Discovery and development of BNC105P, a novel Vascular Disruption Agent exhibiting a superior therapeutic margin
|
|
|
- Eustace Cobb
- 5 years ago
- Views:
Transcription
1 Discovery and development of BNC105P, a novel Vascular Disruption Agent exhibiting a superior therapeutic margin Gabriel Kremmidiotis, Tina Lavranos, Annabell Leske, Donna Beaumont, Jelena Gasic, Allison Hall, Michael O'callaghan, Gurmit Gill, Damien Grobelny, Bernard Flynn. ABSTRACT Vascular Disruption Agents (VDA) target endothelial cells lining blood vessels in solid tumors, causing occlusion of tumor vasculature and tumor cell necrosis due to hypoxia. Tumor vascular disruption is a therapeutic strategy of great potential, however, VDA compounds currently under development display a narrow therapeutic margin between efficacy and toxicity, with cardiovascular toxicity posing as the major dose limiting obstacle. Discovery of new VDA compounds that display a wide therapeutic margin will enable us to fully harness the therapeutic potential of blocking tumor vasculature. In order to identify such compounds we used a novel in vitro screening approach that exploits the different activation states displayed by endothelial cells stimulated by growth factors found in a solid tumor milieu versus endothelial cells that are present in normal tissues and are deprived of such growth factors. Our effort has yielded the compound BNC105. This compound displays 100-fold higher potency against endothelial cells that are actively proliferating or are engaged in angiogenic processes as compared to non-proliferating endothelial cells or endothelium found in stable capillaries. This selectivity profile was not seen with other tubulin polymerisation inhibitors currently in clinical development as VDAs. BNC105 disrupts the vasculature in human breast and colon solid tumors grown in nude mice but does not affect the vasculature in normal organs. One dose of 1mg/kg BNC105 administered iv disrupted 50% of tumor vasculature. Animals treated with 80mg/kg BNC105 did not exhibit any signs of toxicity, demonstrating a therapeutic margin of 80. A single 10mg/kg iv dose of BNC105 disrupted more than 95% of tumor vasculature in breast and colon cancer models. With repeated dosing this resulted in up to 75% tumor growth inhibition. With repeat dosing 40 mg/kg iv BNC105 caused 52% tumor regression and prolongation of survival time. Fourteen percent of tumor bearing animals were cleared of their tumor burden following 2 cycles of BNC105 treatment. BNC105 exhibits pharmacokinetic and pharmacodynamic profiles that justify commencement of clinical development. Bionomics Limited, 31 Dalgleish Street, Thebarton, 5031 South Australia, Australia.
2 INTRODUCTION The disruption of blood vessels that feed tumors represents one of the most promising therapeutic strategies for treating cancer. Until recently, the majority of research underlying this concept has focused on the discovery of agents that inhibit formation of new blood vessels in tumors, a process known as tumor angiogenesis. However, more recently, the importance of disrupting existing tumor vessels has been recognised and research efforts have expanded to the discovery of anti-vascular agents that cause shutdown of tumor vasculature, leading to extensive tumor death (Pilat et al 2004; Cooney et al 2005). Microtubules are filamentous intracellular structures that play a critical role in the function of cells. They act as a scaffold that determines cell shape and provides a network of tracks for intracellular vesicles and organelles to move on. Furthermore, microtubules are responsible for the formation of the mitotic spindle, a network of fibres that forms during cell division and guides the separation of chromosomes (Desai & Mitchison 1997; Nogales 2000). Compounds that target microtubule function by binding to of the protein tubulin have yielded very encouraging results in preclinical animal studies. These compounds, referred to as Vascular Disruption Agents (VDAs), elicit their anti-vascular effect through disruption of the cytoskeleton resulting in endothelial cell swelling (Siemann et al 2005; Tozer et al 2005). The swelling of endothelial cells can result in blood vessel occlusion and shutdown of the blood circulation. This effect is particularly pronounced in tumors. Certain aberrant features make tumor vasculature more susceptible to the cell swelling effects induced by VDAs. Despite encouraging results obtained in animal models the development of tubulin polymerisation inhibitors as vascular disruption agents in the clinic has been very slow, possibly due to toxicity issues associated with disruption of normal endothelial cells. Human endothelial cells may be more susceptible to the in vivo action of VDAs than endothelial cells in experimental animals. There is a clear need for the development of improved VDA compounds that are able to distinguish between tumor and normal endothelial cells. It is reasonable to hypothesise that compounds that exhibit selectivity for tumor vasculature may further enhance the specificity of VDA action. This is likely to alleviate some of the toxicity issues seen in the clinic to-date and result in significant increases in therapeutic index. The ability to influence the cell shape of actively proliferating versus non-proliferating endothelial cells in culture has been one of the most commonly used selection criteria for the identification of tubulin polymerisation inhibitors that have the ability to act as selective disruptors of tumor vasculature. Representative examples of this approach are the published work of Davis et al (2002) on ZD6126 and Dark et al (1997) on CA4P. We have utilised an in vitro system that allows comparative investigation of activated (tumor) versus quiescent (normal) endothelial cells as defined by endothelial cell proliferation under the influence of growth factors and the ability of endothelial cells to engage in the formation and maintenance of capillary tubes in vitro. We utilised these in vitro assays to evaluate a library of over 100 compounds exhibiting tubulin polymerisation inhibition (TPI) activity. The aim of this undertaking was to identify compounds that exhibit higher activity for activated endothelial cells than quiescent endothelial cells. It is reasonable to anticipate that such compounds will exhibit selectivity for the endothelium in tumors, sparing endothelium in normal tissues.
3 MATERIALS & METHODS Proliferation assays were carried out in triplicate in 96-well plates using an MTT assay. Absorbance readings for each compound concentration were normalised to no-compound control cultures. For capillary formation assays cells were plated on Matrigel at 25,000/well in 96-well plates and cultured for 22hrs. In vitro tubulin polymerisation assays utilised bovine neuronal tubulin that was allowed to polymerise in vitro in the presence of different concentrations of BNC105 or CA4. Fluorescence measurements were obtained over a period of 42 minutes at 1 minute intervals. Endothelial cell monolayer permeability was assessed using an in vitro Vascular Permeability Assay kit (ECM640, Chemicon) as previously described (Watts et al, 1997). All in vitro assays were carried out using endothelial cells derived from human umbilical vein (HUVEC) or human abdominal aorta (HAAEC). Primary in vivo evaluations of BNC105P efficacy were carried out using either an orthotopic human breast cancer model or a subcutaneous colon cancer model. The breast model involves the injection of MDA-MB-231 human breast cancer cells under the mammary fat pad of female Balb/c nu/nu mice; the colon model involves the injection of Colo205 cells subcutaneously on the right hind flank of female Balb/c nu/nu mice. The cells are allowed to grow for 2-3 weeks forming a solid tumor of approximately mm 3 in size. BNC105 has low solubility. For in vivo administration we synthesised BNC105 in a disodium phosphate prodrug chemical arrangement, BNC105P. BNC105P is completely soluble in saline and is rapidly converted into the BNC105 drug in vivo. BNC105P solutions were administered by single bolus intravenous injections through the tail vein. RESULTS & DISCUSSION Identification of BNC105, a compound that exhibits high activity against proliferating endothelial cells and comparatively low activity against quiescent endothelial cells. We have constructed a library of TPI compounds based on a benzofuran ring chemical core. We evaluated each of these compounds for its activity on human endothelial cells cultured in the presence or absence of growth factors that promote endothelial cell proliferation (VEGF, hfgf-b, IGF-1, hegf, heparin). In the presence of these growth factors, cells were seeded at a density that allows further growth whereas, in the absence of growth factors cells were seeded at higher densities in order to promote survival and emulate close cell-cell contact interactions that are expected to be in operation between quiescent endothelial cells lining normal blood vessels. Our analyses identified a small number of compounds that exhibit selectivity for activated endothelial cells (Fig. 1). Compound BNC105 exhibited the highest degree of selectivity being 100 times more active against activated endothelial cells than quiescent endothelial cells (Figs. 1, 2). This selectivity was not seen with other TPI drugs currently in clinical trials, namely CA4 (Fig. 2) or ABT751 (Fig. 2). Furthermore, BNC105 exhibited significantly higher potency than CA4 and ABT751 against proliferating endothelial cells. In addition, to studying HUVEC we also investigated the activity of BNC105 against activated and quiescent HAAEC cells. As seen for HUVEC, BNC105 exhibited 100-fold selectivity for activated HAAEC cells (data not shown). These findings demonstrate that the selectivity displayed by BNC105 for activated endothelial cells is not restricted to HUVEC.
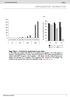
4 Ratio IC50 Quiescent / Growth-Factor Activated Endothelial cells BNC105 CA4 Figure 1. Activity exhibited by Bionomics TPI compounds on HUVEC. HUVEC were cultured either in the presence (activated) or absence (quiescent) of growth factors. Activated and quiescent cell cultures were exposed to a range of concentrations for each of the compounds tested (0.1nM -1000nM). Following a 48hr incubation period cell proliferation was measured using an MTT assay (absorbance readings - 492nm). Absorbance readings were normalised to no-compound control cultures corresponding to activated and quiescent cells respectively. Normalised values were used to derive IC50 values for each compound for activated and quiescent cells. The graph in this figure displays the IC50-quiescent/IC50-activated ratio for each of the compounds tested. BNC105 (compound 0083 shown in Blue) exhibited a ratio of over 100. The compound CA4 (shown in red) was used as a comparator.
5 Activity displayed by BNC105 against activated and quiescent endothelial cells (Control normalised ratio) Activity displayed by CA4 against activated and quiescent endothelial cells (Control normalised ratio) Activated Quiescent Activated Quiescent (Log 10) M (Log 10) M EC50 Activated e-010 Quiescent e-008 EC50 Activated e-009 Quiescent e-009 Activity displayed by ABT751 against activated and quiescent endothelial cells (Control normalised ratio) Activated Quiescent EC50 Activated e-007 (log10) M Quiescent e-007 Figure 2. BNC105 exhibits 100-fold higher activity against activated HUVEC. HUVEC were cultured either in the presence (activated) or absence (quiescent) of growth factors. Activated and quiescent cell cultures were exposed to a range of concentrations of BNC105, CA4 and ABT751. Following a 48hr incubation period cell proliferation was measured using an MTT assay (absorbance readings - 492nm). Absorbance readings for each drug concentration were normalised to vehicle-treated control cultures corresponding to activated and quiescent cells respectively. The graphs in this figure display the activity of BNC105, CA4 and ABT751 against activated and quiescent HUVEC in the concentration range 0.1nM-1000nM.
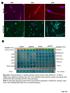
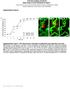
6 BNC105 disrupts the formation of endothelial capillaries but does not disrupt preformed/quiescent capillaries. We investigated the activity of BNC105 on endothelial cells engaged in capillary tube formation in vitro. Endothelial cells were placed on a Matrigel matrix and allowed to form capillary tubes in the presence or absence of the compound under evaluation. Each compound tested was used at concentrations found to display activity in the proliferation assay. BNC105 inhibited the formation of capillary tubes at 1nM (Fig.3). This activity was considerably higher than CA4 which inhibited capillary formation at 10nM and ABT751 that failed to inhibit capillary formation at 100nM (Fig. 3). These results clearly demonstrate that BNC105 exhibits very high potency against activated endothelial cells engaged in formation of capillaries. Subsequently, we investigated the activity of BNC105 on preformed capillaries. Endothelial cells were plated on Matrigel and allowed to form capillary tubes for 22hrs. Subsequently, these capillary tubes were exposed to each of the compounds under study. Our results indicate that although BNC105 inhibited the formation of capillaries at 1nM (Fig. 3), it failed to disrupt preformed capillaries at that concentration (Fig. 4). This result is in agreement with the proliferation assay results providing further proof that this compound exhibits selectivity for activated endothelial cells. In contrast, CA4 disrupted preformed capillary tubes at the 10nM concentration (Fig. 4), at this concentration it also inhibited the formation of capillary tubes (Fig. 3). BNC105 alters endothelial cell morphology and increases the permeability of endothelial cell monolayers. It has been previously shown that CA4 induces cytoskeletal alterations that manifest in a cell blebbing morphology in which actin accumulates around surface blebs (Kanthou & Tozer, 2002). We investigated whether BNC105 induces similar morphological changes. Exposure of HUVEC to 10nM of BNC105 for 15 minutes resulted in cell blebbing (Figure 5A). Drugs interfering with the morphology of endothelial cells compromise the integrity of endothelial cell layers lining blood vessels resulting in increased permeability. The consequences of the BNC105-induced cell morphology changes to endothelial cell permeability were evaluated. HUVEC monolayers were grown to confluency on microporous membranes. The ability of fluorescent dextran to cross the HUVEC monolayer in the presence of different concentrations of BNC105 was assessed. CA4 was used as comparator. This in vitro assay was carried out as previously described (Watts et al, 1997). Based on the effect of BNC105 on the cytoskeletal integrity of HUVEC it was not surprising to see that this drug led to an increase in the permeability of a HUVEC monolayer. Interestingly, CA4 was marginally more potent than BNC105 in this assay (Fig. 5B). This finding provides further support to the notion of BNC105 selectivity. Due to contact inhibition, endothelial cell monolayers in this assay comprise quiescent cells. BNC105 is less potent against these quiescent cells than CA4. In contrast, BNC105 was found to be 10-fold more potent than CA4 against endothelial cells that are either proliferating actively or are engaged in the formation of capillary tubes (Fig.2,3).
7 Figure 3. BNC105 inhibits HUVEC capillary formation in vitro. Cells were plated at 25,000/well in 96-well plates and cultured for 22hrs. The inhibitory activity exhibited by the compounds BNC105, CA4 and ABT751 on the formation of capillary tubes by HUVEC on Matrigel was evaluated. DMSO vehicle negative control was included in the analysis. Figure 4. BNC105 does not disrupt preformed HUVEC capillaries in vitro. The inhibitory activity exhibited by the compounds BNC105 and CA4 on HUVEC capillary tubes formed on Matrigel was evaluated. Pre-formed capillaries were exposed to BNC105 and CA4 concentrations that were found to inhibit capillary formation (Fig. 3). Capillary integrity was examined 5hrs after the addition of the compounds under study. This figure shows pictures of HUVEC capillaries immediately after addition of BNC105 and CA4 and 5hrs later. Capillaries were also stained with Calcein to confirm cell viability.
8 CONTROL 5000 Permeability assay 4000 BNC105 CA BNC105 Blebs A B Figure 5. Effect of BNC105 on endothelial cell morphology (A) and permeability (B) in vitro. (A) HUVEC were cultured in the presence of 10nM BNC105 for 15min. Cells were stained for actin. BNC105 treated cells displayed clear evidence of blebbing. (B) HUVEC monolayer permeability was assessed using an in vitro Vascular Permeability Assay kit (ECM640, Chemicon). HUVEC were seeded on a semi-permeable membrane and allowed to form a confluent monolayer. The HUVEC monolayer was treated with either BNC105, CA4 or vehicle control for 15min. The culture medium was replaced with an FITC-Dextran solution and incubated for 5 minutes. The extent of permeability was measured in terms of fluorescence allowed to pass through the cell monolayer.
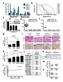
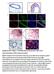
9 Synthesis of a BNC105 prodrug - BNC105P. BNC105 does not have sufficient solubility to allow intravenous administration for future clinical use. In order to accommodate for this we synthesised BNC105P, a disodium phosphate ester form of BNC105. BNC105P is soluble in saline and is rapidly converted to the active drug BNC105 in vivo through the action of non-specific phosphatases. BNC105P disrupts vascular perfusion in human breast cancer xenografts. In order to further substantiate the anti-vascular action of BNC105P we investigated its ability to preferentially shut down the vasculature in solid tumors. Mice carrying solid breast tumors were treated with a single intravenous injection of either BNC105P (1mg/kg 150mg/kg) or CA4P (5mg/kg - 400mg/kg). Twenty four hours after the injection of the compounds the animals were injected with the fluorescent dye H H33342 acts as a marker of blood perfusion in tumors and normal tissues. Following the injection of the dye the animals were euthanized, the tumors were excised for histological examination. Quantitative analysis of H33342 staining in tumor sections revealed that BNC105P treatment caused ~50% vascular shutdown at 1 mg/kg and almost complete vascular shutdown at 10 mg/kg (Fig. 6). In comparison, CA4P caused ~50% vascular shut down at 100 mg/kg (Fig. 6). Interestingly, BNC105P was tolerated without any signs of toxicity at 80 mg/kg and CA4P at 300 mg/kg. These values translate into a TI of 80 for BNC105P and a TI of only 3 for CA4P. Qualitative examination of the data revealed that BNC105P caused dramatic vascular disruption within solid tumors at a dose of 10mg/kg (Fig. 7A). Staining of tumor sections with H&E showed that the vascular shutdown caused by BNC105P resulted in the initiation of necrosis in the core of the tumor (Fig. 7B). Investigation of vascular disruption achieved by a single iv dose of 10 mg/kg BNC105P at shorter time intervals revealed that BNC105P causes 98% vascular disruption as early as 3 hours post-administration and remained above 90% after 24hrs. Increased tumor necrosis became evident 24hrs after administration of BNC105P (data not shown). Quantitative analysis of vascular perfusion in normal organs of animals treated with BNC105P at the MTD (80mg/kg) revealed no changes compared to the organs of vehicle treated animals (Fig. 8). BNC105P disrupts vascular perfusion in human colon cancer xenografts. Following the investigation of BNC105P efficacy in a solid breast cancer model we sought to investigate the efficacy of BNC105P treatment on tumor vasculature in a colon cancer model. For BNC105P treated groups, doses of 10mg/kg and above showed a statistically significant decrease in tumor perfusion (Fig.9A) and a corresponding increase in tumor necrosis (Fig. 9B) compared with the saline treated group. In the CA4P treated groups there was no significant difference in vascular perfusion between any of the CA4P doses tested and the saline treated group (Fig. 9A). There was a trend towards an increase in necrosis in a dose dependent manner in CA4P treated tumors but this was not statistically significant (Fig. 9B).
10 MTD* MTD* TI=80 TI=3 *MTD - no observable signs of toxicity Figure 6. Tumor vascular disruption caused by treatment with different doses of BNC105P and CA4P. Balb/c nu/nu mice bearing MDA-MB-231 solid tumors were treated with a single iv injection of BNC105P, CA4P or saline. BNC105P was injected at concentrations ranging at 1mg/kg-150mg/kg. CA4P was injected in the range of 5mg/kg-400mg/kg. 24hrs following this treatment the animals were injected with H33342 to label tumor vascular perfusion. Tumors were excised, sectioned and visualised for H33342 staining. H33342 staining was quantified using Image analysis software.
11 A B Figure 7. BNC105P disrupts the vasculature in solid tumors. Balb/c nu/nu mice bearing MDA-MB-231 solid tumors were treated with a single iv injection of 10mg/kg BNC105P or saline. 24hrs following this treatment the animals were injected with H33342 to label tumor vascular perfusion. Tumors were excised, sectioned and visualised for H33342 staining (A). Tumor sections were subsequently stained with H&E to detect necrotic cancer cells. H33342 staining (black) was superimposed to the H&E stain(b) Heart Kidney Liver Spleen Saline BNC105P Saline BNC105P 24 hours 72 hours Figure 8. Effect of BNC105P treatment on the vascular perfusion in normal tissues. Balb/c nu/nu mice bearing MDA-MB-231 solid tumors were treated with a single iv injection of 80mg/kg BNC105P or saline. 24 and 72hrs following treatment the animals were injected with H33342 to label vascular perfusion within tumors and normal organs. A number of normal organs were excised, sectioned and visualised for H33342 staining.
12 BNC105P Tumor perfusion (Colo205) BNC105P Tumor necrosis (Colo205) ** ** ** ** * =P< * * * 0 ** =P< mg/kg 20mg/kg 10mg/kg 5mg/kg Saline 80mg/kg 20mg/kg 10mg/kg 5mg/kg Saline BNC105P Dose Treatment BNC105P CA4P Tumor perfusion (Colo205) CA4P Tumor Necrosis (Colo205) A 400mg/kg 200mg/kg 150mg/kg 100mg/kg Saline Treatment CA4P 400mg/kg 200mg/kg 150mg/kg 100mg/kg Saline Treatment CA4P Figure 9. (A) Analysis of vascular perfusion in human colon tumor xenografts treated with BNC105P or CA4P. (B) Analysis of necrosis in human colon tumor xenografts treated with BNC105P or CA4P. Balb/c nu/nu mice bearing subcutaneous Colo205 tumors were treated with a single iv injection of saline, BNC105P, or CA4P at various concentrations. 24hrs following this treatment the animals were injected with H33342 to detect tumor vascular perfusion. Tumors were excised, sectioned and visualised for H33342 staining and were subsequently stained with H&E to visualise necrotic tumor areas. Percentage perfusion and necrosis is shown for each individual animal per group, staining was quantified using Image analysis software. Statistical analysis on perfusion and necrosis data utilized one-way ANOVA and Dunnett s multiple comparison post-test using Prism Software. B
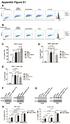
13 Two cycles of BNC105P treatment suppresses tumor growth in a breast cancer model. Balb/c nu/nu mice bearing MDA- MB-231 orthotopic breast solid tumors were treated with two cycles of BNC105P at 40mg/kg. Tumor growth as well as animal health were monitored for up to 72 days post-day 1 of treatment. The results seen in this experiment (Fig. 11) clearly show tumor growth inhibition in animals treated with two cycles of BNC105P. Significant differences in tumor growth between BNC105P treated (n=64) and saline treated (n=20) animals were observed as early as day 4 (p<0.001; unpaired t-test; Prism analysis) through to Day 70. In the BNC105P treated group, 34% of tumors showed delayed/slow growth, 4.7% exhibited no growth, 6.3% regressed and 14% of tumors were cleared. Fig. 10 depicts an example of one Balb/c nu/nu mouse from a total of 14% bearing a solid tumor in which tumor growth was arrested and the tumor disappeared over approximately 20 days following a second cycle of BNC105P treatment. There is a clear progressive drop in tumor volume following the second cycle of BNC105P treatment with the animal being cleared of its tumor burden by day 49. Cleared tumors did not reappear for the duration of this experiment. Figure 10. Example of one Balb/c nu/nu mouse from a total of 14% bearing a MDA-MB-231 solid tumor and treated with BNC105P at 40mg/kg iv 2 doses/1wk apart/3 wks rest, in which tumor growth was arrested and the tumor disappeared over 20 days following a second cycle of treatment.
14 Evaluation of BNC105P Treatment in Tumor Growth Inhibition (Tumor volume ratio +/- SD) Saline BNC105P Figure 11. BNC105P tumor growth inhibition. Balb/c nu/nu mice bearing MDA-MB-231 solid tumors were treated with BNC105P. Animals were iv dosed with a total of two cycles of BNC105P treatment (dose timepoints shown with blue arrows). Tumor growth represented as a ratio to initial tumor volume is shown over a total of 72-days. Significant differences in tumor growth between BNC105P treated (n=64) and saline treated (n=20) animals were observed as early as day 4 (p<0.001; unpaired t-test; Prism analysis) through to Day 49 at which timepoint the vehicle treated animals had to be euthanized due to the very large size of their tumors Day
15 RESULTS & DISCUSSION BNC105P is a very potent Vascular Disruption Agent BNC105 exhibits selectivity for activated endothelium in two independent in vitro assays BNC105P is considerably more potent than CA4P in causing tumor vascular disruption, achieving a significantly improved Therapeutic Index BNC105P disrupts blood vessels in human breast and colon cancer in vivo tumor models BNC105P shows single agent efficacy in breast cancer with a 14% cure rate REFERENCES Cooney MM, Ortiz J, Bukowski RM, Remick SC. Novel vascular targeting/disrupting agents: Combretastatin A4 phosphate and related compounds. Curr Oncol Rep : Dark GG, Hill SA, Prise VE, Tozer GM, Pettit GR, Chaplin DJ. Combretastatin A-4, an agent that displays potent and selective toxicity toward tumor vasculature. Cancer Res May 15;57(10): Davis PD, Dougherty GJ, Blakey DC, Galbraith SM, Tozer GM, Holder AL, Naylor MA, Nolan J, Stratford MR, Chaplin DJ, Hill SA. ZD6126: a novel vascular-targeting agent that causes selective destruction of tumor vasculature. Cancer Res Dec 15;62(24): Desai A, Mitchison TJ. Microtubule polymerization dynamics. Annu Rev Cell Dev Biol : Kanthou C, Tozer GM. The tumor vascular targeting agent combretastatin A-4-phosphate induces reorganization of the actin cytoskeleton and early membrane blebbing in human endothelial cells. Blood Mar 15;99(6): Nogales E. Structural insights into microtubule function. Annu Rev Biochem : Pilat MJ, McCormick J, LoLusso PM. Vascular targeting agents. Curr. Oncol. Rep : Prinz H. Recent advances in the field of tubulin polymerization inhibitors. Expert Rev. Anticancer Ther (6): Siemann DW, Bibby MC, Dark GG, Dicker AP, Eskens FALM, Horsman MR, Marme D, LoLusso PM. Differentiation and definition of vascular-targeted therapies. Clin. Cancer res : Tozer GM, Kanthou C, Bagulegy BC. Disrupting tumor blood vessels. Nature : Watts ME, Woodcock M, Arnold S, Chaplin DJ. Effects of novel and conventional anti-cancer agents on human endothelial permeability: influence of tumor secreted factors. Anticancer Res Jan-Feb;17(1A):71-5. ABBREVIATIONS HUVEC - Human umbilical vein endothelial cells TPI - Tubulin Polymerisation Inhibitor HAAEC - Human abdominal aorta endothelial cells TI - Therapeutic Index
Preclinical Studies of the Novel Vascular Disrupting Agent MN-029
 Preclinical Studies of the Novel Vascular Disrupting Agent MN-029 WENYIN SHI and DIETMAR W. SIEMANN Department of Radiation Oncology, Shands Cancer Center, University of Florida, Gainesville, FL 32610,
Preclinical Studies of the Novel Vascular Disrupting Agent MN-029 WENYIN SHI and DIETMAR W. SIEMANN Department of Radiation Oncology, Shands Cancer Center, University of Florida, Gainesville, FL 32610,
CANCER THERAPEUTICS: A NOVEL APPROACH
 CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
CANCER THERAPEUTICS: A NOVEL APPROACH Mary Dwyer, Ph.D. HBRI and ChemRegen, Inc. SCDMDG Meeting October 23, 212 Outline Introduction Hit, HBRI1: identification & characterization Leads, HBRI2 & HBRI3:
A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified
 Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin
 Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like
 SUPPLEMENTL FIGURE LEGENDS Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like spheroids from a human brain tumor sample or glioma xenograft. () CD133+ tumor cells isolated
SUPPLEMENTL FIGURE LEGENDS Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like spheroids from a human brain tumor sample or glioma xenograft. () CD133+ tumor cells isolated
TITLE: Notch in Pathological Angiogenesis and Lymphangiogenesis
 Award Number: W81XWH-10-1-0304 TITLE: Notch in Pathological Angiogenesis and Lymphangiogenesis PRINCIPAL INVESTIGATOR: Minji Kim CONTRACTING ORGANIZATION: Columbia University New York, NY 10032 REPORT
Award Number: W81XWH-10-1-0304 TITLE: Notch in Pathological Angiogenesis and Lymphangiogenesis PRINCIPAL INVESTIGATOR: Minji Kim CONTRACTING ORGANIZATION: Columbia University New York, NY 10032 REPORT
Related information Actions Suggestions
 QuickSearch: Results Summary > Drug Report Drug lexibulin (iv infusion, cancer), YM BioSciences Company Highest Dev Status Indications Actions Technologies Cytopia Ltd Phase 2 Clinical Multiple myeloma
QuickSearch: Results Summary > Drug Report Drug lexibulin (iv infusion, cancer), YM BioSciences Company Highest Dev Status Indications Actions Technologies Cytopia Ltd Phase 2 Clinical Multiple myeloma
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
SUPPLEMENTARY FIGURES AND TABLES
 SUPPLEMENTARY FIGURES AND TABLES Supplementary Figure S1: CaSR expression in neuroblastoma models. A. Proteins were isolated from three neuroblastoma cell lines and from the liver metastasis of a MYCN-non
SUPPLEMENTARY FIGURES AND TABLES Supplementary Figure S1: CaSR expression in neuroblastoma models. A. Proteins were isolated from three neuroblastoma cell lines and from the liver metastasis of a MYCN-non
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center
 Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Trevigen s Extracellular Matrix Proteins for Triculture of Breast Cancer Spheroids, Endothelial Tubules, and Human Mesenchymal Stem Cells
 Trevigen s Extracellular Matrix Proteins for Triculture of Breast Cancer Spheroids, Endothelial Tubules, and Human Mesenchymal Stem Cells Gabriel Benton, Ph.D. and Jay George, Ph.D., Trevigen, Inc. Gaithersburg,
Trevigen s Extracellular Matrix Proteins for Triculture of Breast Cancer Spheroids, Endothelial Tubules, and Human Mesenchymal Stem Cells Gabriel Benton, Ph.D. and Jay George, Ph.D., Trevigen, Inc. Gaithersburg,
Lankenau Institute for Medical Research Annual Progress Report: 2010 Formula Grant
 Lankenau Institute for Medical Research Annual Progress Report: 21 Formula Grant Reporting Period July 1, 211 December 31, 211 Formula Grant Overview The Lankenau Institute for Medical Research received
Lankenau Institute for Medical Research Annual Progress Report: 21 Formula Grant Reporting Period July 1, 211 December 31, 211 Formula Grant Overview The Lankenau Institute for Medical Research received
Induction of Solid Mammary, Subcutaneous and Lung Cancers in Rats and Mice. To investigate rodent models of cancer
 THE UNIVERSITY OF QUEENSLAND Page 1 of 6 SOP No: AHP 30 DATE ISSUED: 29/09/2017 SUBJECT: REASON FOR USE: Induction of Solid Mammary, Subcutaneous and Lung Cancers in Rats and Mice To investigate rodent
THE UNIVERSITY OF QUEENSLAND Page 1 of 6 SOP No: AHP 30 DATE ISSUED: 29/09/2017 SUBJECT: REASON FOR USE: Induction of Solid Mammary, Subcutaneous and Lung Cancers in Rats and Mice To investigate rodent
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH- TITLE: PRINCIPAL INVESTIGATOR: CONTRACTING ORGANIZATION: REPORT DATE: TYPE OF REPORT: Annual PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
AD Award Number: W81XWH- TITLE: PRINCIPAL INVESTIGATOR: CONTRACTING ORGANIZATION: REPORT DATE: TYPE OF REPORT: Annual PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
SHREE ET AL, SUPPLEMENTAL MATERIALS. (A) Workflow for tumor cell line derivation and orthotopic implantation.
 SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
(PDGF), 9 ( -2 (FGF-2), SMO
 Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG)
 In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG) 1 Dr Saeb Aliwaini 13/11/2015 Migration in vivo Primary tumors are responsible for only about 10%
In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG) 1 Dr Saeb Aliwaini 13/11/2015 Migration in vivo Primary tumors are responsible for only about 10%
Dual Targeting Nanoparticle Stimulates the Immune
 Dual Targeting Nanoparticle Stimulates the Immune System to Inhibit Tumor Growth Alyssa K. Kosmides, John-William Sidhom, Andrew Fraser, Catherine A. Bessell, Jonathan P. Schneck * Supplemental Figure
Dual Targeting Nanoparticle Stimulates the Immune System to Inhibit Tumor Growth Alyssa K. Kosmides, John-William Sidhom, Andrew Fraser, Catherine A. Bessell, Jonathan P. Schneck * Supplemental Figure
Specific Targeting and Delivery of Therapeutics to Cancer Cells Based on the Tumor Microenvironment
 Specific Targeting and Delivery of Therapeutics to Cancer Cells Based on the Tumor Microenvironment Damien Thévenin Department of Chemistry Bioscience in the 21st century (Sep. 14, 218) Anticancer Drugs
Specific Targeting and Delivery of Therapeutics to Cancer Cells Based on the Tumor Microenvironment Damien Thévenin Department of Chemistry Bioscience in the 21st century (Sep. 14, 218) Anticancer Drugs
Developing Best in Class BET Inhibitors for Oncology & AI: from Discovery to the Clinic. Kevin G. McLure, PhD EpiCongress July 2014
 Developing Best in Class BET Inhibitors for Oncology & AI: from Discovery to the Clinic Kevin G. McLure, PhD EpiCongress July 2014 Zenith Epigenetics Overview Formed from Resverlogix as an independent
Developing Best in Class BET Inhibitors for Oncology & AI: from Discovery to the Clinic Kevin G. McLure, PhD EpiCongress July 2014 Zenith Epigenetics Overview Formed from Resverlogix as an independent
CONTRACTING ORGANIZATION: University of Colorado Health Sciences Aurora, CO 80045
 AD Award Number: W81XWH-05-1-0464 TITLE: Muscle contraction arrests tumor growth PRINCIPAL INVESTIGATOR: Kim C. Westerlind, Ph.D. CONTRACTING ORGANIZATION: University of Colorado Health Sciences Aurora,
AD Award Number: W81XWH-05-1-0464 TITLE: Muscle contraction arrests tumor growth PRINCIPAL INVESTIGATOR: Kim C. Westerlind, Ph.D. CONTRACTING ORGANIZATION: University of Colorado Health Sciences Aurora,
B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small cell lung cancer
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
Reduction of metastatic and angiogenic potency of malignant cancer by Eupatorium. fortunei via suppression of MMP-9 activity and VEGF production
 Supplementary Information Reduction of metastatic and angiogenic potency of malignant cancer by Eupatorium fortunei via suppression of MMP-9 activity and VEGF production Aeyung Kim, Minju Im, Nam-Hui Yim
Supplementary Information Reduction of metastatic and angiogenic potency of malignant cancer by Eupatorium fortunei via suppression of MMP-9 activity and VEGF production Aeyung Kim, Minju Im, Nam-Hui Yim
TITLE: Role of ADAM15 in Tumor/Endothelial Interactions Prostate Cancer Regression
 AD Award Number: W81XWH-07-1-0030 TITLE: Role of ADAM15 in Tumor/Endothelial Interactions Prostate Cancer Regression PRINCIPAL INVESTIGATOR: Mark L. Day CONTRACTING ORGANIZATION: University of Michigan
AD Award Number: W81XWH-07-1-0030 TITLE: Role of ADAM15 in Tumor/Endothelial Interactions Prostate Cancer Regression PRINCIPAL INVESTIGATOR: Mark L. Day CONTRACTING ORGANIZATION: University of Michigan
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Catechin s anti-angiogenic effects in epithelial ovarian cancer
 Catechin s anti-angiogenic effects in epithelial ovarian cancer Brian Krug Background Epithelial ovarian cancer (EOC) is a common and lethal malignancy of the female reproductive tract (2). Often detected
Catechin s anti-angiogenic effects in epithelial ovarian cancer Brian Krug Background Epithelial ovarian cancer (EOC) is a common and lethal malignancy of the female reproductive tract (2). Often detected
Endothelial Tube Formation Assay (In Vitro Angiogenesis)
 Product Manual Endothelial Tube Formation Assay (In Vitro Angiogenesis) Catalog Number CBA-200 50 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Angiogenesis, or neovascularization,
Product Manual Endothelial Tube Formation Assay (In Vitro Angiogenesis) Catalog Number CBA-200 50 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Angiogenesis, or neovascularization,
Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF
 Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF- 02341066 Assay IC 50 nm Selectivity Ratio d Biochemical Activity In Vitro c-met/hgfr enzyme (Ki, nm) a 4 NA Cellular Activity
Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF- 02341066 Assay IC 50 nm Selectivity Ratio d Biochemical Activity In Vitro c-met/hgfr enzyme (Ki, nm) a 4 NA Cellular Activity
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
Discovery of Antitubulin Agents with Antiangiogenic Activity as Single Entities with Multitarget Chemotherapy Potential
 This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acsmedchemlett
This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acsmedchemlett
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were
 Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were
 Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
ENMD-2076, an Oral Aurora A and Angiogenesis Kinase Inhibitor
 ENMD-2076, an Oral Aurora A and Angiogenesis Kinase Inhibitor Dr. Mark R. Bray VP Research, EntreMed, Inc. AACR Annual Meeting New Drugs on the Horizon 2 April 2008 1 Disclosure Information: AACR New Drugs
ENMD-2076, an Oral Aurora A and Angiogenesis Kinase Inhibitor Dr. Mark R. Bray VP Research, EntreMed, Inc. AACR Annual Meeting New Drugs on the Horizon 2 April 2008 1 Disclosure Information: AACR New Drugs
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Antitumor Activity of CUDC-305, a Novel Oral HSP90 Inhibitor, in Solid and Hematological Tumor Xenograft Models
 Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Supplementary Information
 Supplementary Information Lymphatic endothelial cells support tumor growth in breast cancer Esak Lee a, Niranjan B. Pandey a, and Aleksander S. Popel a,b a Department of Biomedical Engineering, Johns Hopkins
Supplementary Information Lymphatic endothelial cells support tumor growth in breast cancer Esak Lee a, Niranjan B. Pandey a, and Aleksander S. Popel a,b a Department of Biomedical Engineering, Johns Hopkins
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells
 CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
RIP-Tag2 mouse model as a Paradigm for Target. Search in NETs
 RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS
 7 CHAPTER SEVEN ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS 136 7.1 INTRODUCTION Breast cancer is one of the leading causes of death in women (Stephens et al., 2012). Several chemotherapeutic
7 CHAPTER SEVEN ELUCIDATION OF ANTICANCER MECHANSIMS OF ISOLATED PHYTO- CONSTITUENTS 136 7.1 INTRODUCTION Breast cancer is one of the leading causes of death in women (Stephens et al., 2012). Several chemotherapeutic
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis
 The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
mm Distance (mm)
 b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the
 Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
Supplementary Figure 1: Digitoxin induces apoptosis in primary human melanoma cells but not in normal melanocytes, which express lower levels of the cardiac glycoside target, ATP1A1. (a) The percentage
3D Tissue Models. Simple, Low Cost Fabrication. Simple, Robust Protocols
 3D Tissue Models SynVivo is a physiological, cell-based microfluidic platform that provides a morphologically and physiologically realistic microenvironment allowing real-time study of cellular behavior,
3D Tissue Models SynVivo is a physiological, cell-based microfluidic platform that provides a morphologically and physiologically realistic microenvironment allowing real-time study of cellular behavior,
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Ch. 3 CELLS AND TISSUES. Copyright 2010 Pearson Education, Inc.
 Ch. 3 CELLS AND TISSUES Generalized Cell All cells: Human cells have three basic parts: Plasma membrane flexible outer boundary Cytoplasm intracellular fluid containing organelles Nucleus control center
Ch. 3 CELLS AND TISSUES Generalized Cell All cells: Human cells have three basic parts: Plasma membrane flexible outer boundary Cytoplasm intracellular fluid containing organelles Nucleus control center
Appendix Figure S1 A B C D E F G H
 ppendix Figure S1 C D E F G H ppendix Figure S1. RT and chemotherapy alter PD-L1 expression in PDC cells. Flow cytometric analysis of PD-L1 expression in () KPC and () Pan02 cells following treatment with
ppendix Figure S1 C D E F G H ppendix Figure S1. RT and chemotherapy alter PD-L1 expression in PDC cells. Flow cytometric analysis of PD-L1 expression in () KPC and () Pan02 cells following treatment with
Development of a Modified Hollow Fibre Assay for Studying Agents Targeting the Tumour Neovasculature
 Development of a Modified Hollow Fibre Assay for Studying Agents Targeting the Tumour Neovasculature S.D. SHNYDER 1, J. HASAN 2, P.A. COOPER 1, E. PILARINOU 3, E. JUBB 1, G.C. JAYSON 2 and M.C. BIBBY 1
Development of a Modified Hollow Fibre Assay for Studying Agents Targeting the Tumour Neovasculature S.D. SHNYDER 1, J. HASAN 2, P.A. COOPER 1, E. PILARINOU 3, E. JUBB 1, G.C. JAYSON 2 and M.C. BIBBY 1
Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263
 Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263 Malcolm A. Smith, John M. Maris, Stephen T. Keir, Henry S. Friedman, Richard B. Lock, Hernan Carol, Mayamin Tajbakhsh,
Pediatric Preclinical Testing Program (PPTP) evaluation of the Bcl-2 inhibitor ABT-263 Malcolm A. Smith, John M. Maris, Stephen T. Keir, Henry S. Friedman, Richard B. Lock, Hernan Carol, Mayamin Tajbakhsh,
ANTITUMOR EFFECT OF AN ANTI-ENDOTHELIAL CELL MONOCLONAL ANTIBODY BVE-1 ON SOLID TUMOR XENOGRAFT IN NUDE MICE
 92 Chinese Journal of Cancer Research 11(2): 92 --96, 1999 ANTITUMOR EFFECT OF AN ANTI-ENDOTHELIAL CELL MONOCLONAL ANTIBODY BVE-1 ON SOLID TUMOR XENOGRAFT IN NUDE MICE LI Peiyu z}~r~]~-,1 YUAN Mei 3~'J~,
92 Chinese Journal of Cancer Research 11(2): 92 --96, 1999 ANTITUMOR EFFECT OF AN ANTI-ENDOTHELIAL CELL MONOCLONAL ANTIBODY BVE-1 ON SOLID TUMOR XENOGRAFT IN NUDE MICE LI Peiyu z}~r~]~-,1 YUAN Mei 3~'J~,
Supplemental Figures:
 Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Supplemental Figures: Figure 1: Intracellular distribution of VWF by electron microscopy in human endothelial cells. a) Immunogold labeling of LC3 demonstrating an LC3-positive autophagosome (white arrow)
Endothelial PGC 1 - α 1 mediates vascular dysfunction in diabetes
 Endothelial PGC-1α mediates vascular dysfunction in diabetes Reporter: Yaqi Zhou Date: 04/14/2014 Outline I. Introduction II. Research route & Results III. Summary Diabetes the Epidemic of the 21st Century
Endothelial PGC-1α mediates vascular dysfunction in diabetes Reporter: Yaqi Zhou Date: 04/14/2014 Outline I. Introduction II. Research route & Results III. Summary Diabetes the Epidemic of the 21st Century
Supplemental Information. Differential Effects of EGFL6 on Tumor. versus Wound Angiogenesis
 Cell Reports, Volume 21 Supplemental Information Differential Effects of EGFL6 on Tumor versus Wound Angiogenesis Kyunghee Noh, Lingegowda S. Mangala, Hee-Dong Han, Ningyan Zhang, Sunila Pradeep, Sherry
Cell Reports, Volume 21 Supplemental Information Differential Effects of EGFL6 on Tumor versus Wound Angiogenesis Kyunghee Noh, Lingegowda S. Mangala, Hee-Dong Han, Ningyan Zhang, Sunila Pradeep, Sherry
New Horizons in radionuclide therapy. John Buscombe Royal Free Hospital
 New Horizons in radionuclide therapy John Buscombe Royal Free Hospital Date for you diary Interested in Radionuclide imaging and therapy using antibodies and peptides IRIST 2008 Krakow Poland 18-21 June
New Horizons in radionuclide therapy John Buscombe Royal Free Hospital Date for you diary Interested in Radionuclide imaging and therapy using antibodies and peptides IRIST 2008 Krakow Poland 18-21 June
TITLE: Targeting hosphatidylserince for adioimmunotherapy of east ancer rain etastasis. CONTRACTING ORGANIZATION: U Southwestern Center
 Award Number: W81XWH-12-1-0316 TITLE: Targeting hosphatidylserince for adioimmunotherapy of east ancer rain etastasis PRINCIPAL INVESTIGATOR: Rolf A. Brekken CONTRACTING ORGANIZATION: U Southwestern Center
Award Number: W81XWH-12-1-0316 TITLE: Targeting hosphatidylserince for adioimmunotherapy of east ancer rain etastasis PRINCIPAL INVESTIGATOR: Rolf A. Brekken CONTRACTING ORGANIZATION: U Southwestern Center
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1
 Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information)
 BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information) David Dankort, David P. Curley, Robert A. Cartlidge, Betsy Nelson, Anthony N. Karnezis,
BRaf V600E cooperates with Pten silencing to elicit metastatic melanoma (Nature Genetics Supplementary Information) David Dankort, David P. Curley, Robert A. Cartlidge, Betsy Nelson, Anthony N. Karnezis,
TITLE: Investigation of the Akt/PKB Kinase in the Development of Hormone-Independent Prostate Cancer
 AD Award Number: TITLE: Investigation of the Akt/PKB Kinase in the Development of Hormone-Independent Prostate Cancer PRINCIPAL INVESTIGATOR: Linda A. degraffenried, Ph.D. CONTRACTING ORGANIZATION: The
AD Award Number: TITLE: Investigation of the Akt/PKB Kinase in the Development of Hormone-Independent Prostate Cancer PRINCIPAL INVESTIGATOR: Linda A. degraffenried, Ph.D. CONTRACTING ORGANIZATION: The
BNC105 Results Presentation
 Creating and developing innovative therapies BNC105 Results Presentation Dr Deborah Rathjen CEO & Managing Director, Bionomics Limited Dr José Iglesias Chief Medical Officer, Bionomics Limited Dr Tom Hutson
Creating and developing innovative therapies BNC105 Results Presentation Dr Deborah Rathjen CEO & Managing Director, Bionomics Limited Dr José Iglesias Chief Medical Officer, Bionomics Limited Dr Tom Hutson
Cell Migration and Invasion Assays INCUCYTE LIVE-CELL ANALYSIS SYSTEM. Real-time automated measurements of cell motility inside your incubator
 Cell Migration and Invasion Assays INCUCYTE LIVE-CELL ANALYSIS SYSTEM Real-time automated measurements of cell motility inside your incubator See the whole story Real-time cell motility visualization and
Cell Migration and Invasion Assays INCUCYTE LIVE-CELL ANALYSIS SYSTEM Real-time automated measurements of cell motility inside your incubator See the whole story Real-time cell motility visualization and
TITLE: Antiangiogenic Action of Chemically Modified Tetracyclines in Breast Cancer
 AD Award Number: DAMD17-01-1-0346 TITLE: Antiangiogenic Action of Chemically Modified Tetracyclines in Breast Cancer PRINCIPAL INVESTIGATOR: Sanford R. Simon, Ph.D. CONTRACTING ORGANIZATION: The Research
AD Award Number: DAMD17-01-1-0346 TITLE: Antiangiogenic Action of Chemically Modified Tetracyclines in Breast Cancer PRINCIPAL INVESTIGATOR: Sanford R. Simon, Ph.D. CONTRACTING ORGANIZATION: The Research
Cancer Treatment by Alternating Electric Fields (TTFields); Physical Basis & Clinical Trial Results. Madrid, March 2015
 1 Cancer Treatment by Alternating Electric Fields (TTFields); Physical Basis & Clinical Trial Results Madrid, March 2015 2 Cancer Treatments Surgical - whenever possible, Effective mostly in Early stages,
1 Cancer Treatment by Alternating Electric Fields (TTFields); Physical Basis & Clinical Trial Results Madrid, March 2015 2 Cancer Treatments Surgical - whenever possible, Effective mostly in Early stages,
Boucher et al NCOMMS B
 1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
Cell Category? Prokaryote
 CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
Supplemental Material
 Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Functional genomics reveal that the serine synthesis pathway is essential in breast cancer
 Functional genomics reveal that the serine synthesis pathway is essential in breast cancer Results Presented by Stacey Lin Lloyd Lab http://www.amsbio.com/expression-ready-lentiviral-particles.aspx Overview
Functional genomics reveal that the serine synthesis pathway is essential in breast cancer Results Presented by Stacey Lin Lloyd Lab http://www.amsbio.com/expression-ready-lentiviral-particles.aspx Overview
COMBRETASTATIN A-4 PRODRUG: A POTENT INHIBITOR OF MALIGNANT HEMANGIOENDOTHELIOMA CELL PROLIFERATION
 Int. J. Cancer: 87, 838 843 (2000) 2000 Wiley-Liss, Inc. Publication of the International Union Against Cancer COMBRETASTATIN A-4 PRODRUG: A POTENT INHIBITOR OF MALIGNANT HEMANGIOENDOTHELIOMA CELL PROLIFERATION
Int. J. Cancer: 87, 838 843 (2000) 2000 Wiley-Liss, Inc. Publication of the International Union Against Cancer COMBRETASTATIN A-4 PRODRUG: A POTENT INHIBITOR OF MALIGNANT HEMANGIOENDOTHELIOMA CELL PROLIFERATION
SUPPLEMENTARY INFORMATION
 b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
Vascular-Targeting Therapies for Treatment of Malignant Disease
 2491 Vascular-Targeting Therapies for Treatment of Malignant Disease Dietmar W. Siemann, Ph.D. 1 David J. Chaplin, Ph.D. 2 Michael R. Horsman, Ph.D., D.M.Sc. 3 1 Department of Radiation Oncology, University
2491 Vascular-Targeting Therapies for Treatment of Malignant Disease Dietmar W. Siemann, Ph.D. 1 David J. Chaplin, Ph.D. 2 Michael R. Horsman, Ph.D., D.M.Sc. 3 1 Department of Radiation Oncology, University
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Cancer Metronomic Therapy Milan, February 26, 2016
 Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES
 8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES 8.1. Introduction Nanobiotechnology, an emerging field of nanoscience, utilizes nanobased-systems for various biomedical applications.
8. CHAPTER IV. ANTICANCER ACTIVITY OF BIOSYNTHESIZED SILVER NANOPARTICLES 8.1. Introduction Nanobiotechnology, an emerging field of nanoscience, utilizes nanobased-systems for various biomedical applications.
(A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square),
 Supplementary Figures and Tables Figure S1. Validation of EMT-selective small molecules (A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square), and HMLE_Twist (black diamond) cells
Supplementary Figures and Tables Figure S1. Validation of EMT-selective small molecules (A) Dose response curves of HMLE_shGFP (blue circle), HMLE_shEcad (red square), and HMLE_Twist (black diamond) cells
Figure S1: Effects on haptotaxis are independent of effects on cell velocity A)
 Supplemental Figures Figure S1: Effects on haptotaxis are independent of effects on cell velocity A) Velocity of MV D7 fibroblasts expressing different GFP-tagged Ena/VASP family proteins in the haptotaxis
Supplemental Figures Figure S1: Effects on haptotaxis are independent of effects on cell velocity A) Velocity of MV D7 fibroblasts expressing different GFP-tagged Ena/VASP family proteins in the haptotaxis
Supplemental Figure S1A Notch1
 Supplemental Figure S1A Notch1 erage) epth of Cove ormalized De Log1(No Notch exons Figure S1: A) Relative coverage of Notch1 and Notch 2 exons in HCC2218, HCC1187, MB157, MDA-MB157 cell lines. Blue color
Supplemental Figure S1A Notch1 erage) epth of Cove ormalized De Log1(No Notch exons Figure S1: A) Relative coverage of Notch1 and Notch 2 exons in HCC2218, HCC1187, MB157, MDA-MB157 cell lines. Blue color
- 1 - Cell types Monocytes THP-1 cells Macrophages. LPS Treatment time (Hour) IL-6 level (pg/ml)
 Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplementary Table ST1: The dynamic effect of LPS on IL-6 production in monocytes and THP-1 cells after GdA treatment. Monocytes, THP-1 cells and macrophages (5x10 5 ) were incubated with 10 μg/ml of
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Lankenau Institute for Medical Research Annual Progress Report: 2011 Formula Grant
 Lankenau Institute for Medical Research Annual Progress Report: Formula Grant Reporting Period January, June, Formula Grant Overview The Lankenau Institute for Medical Research received $6,99 in formula
Lankenau Institute for Medical Research Annual Progress Report: Formula Grant Reporting Period January, June, Formula Grant Overview The Lankenau Institute for Medical Research received $6,99 in formula
Figure S1, related to Figure 1. Escaper p38a-expressing cancer cells repopulate the tumors (A) Scheme of the mt/mg reporter that expresses a
 Cancer Cell, Volume 33 Supplemental Information Targeting p38a Increases DNA Damage, Chromosome Instability, and the Anti-tumoral Response to Taxanes in Breast Cancer Cells Begoña Cánovas, Ana Igea, Alessandro
Cancer Cell, Volume 33 Supplemental Information Targeting p38a Increases DNA Damage, Chromosome Instability, and the Anti-tumoral Response to Taxanes in Breast Cancer Cells Begoña Cánovas, Ana Igea, Alessandro
Real-time imaging reveals the single steps of brain metastasis fo mation r
 Real-time imaging reveals the single steps of brain metastasis fo mation r Yvonne Kienast, Louisa von Baumgarten, Martin Fuhrmann, Wolfgang E.F. Klinkert, Roland Goldbrunner, Jochen Herms and Frank Winkler
Real-time imaging reveals the single steps of brain metastasis fo mation r Yvonne Kienast, Louisa von Baumgarten, Martin Fuhrmann, Wolfgang E.F. Klinkert, Roland Goldbrunner, Jochen Herms and Frank Winkler
The Novel Vascular Disrupting Agent ANG501 Induces Cell Cycle Arrest and Enhances Endothelial Cell Sensitivity to Radiation
 ORIGINAL RESEARCH The Novel Vascular Disrupting Agent ANG51 Induces Cell Cycle Arrest and Enhances Endothelial Cell Sensitivity to Radiation Shona T. Dougherty 1, Sean E. Walker 1, Peter D. Davis 2 and
ORIGINAL RESEARCH The Novel Vascular Disrupting Agent ANG51 Induces Cell Cycle Arrest and Enhances Endothelial Cell Sensitivity to Radiation Shona T. Dougherty 1, Sean E. Walker 1, Peter D. Davis 2 and
Phase 1 Trial of ALN-VSP in Cancers Involving the Liver. Annual Meeting of the Controlled Release Society August 2, 2011
 Phase 1 Trial of ALN-VSP in Cancers Involving the Liver Annual Meeting of the Controlled Release Society August 2, 2011 Agenda ALN-VSP: Background Phase 1 Trial Study Design Safety Data Tumor Response
Phase 1 Trial of ALN-VSP in Cancers Involving the Liver Annual Meeting of the Controlled Release Society August 2, 2011 Agenda ALN-VSP: Background Phase 1 Trial Study Design Safety Data Tumor Response
The nab Platform : From Bench to the Clinic and Beyond. Neil P. Desai, PhD Abraxis BioScience LLC
 The nab Platform : From Bench to the Clinic and Beyond Neil P. Desai, PhD Abraxis BioScience LLC Policy Issues in Nanotechnology and Oncology, National Cancer Policy Forum Workshop, Institute of Medicine,
The nab Platform : From Bench to the Clinic and Beyond Neil P. Desai, PhD Abraxis BioScience LLC Policy Issues in Nanotechnology and Oncology, National Cancer Policy Forum Workshop, Institute of Medicine,
Routine Assessment of Cancer Cell Cytotoxicity in a Novel Three Dimensional Culture Assay. Introduction
 Routine Assessment of Cancer Cell Cytotoxicity in a Novel Three Dimensional Culture Assay Application Note 2 Introduction Cancer is a leading cause of death worldwide, accounting for around 7.6 million
Routine Assessment of Cancer Cell Cytotoxicity in a Novel Three Dimensional Culture Assay Application Note 2 Introduction Cancer is a leading cause of death worldwide, accounting for around 7.6 million
AWARD NUMBER: W81XWH TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers
 AWARD NUMBER: W81XWH-12-1-0420 TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers PRINCIPAL INVESTIGATOR: Ryan B. Corcoran, M.D., Ph.D. CONTRACTING
AWARD NUMBER: W81XWH-12-1-0420 TITLE: An in Vivo shrna-drug Screen to Identify Novel Targeted Therapy Combinations for KRAS Mutant Cancers PRINCIPAL INVESTIGATOR: Ryan B. Corcoran, M.D., Ph.D. CONTRACTING
5K ALDEFLUOR-positive/ CXCR1-negative. 5K ALDEFLUOR-positive/ CXCR1-positive BAAA BAAA CXCR1-APC BAAA BAAA CXCR1-APC
 A +DEAB -DEAB K ALDEFLUOR-positive/ CXCR-negative BAAA BAAA CXCR-APC B +DEAB -DEAB K ALDEFLUOR-positive/ CXCR-positive BAAA BAAA CXCR-APC C Supplemental Figure. Tumorigenicity of the ALDEFLUOR-positive/CXCR-positive
A +DEAB -DEAB K ALDEFLUOR-positive/ CXCR-negative BAAA BAAA CXCR-APC B +DEAB -DEAB K ALDEFLUOR-positive/ CXCR-positive BAAA BAAA CXCR-APC C Supplemental Figure. Tumorigenicity of the ALDEFLUOR-positive/CXCR-positive
Supplemental materials for
 Supplemental materials for Pericyte-targeting prodrug overcomes tumor resistance to vascular disrupting agents Minfeng Chen, Xueping Lei, Changzheng Shi, Maohua Huang, Xiaobo Li, Baojian Wu, Zhengqiu Li,
Supplemental materials for Pericyte-targeting prodrug overcomes tumor resistance to vascular disrupting agents Minfeng Chen, Xueping Lei, Changzheng Shi, Maohua Huang, Xiaobo Li, Baojian Wu, Zhengqiu Li,
TEB. Id4 p63 DAPI Merge. Id4 CK8 DAPI Merge
 a Duct TEB b Id4 p63 DAPI Merge Id4 CK8 DAPI Merge c d e Supplementary Figure 1. Identification of Id4-positive MECs and characterization of the Comma-D model. (a) IHC analysis of ID4 expression in the
a Duct TEB b Id4 p63 DAPI Merge Id4 CK8 DAPI Merge c d e Supplementary Figure 1. Identification of Id4-positive MECs and characterization of the Comma-D model. (a) IHC analysis of ID4 expression in the
NKTR-214 plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology
 plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics World Preclinical Congress, 2017
plus NKTR-262, a Scientifically-Guided Rational Combination Approach for Immune Oncology Jonathan Zalevsky SVP, Biology and Preclinical Development Nektar Therapeutics World Preclinical Congress, 2017
MicroRNA 132 mediated loss of p120rasgap activates endothelium to facilitate pathological angiogenesis
 MicroRNA 132 mediated loss of p12rasgap activates endothelium to facilitate pathological angiogenesis Sudarshan Anand, Bharat K. Majeti, Lisette M. Acevedo, Eric A. Murphy, Rajesh Mukthavaram, Lea Scheppke,
MicroRNA 132 mediated loss of p12rasgap activates endothelium to facilitate pathological angiogenesis Sudarshan Anand, Bharat K. Majeti, Lisette M. Acevedo, Eric A. Murphy, Rajesh Mukthavaram, Lea Scheppke,
Human Umbilical Vein Endothelial Cell Manual
 Human Umbilical Vein Endothelial Cell Manual INSTRUCTION MANUAL ZBM0079.03 SHIPPING CONDITIONS Human Umbilical Vein Endothelial Cells, cryopreserved Cryopreserved cells are shipped on dry ice and should
Human Umbilical Vein Endothelial Cell Manual INSTRUCTION MANUAL ZBM0079.03 SHIPPING CONDITIONS Human Umbilical Vein Endothelial Cells, cryopreserved Cryopreserved cells are shipped on dry ice and should
Antivascular effects of electrochemotherapy: implications in treatment of bleeding metastases
 For reprint orders, please contact reprints@expert-reviews.com Antivascular effects of electrochemotherapy: implications in treatment of bleeding metastases Expert Rev. Anticancer Ther. 10(5), 729 746
For reprint orders, please contact reprints@expert-reviews.com Antivascular effects of electrochemotherapy: implications in treatment of bleeding metastases Expert Rev. Anticancer Ther. 10(5), 729 746
VWF other roles than hemostasis. Summary 1: VWF & hemostasis synthesis 11/4/16. Structure/function relationship & functions kDa.
 VWF other roles than hemostasis Len$ng PJ, Casari C et al JTH 2012 Summary 1: VWF & hemostasis synthesis Structure/function relationship & functions (HMWM) 20.000kDa multimerization propeptide FVIII GPIb
VWF other roles than hemostasis Len$ng PJ, Casari C et al JTH 2012 Summary 1: VWF & hemostasis synthesis Structure/function relationship & functions (HMWM) 20.000kDa multimerization propeptide FVIII GPIb
