biomolecules. Inorganic & Organic Chemistry. Water. Lecture 2: Inorganic and organic chemistry.
 EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
Sample Questions BSC1010C Chapters 5-7
 Sample Questions BSC1010C Chapters 5-7 1. Which type of lipid is most important in biological membranes? a. oils b. fats c. wax d. phospholipids e. triglycerides 2. Which type of interaction stabilizes
Sample Questions BSC1010C Chapters 5-7 1. Which type of lipid is most important in biological membranes? a. oils b. fats c. wax d. phospholipids e. triglycerides 2. Which type of interaction stabilizes
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) One-Step Encapsulation and Triggered Release Based on
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) One-Step Encapsulation and Triggered Release Based on
Polyoxometalate Macroion Induced Phase and Morphology
 Polyoxometalate Macroion Induced Phase and Morphology Instability of Lipid Membrane Benxin Jing a, Marie Hutin c, Erin Connor a, Leroy Cronin c,* and Yingxi Zhu a,b,* a Department of Chemical and Biomolecular
Polyoxometalate Macroion Induced Phase and Morphology Instability of Lipid Membrane Benxin Jing a, Marie Hutin c, Erin Connor a, Leroy Cronin c,* and Yingxi Zhu a,b,* a Department of Chemical and Biomolecular
The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions
 Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
2.1.1 Biological Molecules
 2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
3. When he discovered enzymes in 1897, Eduard Buchner was investigating the causes of. disease antisepsis spontaneous generation fermentation
 Chapter 2 1. Which of the following elements forms the backbone of organic molecules? carbon fluorine chlorine sodium carbon 2. The smallest chemical unit of matter is the. neutron element atom proton
Chapter 2 1. Which of the following elements forms the backbone of organic molecules? carbon fluorine chlorine sodium carbon 2. The smallest chemical unit of matter is the. neutron element atom proton
Basic Structure of a Cell
 Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Name a property of. water why is it necessary for life?
 02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
02.09.18 Name a property of + water why is it necessary for life? n Cohesion n Adhesion n Transparency n Density n Solvent n Heat capacity + Macromolecules (2.3 & some of 2.4) + Organic Molecules All molecules
Plant Power! From traditional crops to alt proteins. Atze Jan van der Goot. Food Protein Vision, Amsterdam, 8 March 2018
 Plant Power! From traditional crops to alt proteins Atze Jan van der Goot Food Protein Vision, Amsterdam, 8 March 2018 The problem: Making same food requires more recourses Tilman, PNAS 20260 (2011) Global
Plant Power! From traditional crops to alt proteins Atze Jan van der Goot Food Protein Vision, Amsterdam, 8 March 2018 The problem: Making same food requires more recourses Tilman, PNAS 20260 (2011) Global
FIRST MIDTERM EXAMINATION
 FIRST MIDTERM EXAMINATION 1. True or false: because enzymes are produced by living organisms and because they allow chemical reactions to occur that would not otherwise occur, enzymes represent an exception
FIRST MIDTERM EXAMINATION 1. True or false: because enzymes are produced by living organisms and because they allow chemical reactions to occur that would not otherwise occur, enzymes represent an exception
Human Epithelial Cells
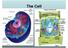 The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
LIFE IS CELLULAR. Cell Theory. Cells Are Small. Prokaryotic Cell 10/4/15. Chapter 7 Cell Structure and Function
 Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
2-2 Properties of Water
 2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
2-2 Properties of Water 1 A. The Water Molecule o o o Water is polar Hydrogen bonds form between water molecules Properties of Water: cohesion adhesion capillary action high specific heat ice floats good
Chapter 5: Analysis of water content, total solids & water activity
 Chapter 5: Analysis of water content, total solids & water activity 1 Water is an essential constituent of many foods. It may occur as an intracellular or extracellular component in vegetable and animal
Chapter 5: Analysis of water content, total solids & water activity 1 Water is an essential constituent of many foods. It may occur as an intracellular or extracellular component in vegetable and animal
SAM Teachers Guide Lipids and Carbohydrates
 SAM Teachers Guide Lipids and Carbohydrates Overview Students will explore the structure and function of two of the four major macromolecules, lipids and carbohydrates. They will look specifically at the
SAM Teachers Guide Lipids and Carbohydrates Overview Students will explore the structure and function of two of the four major macromolecules, lipids and carbohydrates. They will look specifically at the
Chapter 11 Nutrition: Food for Thought
 Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Plasma Membrane Function
 Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
The Structure and Function of Large Biological Molecules. Chapter 5
 The Structure and Function of Large Biological Molecules Chapter 5 The Molecules of Life Living things made up of 4 classes of large biological molecules (macromolecules) : 1. Carbohydrates 2. Lipids 3.
The Structure and Function of Large Biological Molecules Chapter 5 The Molecules of Life Living things made up of 4 classes of large biological molecules (macromolecules) : 1. Carbohydrates 2. Lipids 3.
CHEMISTRY OF LIFE 05 FEBRUARY 2014
 CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
3.1 Carbon is Central to the Living World
 BIOL 100 Ch. 3 1 3.1 Carbon is Central to the Living World Carbon Central element to life Most biological molecules are built on a carbon framework. Organic molecules Humans 18.5% Carbon Why is Carbon
BIOL 100 Ch. 3 1 3.1 Carbon is Central to the Living World Carbon Central element to life Most biological molecules are built on a carbon framework. Organic molecules Humans 18.5% Carbon Why is Carbon
2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE
 Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE Matter Cycles The atoms in living systems are common on this planet.
Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE Matter Cycles The atoms in living systems are common on this planet.
OCR (A) Biology A-level
 OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
Biochemical Concepts. Section 4.6 The Chemistry of Water. Pre-View 4.6. A Covalent Polar Molecule
 Biochemical Concepts Section 4.6 The Chemistry of Water Pre-View 4.6 Polar molecule a molecule that has a partial positive charge on one end and a partial negative charge on the other end Hydrogen bond
Biochemical Concepts Section 4.6 The Chemistry of Water Pre-View 4.6 Polar molecule a molecule that has a partial positive charge on one end and a partial negative charge on the other end Hydrogen bond
Week 2. Macromolecules
 Week 2 In living organisms, smaller molecules are often attached to each other to make larger molecules. These smaller molecules are sometimes called monomers, and the larger molecules made from these
Week 2 In living organisms, smaller molecules are often attached to each other to make larger molecules. These smaller molecules are sometimes called monomers, and the larger molecules made from these
and hydrophilic and how they relate to solubility.
 o o o and hydrophilic and how they relate to solubility. o o o o o o o o Page 1: Introduction Page 2: 1. Hydrocarbons are referred to as organic molecules with a "backbone." Take a snapshot of the hydrocarbon
o o o and hydrophilic and how they relate to solubility. o o o o o o o o Page 1: Introduction Page 2: 1. Hydrocarbons are referred to as organic molecules with a "backbone." Take a snapshot of the hydrocarbon
Data File. Sephadex ion exchange media. Ion exchange chromatography. Introduction. Sephadex ion exchangers General description
 A m e r s h a m B i o s c i e n c e s Sephadex ion exchange media Data File Ion exchange chromatography Based on well documented and well proven Sephadex base matrix Simple and economical to use Very high
A m e r s h a m B i o s c i e n c e s Sephadex ion exchange media Data File Ion exchange chromatography Based on well documented and well proven Sephadex base matrix Simple and economical to use Very high
Chapters 2 and 3. Pages and Pages Prayer Attendance Homework
 Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
1.2 introduction to the cell. me239 mechanics of the cell. 1.2 introduction to the cell. 1.2 introduction to the cell.
 2. introduction to mechanics prokaryotic cells Figure 1.1 Prokaryotic cell. Cell without a nucleus. the inner life of a cell, viel & lue, harvard [2006] me239 mechanics of the cell 1 eukaryotic cells 1.2
2. introduction to mechanics prokaryotic cells Figure 1.1 Prokaryotic cell. Cell without a nucleus. the inner life of a cell, viel & lue, harvard [2006] me239 mechanics of the cell 1 eukaryotic cells 1.2
Chapter 1 Membrane Structure and Function
 Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Supplementary Material
 Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2018 Supplementary Material 1 Supplemental Figures Supplemental Figure S1. Mechanical properties
Electronic Supplementary Material (ESI) for Integrative Biology. This journal is The Royal Society of Chemistry 2018 Supplementary Material 1 Supplemental Figures Supplemental Figure S1. Mechanical properties
Organic Compounds. (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids
 Organic Compounds (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids Carbon s Bonding Behavior Outer shell of carbon has 4 electrons; can hold 8 Each carbon atom can form covalent bonds with
Organic Compounds (Carbon Compounds) Carbohydrates Lipids Proteins Nucleic Acids Carbon s Bonding Behavior Outer shell of carbon has 4 electrons; can hold 8 Each carbon atom can form covalent bonds with
Introduction to Membrane
 Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
Heinrich Strathmann Introduction to Membrane Science and Technology WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa V Preface Symbols XIII XV 1 Introduction 2 1.1 Overview of Membrane Science and Technology
The Structure and Function of Large Biological Molecules
 Chapter 5 The Structure and Function of Large Biological Molecules PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley
Chapter 5 The Structure and Function of Large Biological Molecules PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley
CARBOHYDRATE BOOKLET ANSWERS
 CARBOHYDRATE BOOKLET ANSWERS FOR: QER QUESTION APPLICATION AND EXTENSION QUESTIONS. FEEDBACK CAN BE VIEWED AT: https://thiacin.com/carbohydrateqerappextfeedback LONG ANSWER QUESTIONS WITH QER The long
CARBOHYDRATE BOOKLET ANSWERS FOR: QER QUESTION APPLICATION AND EXTENSION QUESTIONS. FEEDBACK CAN BE VIEWED AT: https://thiacin.com/carbohydrateqerappextfeedback LONG ANSWER QUESTIONS WITH QER The long
Unit 1: Level of organization Chemistry (macromolecules)
 Unit 1: Level of organization hemistry (macromolecules) Levels of organization: Unit 1 Small to large: atoms molecules macromolecules Then larger (microscopic stuff): cells and their parts (organelles)
Unit 1: Level of organization hemistry (macromolecules) Levels of organization: Unit 1 Small to large: atoms molecules macromolecules Then larger (microscopic stuff): cells and their parts (organelles)
The power point notes are behind a locked page, so you ll need the following password to access them: kapabio123
 AP Biology Summer Assignment Due date: Week of September 8, 2017. Purpose: The following chapters are mostly review from PreAP biology. Completing this assignment during the summer prior to the school
AP Biology Summer Assignment Due date: Week of September 8, 2017. Purpose: The following chapters are mostly review from PreAP biology. Completing this assignment during the summer prior to the school
EQ What are the 4 biomolecules and their monomers?
 EQ What are the 4 biomolecules and their monomers? Monomers: Fatty Acids Amino acids Nucleotides Monosaccharides Drawings Chemistry of Life Chapter 3 Biomolecules Chapter 3 Section 3 Building Blocks of
EQ What are the 4 biomolecules and their monomers? Monomers: Fatty Acids Amino acids Nucleotides Monosaccharides Drawings Chemistry of Life Chapter 3 Biomolecules Chapter 3 Section 3 Building Blocks of
PHOTOSYNTHESIS (7.5A)
 PHOTOSYNTHESISS (7.5A) 1) What kind of energy is necessary to initiate the process of photosynthesis? A. radiant B. heat C. electrical D. wind 2) What happens to the radiant energy absorbed by plants during
PHOTOSYNTHESISS (7.5A) 1) What kind of energy is necessary to initiate the process of photosynthesis? A. radiant B. heat C. electrical D. wind 2) What happens to the radiant energy absorbed by plants during
Macromolcules, Enzymes, & Cells Intro
 Name: Date: 1. The distortion (change in shape) of enzyme molecules which occurs at high temperatures is known as 5. A characteristic shared by all enzymes, hormones, and antibodies is that their function
Name: Date: 1. The distortion (change in shape) of enzyme molecules which occurs at high temperatures is known as 5. A characteristic shared by all enzymes, hormones, and antibodies is that their function
Chapter 5 Structure and Function Of Large Biomolecules
 Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
Domain 2: Matter. 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. (EK2.A.3) 1.
 Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. (EK2.A.3) 1. Matter exchange The atoms in living systems are common on this planet.
Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. (EK2.A.3) 1. Matter exchange The atoms in living systems are common on this planet.
First discovered in 1665 since then every organism observed with microscopes shows cells
 The Cell Cell theory (1838): 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
The Cell Cell theory (1838): 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
From Atoms to Cells: Fundamental Building Blocks. Models of atoms. A chemical connection
 From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
Chapter 3. Chapter 3. Bellringer. Objectives. Chapter 3. Chapter 3. Vocabulary. Cell Wall. Identify the different parts of a eukaryotic cell.
 Bellringer Objectives Identify the different parts of a eukaryotic cell. Explain the function of each part of a eukaryotic cell. Plant cells and animal cells have many cell parts in common. But they also
Bellringer Objectives Identify the different parts of a eukaryotic cell. Explain the function of each part of a eukaryotic cell. Plant cells and animal cells have many cell parts in common. But they also
In this study, effect of different high-boiling-organic solvent (ethanolamine, diethylene glycol and
 ISESCO JOURNAL of Science and Technology Vol. 12 No 21 High Boiling Solvent Pre-treatment of Hazelnut Shells for Enzymatic Hydrolysis Emir Zafer Hoşgün, Berrin Bozan Anadolu University, Engineering Faculty,
ISESCO JOURNAL of Science and Technology Vol. 12 No 21 High Boiling Solvent Pre-treatment of Hazelnut Shells for Enzymatic Hydrolysis Emir Zafer Hoşgün, Berrin Bozan Anadolu University, Engineering Faculty,
Cells & Transport. Chapter 7.1, 7.2, & 7.4
 Cells & Transport Chapter 7.1, 7.2, & 7.4 Do Now How big is a cell? How many cells are we made of? How many cells is the smallest living organism made of? Objectives Describe how cells were discovered
Cells & Transport Chapter 7.1, 7.2, & 7.4 Do Now How big is a cell? How many cells are we made of? How many cells is the smallest living organism made of? Objectives Describe how cells were discovered
Statement Starch Cellulose Glycogen glycosidic bonds present polymer of α-glucose unbranched chains only only found in plants
 1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
1 The statements in the table below refer to three polysaccharide molecules. Complete the table. If the statement is correct, place a tick ( ) in the box and if the statement is incorrect place a cross
Algal Biofuels Research: Using basic science to maximize fuel output. Jacob Dums, PhD candidate, Heike Sederoff Lab March 9, 2015
 Algal Biofuels Research: Using basic science to maximize fuel output Jacob Dums, PhD candidate, jtdums@ncsu.edu Heike Sederoff Lab March 9, 2015 Outline Research Approach Dunaliella Increase Oil Content
Algal Biofuels Research: Using basic science to maximize fuel output Jacob Dums, PhD candidate, jtdums@ncsu.edu Heike Sederoff Lab March 9, 2015 Outline Research Approach Dunaliella Increase Oil Content
POLYMER REINFORCEMENT
 POLYMER REINFORCEMENT CG2 NanoCoatings Inc. 2007 CG 2 NanoCoatings Inc. 21 Pine Needles Court, Suite 200, Ottawa, ON K2S 1G5 CANADA T 613.435.7747 F 413.638.3933 W www.cg2nanocoatings.com POLYMER REINFORCEMENT
POLYMER REINFORCEMENT CG2 NanoCoatings Inc. 2007 CG 2 NanoCoatings Inc. 21 Pine Needles Court, Suite 200, Ottawa, ON K2S 1G5 CANADA T 613.435.7747 F 413.638.3933 W www.cg2nanocoatings.com POLYMER REINFORCEMENT
Carbohydrates. 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds.
 Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
Name: Class: Date: Grade 10 Science Related Reading/Biology Carbohydrates Biology Gr10 1. Using the terms provided below, complete the concept map showing the characteristics of organic compounds. maltose
CONSIDERATIONS IN PROTEIN INGREDIENT USE: THE IMPACT OF PROCESSING AND MOLECULAR INTERACTIONS
 CONSIDERATIONS IN PROTEIN INGREDIENT USE: THE IMPACT OF PROCESSING AND MOLECULAR INTERACTIONS Baraem (Pam) Ismail Associate Professor Department of Food Science and Nutrition University of Minnesota May
CONSIDERATIONS IN PROTEIN INGREDIENT USE: THE IMPACT OF PROCESSING AND MOLECULAR INTERACTIONS Baraem (Pam) Ismail Associate Professor Department of Food Science and Nutrition University of Minnesota May
Objectives. To determine the differences between plant and animal cells To discover the structure and function of cellular organelles.
 Cell Organelles 3.2 Objectives To determine the differences between plant and animal cells To discover the structure and function of cellular organelles. Basic Cellular Structures Cell membrane (cytoplasmic
Cell Organelles 3.2 Objectives To determine the differences between plant and animal cells To discover the structure and function of cellular organelles. Basic Cellular Structures Cell membrane (cytoplasmic
The Cell and Its Chemical Compounds
 Cell Theory Cell - The basic unit of structure and function in living things. All of an organism s process or functions are carried out in the cell. Robert Hooke - One of the first people to observe cells
Cell Theory Cell - The basic unit of structure and function in living things. All of an organism s process or functions are carried out in the cell. Robert Hooke - One of the first people to observe cells
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
Biology. Slide 1 of 37. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Biology 1 of 37 2 of 37 The Chemistry of Carbon The Chemistry of Carbon Organic chemistry is the study of all compounds that contain bonds between carbon atoms. 3 of 37 Macromolecules Macromolecules Macromolecules
Standard 2 Exam Biology. 2. This macromolecule is responsible for short term energy storage and structural support in plants
 1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
MY BIOLOGY FINAL EXAM WORKBOOK
 NAME PER DATE MY BIOLOGY FINAL EXAM WORKBOOK DIRECTIONS: This study work book is due on the day of your final exam. Start now! After you have completed this study guide, you need to memorize it! 1. Look
NAME PER DATE MY BIOLOGY FINAL EXAM WORKBOOK DIRECTIONS: This study work book is due on the day of your final exam. Start now! After you have completed this study guide, you need to memorize it! 1. Look
ETHYLENE GLYCOL. Table 1.1 Physical properties of Ethylene glycol
 ETHYLENE GLYCOL Introduction [1]: Glycols are dihydric alcohols having an aliphatic carbon chain. They have the general chemical formula C n H 2n (OH) 2. is the simplest and the most important of the glycols.
ETHYLENE GLYCOL Introduction [1]: Glycols are dihydric alcohols having an aliphatic carbon chain. They have the general chemical formula C n H 2n (OH) 2. is the simplest and the most important of the glycols.
Mechanochemical Modification of Lignin and Application of the Modified Lignin for Polymer Materials
 Mechanochemical Modification of Lignin and Application of the Modified Lignin for Polymer Materials Jinwen Zhang Composite Materials and Engineering Center Washington State University Significance Petroleum-based
Mechanochemical Modification of Lignin and Application of the Modified Lignin for Polymer Materials Jinwen Zhang Composite Materials and Engineering Center Washington State University Significance Petroleum-based
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
Biological Molecules. Carbohydrates, Proteins, Lipids, and Nucleic Acids
 Biological Molecules Carbohydrates, Proteins, Lipids, and Nucleic Acids Organic Molecules Always contain Carbon (C) and Hydrogen (H) Carbon is missing four electrons Capable of forming 4 covalent bonds
Biological Molecules Carbohydrates, Proteins, Lipids, and Nucleic Acids Organic Molecules Always contain Carbon (C) and Hydrogen (H) Carbon is missing four electrons Capable of forming 4 covalent bonds
Cell Membranes Valencia college
 6 Cell Membranes Valencia college 6 Cell Membranes Chapter objectives: The Structure of a Biological Membrane The Plasma Membrane Involved in Cell Adhesion and Recognition Passive Processes of Membrane
6 Cell Membranes Valencia college 6 Cell Membranes Chapter objectives: The Structure of a Biological Membrane The Plasma Membrane Involved in Cell Adhesion and Recognition Passive Processes of Membrane
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2009 Examination #5 ANSWER KEY For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2009 Examination #5 ANSWER KEY For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles
 Supporting information for: Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles Georgios Rizis, Theo G. M. van de Ven*, Adi Eisenberg* Department of Chemistry, McGill University,
Supporting information for: Homopolymers as Structure-Driving Agents in Semicrystalline Block Copolymer Micelles Georgios Rizis, Theo G. M. van de Ven*, Adi Eisenberg* Department of Chemistry, McGill University,
Cellulase Inhibitors/Deactivators in Lignocellulosic Biomass
 Cellulase Inhibitors/Deactivators in Lignocellulosic Biomass Youngmi Kim *, Eduardo Ximenes, Nathan S. Mosier and Michael R. Ladisch LORRE, Purdue Univ. 32 nd Symposium on Biotechnology for Fuels and Chemicals
Cellulase Inhibitors/Deactivators in Lignocellulosic Biomass Youngmi Kim *, Eduardo Ximenes, Nathan S. Mosier and Michael R. Ladisch LORRE, Purdue Univ. 32 nd Symposium on Biotechnology for Fuels and Chemicals
Organic compounds. Lipids, Carbohydrates, Proteins, and Nucleic Acids
 Organic compounds Lipids, Carbohydrates, Proteins, and Nucleic Acids Essential for life Organic compounds: Contain carbon Most are covalently bonded Example: C 6 H 12 O 6 (Glucose) Inorganic Compounds:
Organic compounds Lipids, Carbohydrates, Proteins, and Nucleic Acids Essential for life Organic compounds: Contain carbon Most are covalently bonded Example: C 6 H 12 O 6 (Glucose) Inorganic Compounds:
H 2 O. Liquid, solid, and vapor coexist in the same environment
 Water H 2 O Liquid, solid, and vapor coexist in the same environment WATER MOLECULES FORM HYDROGEN BONDS Water is a fundamental requirement for life, so it is important to understand the structural and
Water H 2 O Liquid, solid, and vapor coexist in the same environment WATER MOLECULES FORM HYDROGEN BONDS Water is a fundamental requirement for life, so it is important to understand the structural and
Biochemistry. Chapter 6
 Biochemistry Chapter 6 Game Plan for Today. - Collect your papers - Hand back quests - Go over Amoeba Sister Chart - Biochem Notes - Video Carbohydrate Lab Food Label Lab! Testing For Carbohydrates Benedict's
Biochemistry Chapter 6 Game Plan for Today. - Collect your papers - Hand back quests - Go over Amoeba Sister Chart - Biochem Notes - Video Carbohydrate Lab Food Label Lab! Testing For Carbohydrates Benedict's
Diluted acid pretreatment for an integrated microalgae bio-refinery to produce lipid- and carbohydrate-based biofuels
 Diluted acid pretreatment for an integrated microalgae bio-refinery to produce lipid- and carbohydrate-based biofuels Tao Dong, Lieve Laurens, Nick Nagle, Stefanie Van Wychen, Nicholas Sweeney, Philip
Diluted acid pretreatment for an integrated microalgae bio-refinery to produce lipid- and carbohydrate-based biofuels Tao Dong, Lieve Laurens, Nick Nagle, Stefanie Van Wychen, Nicholas Sweeney, Philip
Topic 3: Molecular Biology
 Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
Topic 3: Molecular Biology 3.2 Carbohydrates and Lipids Essen=al Understanding: Carbon, hydrogen and oxygen are used to supply and store energy. Carbohydrates CARBOHYDRATES CHO sugars Primarily consist
Explain the reason for this difference in resolving power.
 1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
BIO 12 UNIT 2a CELL COMPOUNDS AND BIOLOGICAL MOLECULES
 IO 12 UNIT 2a LL OMPOUNS N IOLOGIL MOLULS 1. Water has many characteristics beneficial to life. ecause of (a) bonding between water molecules, it is a liquid at temperatures suitable for life. Water is
IO 12 UNIT 2a LL OMPOUNS N IOLOGIL MOLULS 1. Water has many characteristics beneficial to life. ecause of (a) bonding between water molecules, it is a liquid at temperatures suitable for life. Water is
2.2 Properties of Water
 2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
Macromolecules. Ch. 5 Macromolecules BIOL 222. Overview: The Molecules of Life. Macromolecules
 Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Ch. 5 Macromolecules BIOL 222 Overview: The Molecules of Life Macromolecules large molecules composed of thousands of covalently connected atoms Built from carbon backbone Also contain large numbers of
Biology Chapter 2 Review
 Biology Chapter 2 Review Vocabulary: Define the following words on a separate piece of paper. Element Compound Ion Ionic Bond Covalent Bond Molecule Hydrogen Bon Cohesion Adhesion Solution Solute Solvent
Biology Chapter 2 Review Vocabulary: Define the following words on a separate piece of paper. Element Compound Ion Ionic Bond Covalent Bond Molecule Hydrogen Bon Cohesion Adhesion Solution Solute Solvent
By Daniel C. Perrinez Morse Hall 103. Faculty Contact: Ihab H. Farag, Sc.D., P.E.
 By Daniel C. Perrinez Morse Hall 103 Faculty Contact: Ihab H. Farag, Sc.D., P.E. Overview Background Project Process Description Approach Chemicals, Equipment, and Wastes Status & Future Steps http://www.rbgsyd.nsw.gov.au/
By Daniel C. Perrinez Morse Hall 103 Faculty Contact: Ihab H. Farag, Sc.D., P.E. Overview Background Project Process Description Approach Chemicals, Equipment, and Wastes Status & Future Steps http://www.rbgsyd.nsw.gov.au/
FEEDBACK TUTORIAL LETTER TEST 2 SECOND SEMESTER 2018 BASIC SCIENCE [BSC410S]
![FEEDBACK TUTORIAL LETTER TEST 2 SECOND SEMESTER 2018 BASIC SCIENCE [BSC410S] FEEDBACK TUTORIAL LETTER TEST 2 SECOND SEMESTER 2018 BASIC SCIENCE [BSC410S]](/thumbs/87/96785039.jpg) FEEDBACK TUTORIAL LETTER TEST 2 SECOND SEMESTER 2018 BASIC SCIENCE [BSC410S] 1 FACULTY OF HEALTH AND APPLIED SCIENCES DEPARTMENT OF NATURAL AND APPLIED SCIENCES BSC PROGRAMME NAME ST. #:. QUALIFICATION(S):
FEEDBACK TUTORIAL LETTER TEST 2 SECOND SEMESTER 2018 BASIC SCIENCE [BSC410S] 1 FACULTY OF HEALTH AND APPLIED SCIENCES DEPARTMENT OF NATURAL AND APPLIED SCIENCES BSC PROGRAMME NAME ST. #:. QUALIFICATION(S):
2 3 Carbon Compounds (Macromolecules)
 2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
2 3 Carbon Compounds (Macromolecules) Slide 1 of 37 Organic Chemistry Organic chemistry is the study of all compounds that contain bonds between carbon atoms. Slide 2 of 37 Carbon Living organisms are
Macromolecules. Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own.
 Macromolecules Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own. Macromolecules are giant molecules made up of thousands or hundreds
Macromolecules Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own. Macromolecules are giant molecules made up of thousands or hundreds
Carbohydrates, Lipids, Proteins, and Nucleic Acids
 Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Don t Freak Out. Test on cell organelle on Friday!
 Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Unit 3: Chemistry of Life Mr. Nagel Meade High School
 Unit 3: Chemistry of Life Mr. Nagel Meade High School IB Syllabus Statements 3.2.1 Distinguish between organic and inorganic compounds. 3.2.2 Identify amino acids, glucose, ribose and fatty acids from
Unit 3: Chemistry of Life Mr. Nagel Meade High School IB Syllabus Statements 3.2.1 Distinguish between organic and inorganic compounds. 3.2.2 Identify amino acids, glucose, ribose and fatty acids from
the properties of carbon
 Carbon Compounds Learning Objectives Describe the unique qualities of carbon. Describe the structures and functions of each of the four groups of macromolecules. For each macromolecule you will need to
Carbon Compounds Learning Objectives Describe the unique qualities of carbon. Describe the structures and functions of each of the four groups of macromolecules. For each macromolecule you will need to
Improvement of enzymatic hydrolysis of a marine macro-alga by dilute acid hydrolysis pretreatment
 Improvement of enzymatic hydrolysis of a marine macro-alga by dilute acid hydrolysis pretreatment Parviz Yazdani 1*, Keikhosro Karimi 1,2, Mohammad J. Taherzadeh 2 1 Department of Chemical Engineering,
Improvement of enzymatic hydrolysis of a marine macro-alga by dilute acid hydrolysis pretreatment Parviz Yazdani 1*, Keikhosro Karimi 1,2, Mohammad J. Taherzadeh 2 1 Department of Chemical Engineering,
Peroxisomes. Endomembrane System. Vacuoles 9/25/15
 Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
Life s molecular diversity is based on the. properties of carbon. Chain Ring Branching chain
 Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
The building blocks of life.
 The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
Exam 1 SC135 spring 2011 Page 1
 xam 1 S135 spring 2011 Page 1 Name: ate: 1 Which other item is worth the same (has the same weight) as your lecture exams toward your final grade? quizzes writing assignments participation presentation
xam 1 S135 spring 2011 Page 1 Name: ate: 1 Which other item is worth the same (has the same weight) as your lecture exams toward your final grade? quizzes writing assignments participation presentation
New aspects for bipolar plates and gaskets for fuel cells and redox flow batteries
 New aspects for bipolar plates and gaskets for fuel cells and redox flow batteries Dr. Rouven Henkel Eisenhuth GmbH & Co. KG D - 37520 Osterode GERMANY Product range injection molding 3 main divisions
New aspects for bipolar plates and gaskets for fuel cells and redox flow batteries Dr. Rouven Henkel Eisenhuth GmbH & Co. KG D - 37520 Osterode GERMANY Product range injection molding 3 main divisions
3. Describe the study in mimicry, using king snakes and coral snakes. Identify the control in the experiment.
 Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
Unit 2 Notes: Cells. What you need to know:
 1 Unit 2 Notes: Cells What you need to know: 1. MC.2.B.1: Construct a hierarchy of life from cells to ecosystems. (ex: cell, tissue, organ etc) 2. NS.12.B.4: Relate the development of the cell theory to
1 Unit 2 Notes: Cells What you need to know: 1. MC.2.B.1: Construct a hierarchy of life from cells to ecosystems. (ex: cell, tissue, organ etc) 2. NS.12.B.4: Relate the development of the cell theory to
Permeation of organic molecules in water and ethanol-water solutions by reverse osmosis
 Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-20 September 2007 Permeation of organic molecules in water and ethanol-water solutions by reverse osmosis J. Labanda, J.
Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-20 September 2007 Permeation of organic molecules in water and ethanol-water solutions by reverse osmosis J. Labanda, J.
What are the parts of a eukaryotic cell? What is the function of each part of a eukaryotic cell?
 CHAPTER 3 SECTION 2 Cells: The Basic Units of Life Eukaryotic Cells BEFORE YOU READ After you read this section, you should be able to answer these questions: What are the parts of a eukaryotic cell? What
CHAPTER 3 SECTION 2 Cells: The Basic Units of Life Eukaryotic Cells BEFORE YOU READ After you read this section, you should be able to answer these questions: What are the parts of a eukaryotic cell? What
Extraction News Notes Blog
 Extraction News Notes Blog www.infinitysupercritical.com BY: Infinity Supercritical Staff EG TAGS: Supercritical CO2 Oil Extraction, Cannabis, Oil Concentrates Sustainable Production of Cannabinoids with
Extraction News Notes Blog www.infinitysupercritical.com BY: Infinity Supercritical Staff EG TAGS: Supercritical CO2 Oil Extraction, Cannabis, Oil Concentrates Sustainable Production of Cannabinoids with
