Omega-3 Polyunsaturated Fatty Acids Antagonize Macrophage Inflammation via Activation of AMPK/SIRT1 Pathway
|
|
|
- Antonia Ford
- 5 years ago
- Views:
Transcription
1 Georgia State University Georgia State University Biology Faculty Publications Department of Biology Omega-3 Polyunsaturated Fatty Acids Antagonize Macrophage Inflammation via Activation of AMPK/SIRT1 Pathway Bingzhong Xue Georgia State University, bxue@gsu.edu Zhenggang Yang Wake Forest University Xianfeng Wang Wake Forest University Hang Shi Georgia State University, hshi3@gsu.edu Follow this and additional works at: Part of the Biology Commons Recommended Citation Xue B, Yang Z, Wang X, Shi H (2012) Omega-3 Polyunsaturated Fatty Acids Antagonize Macrophage Inflammation via Activation of AMPK/SIRT1 Pathway. PLoS ONE 7(10): e doi: This Article is brought to you for free and open access by the Department of Biology at Georgia State University. It has been accepted for inclusion in Biology Faculty Publications by an authorized administrator of Georgia State University. For more information, please contact scholarworks@gsu.edu.
2 Omega-3 Polyunsaturated Fatty Acids Antagonize Macrophage Inflammation via Activation of AMPK/SIRT1 Pathway Bingzhong Xue 1,2 *, Zhenggang Yang 2, Xianfeng Wang 2, Hang Shi 1,2 * 1 Department of Biology, Georgia State University, Atlanta, Georgia, United States of America, 2 Department of Internal Medicine, Wake Forest University School of Medicine, Medical Center Boulevard, Winston-Salem, North Carolina, United States of America Abstract Macrophages play a key role in obesity-induced inflammation. Omega-3 polyunsaturated fatty acids (v-3 PUFAs) eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) exert anti-inflammatory functions in both humans and animal models, but the exact cellular signals mediating the beneficial effects are not completely understood. We previously found that two nutrient sensors AMP-activated protein kinase (AMPK) and SIRT1 interact to regulate macrophage inflammation. Here we aim to determine whether v-3 PUFAs antagonize macrophage inflammation via activation of AMPK/SIRT1 pathway. Treatment of v-3 PUFAs suppresses lipopolysaccharide (LPS)-induced cytokine expression in macrophages. Luciferase reporter assays, electrophoretic mobility shift assays (EMSA) and Chromatin immunoprecipitation (ChIP) assays show that treatment of macrophages with v-3 PUFAs significantly inhibits LPS-induced NF-kB signaling. Interestingly, DHA also increases expression, phosphorylation and activity of the major isoform a1ampk, which further leads to SIRT1 overexpression. More importantly, DHA mimics the effect of SIRT1 on deacetylation of the NF-kB subunit p65, and the ability of DHA to deacetylate p65 and inhibit its signaling and downstream cytokine expression require SIRT1. In conclusion, v-3 PUFAs negatively regulate macrophage inflammation by deacetylating NF-kB, which acts through activation of AMPK/SIRT1 pathway. Our study defines AMPK/SIRT1 as a novel cellular mediator for the anti-inflammatory effects of v-3 PUFAs. Citation: Xue B, Yang Z, Wang X, Shi H (2012) Omega-3 Polyunsaturated Fatty Acids Antagonize Macrophage Inflammation via Activation of AMPK/SIRT1 Pathway. PLoS ONE 7(10): e doi: /journal.pone Editor: Haiyan Xu, Warren Alpert Medical School of Brown University, United States of America Received April 10, 2012; Accepted August 28, 2012; Published October 5, 2012 Copyright: ß 2012 Xue et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: This work was supported by United States National Institutes of Health (NIH) grants R01DK084172, American Heart Association (AHA) Mid-Atlantic Affiliate Grant-in-Aid 11GRNT and P30 AG21332, a NIH center grant for Wake Forest School of Medicine Claude D. Pepper Older Americans Independence Center (to H.S.), and NIH grant R01HL and AHA Scientist Development Grant 10SDG (to B.X.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing Interests: The authors have declared that no competing interests exist. * hshi3@gsu.edu (HS); bxue@gsu.edu (BX) Introduction Chronic inflammation has emerged as one of the key physiological mechanism linking obesity to insulin resistance/type 2 diabetes [1]. Obesity-associated chronic inflammation features increased production of pro-inflammatory cytokines and activation of the inflammatory pathways in key metabolic tissues [1]. It is increasingly recognized that adipose tissue plays a key role in obesity-induced inflammation [1]. Further studies provided solid evidence that adipose tissue in obesity displays increased infiltration of macrophages, and that a major source of the adipose inflammation comes from infiltrated macrophages [2,3]. The role of macrophages in obesity-induced inflammation and insulin resistance has been extensively investigated in a number of genetic models [4,5,6,7]. For instance, targeted deletion of IKK-b in myeloid lineage cells protected mice from high-fat (HF) dietinduced inflammation and insulin resistance [4]. Similarly, JNK1 deletion in hematopoietic cells including macrophages also ameliorated obesity-induced inflammation and insulin resistance in mice [5]. In contrast, myeloid specific deletion of peroxisome proliferator activated receptor-c (PPAR-c) increased systemic inflammation and impaired insulin sensitivity in mice [6,7]. These genetic studies demonstrate that altered macrophage inflammation plays a critical role in obesity-induced inflammation and thereby leads to systemic insulin resistance in obesity. Therefore, searching for novel agents that can antagonize macrophage inflammation may represent a therapeutic strategy for the prevention and treatment of insulin resistance and type 2 diabetes. v-3 polyunsaturated fatty acids (v-3 PUFAs) have shown potent anti-inflammatory effects in disease models featuring chronic inflammation [8,9](see reviews [10,11,12]). The mechanisms underlying v-3 PUFAs anti-inflammatory functions have received investigation. Several plausible theories have been advanced to explain the ability of v-3 PUFAs to antagonize inflammation and include competitive inhibition of conversion of arachidonate to pro-inflammatory lipid intermediates, serving as endogenous ligands for PPARc, generation of anti-inflammatory lipid mediators such as resolvins and protectins, and activation of GPR120 [11,13,14,15,16,17,18]. However, the cellular signals mediating v- 3 PUFAs anti-inflammatory effects are not completely understood. We previously found that two nutrient sensors AMP-activated protein kinase (AMPK) and SIRT1 interact to regulate macrophage inflammation [19]. Indeed, AMPK activation deacetylates NF-kB, which acts through SIRT1, and therefore leads to inhibition of NF-kB signaling and cytokine expression [19]. Our PLOS ONE 1 October 2012 Volume 7 Issue 10 e45990
3 observations raise an interesting question as to whether the antiinflammatory effects of v-3 PUFAs may be through activation of the AMPK/SIRT1 pathways. To address this hypothesis, we measured cytokine expression, and examined NF-kB signaling in v-3 PUFA-treated macrophages using luciferase reporter assays, electrophoretic mobility shift assays (EMSA) and Chromatin immunoprecipitation (ChIP) assays. We also examined the effects of v-3 PUFAs on AMPK expression, phosphorylation and activity, and SIRT1 expression in macrophages. We further tested the ability of v-3 PUFAs to deacetylate the NF-kB subunit p65 and determined whether SIRT1 is required for v-3 PUFAs to inhibit NF-kB signaling and its downstream cytokine expression in SIRT1-knockdown macrophages. Results v-3 PUFAs suppress LPS-induced cytokine expression in macrophages via antagonizing NF-kB pathway We first determined the ability of v-3 PUFAs to antagonize macrophage inflammation. We found that pre-treatment of Raw264.7 macrophages with v-3 PUFA mixture EPA/DHA (50 mm each) significantly suppressed LPS-induced expression of pro-inflammatory genes including TNF-a, IL-6, IL-1b, and inos (Fig. 1). This is consistent with the findings we and others have previously reported in macrophages [20,21,22]. To explore whether v-3 PUFAs acts on the NF-kB pathway to antagonize cytokine expression, we established a NF-kB reporter system where 293T cells were transiently transfected with expression vectors for TLR4 and its cofactor MD-2, together with NF-kB luciferase reporter constructs. Cells were then pre-treated with EPA/DHA mixture (50 mm each) overnight and stimulated with LPS (100 ng/ml). Fig. 2A shows that v-3 PUFAs significantly suppressed NF-kB luciferase reporter activity induced by LPS in 293T cells transfected with TLR4 and MD-2 expression vectors. To further determine whether v-3 PUFAs antagonize the NF-kB signaling in macrophages with endogenous TLR4 and its signaling machinery, Raw264.7 cells were transfected with NF-kB luciferase reporter constructs alone. In consistence, pre-treatment of v-3 PUFAs inhibited LPS-induced NF-kB luciferase reporter activity in macrophages (Fig. 2B). We next determined what step(s) in the TLR4 signaling cascade v-3 PUFAs act on to antagonize NF-kB. Raw cells were transfected with expression vectors of constitutively active (CA) form of MyD88, an immediate adaptor protein of TLR4. We found that v-3 PUFAs were still able to inhibit NF-kB reporter activation induced by CA-MyD88 (Fig. 2C), suggesting that v-3 PUFAs likely act on the downstream signal(s) of TLR4 to inhibit NF-kB. We then performed EMSA to further confirm the inhibitory effect of v-3 PUFAs on endogenous NF-kB signaling in macrophages. As shown in Fig. 2D, treatment of Raw cells with v-3 PUFAs prevented NF-kB DNA binding stimulated by LPS (100 ng/ml). We next performed chromatin immunoprecipitation (ChIP) assays to examine the NF-kB subunit p65 binding to the consensus sequence of the IL-6 promoter. Similarly, v-3 PUFAs blocked LPS-induced p65 DNA binding to the IL-6 promoter (Fig. 2E, left panel). Using SYBR Green PCR to quantitate the immunoprecipitated DNA from the ChIP assays, we further confirmed the inhibitory effects of v-3 PUFAs on p65 DNA binding to the IL-6 promoter (Fig. 2E, right panel). These data suggest that the anti-inflammatory effect of v-3 PUFAs is mainly mediated via inactivation of NF-kB signaling. v-3 PUFAs activate AMPK and enhances SIRT1 expression in macrophages We previously found that two nutrient sensors AMP-activated protein kinase (AMPK) and SIRT1 interact to regulate macrophage inflammation [19]. Therefore, we determined whether v-3 PUFAs antagonize macrophage inflammation via activation of AMPK/SIRT1 pathways. We found that treatment of Raw264.7 macrophages with the v-3 PUFA DHA for 24 hours significantly stimulated AMPK phosphorylation and a1ampk activity (Fig. 3A and 3B). DHA treatment also increased a1ampk protein levels (Fig. 3A), which may contribute to the increased AMPK phosphorylation and activity. AMPK and SIRT1 show striking similarities in sensing nutrient supply and regulating metabolic pathways and are likely to interact to perform these functions. We previously demonstrated that activation of AMPK increases SIRT1 expression in macrophages [19]. Here we determined whether activation of AMPK signaling by v-3 PUFAs increases SIRT1 expression. Using macrophages with a1ampk knockdown by lentiviral ShRNA [19], we found that DHA treatment significantly enhanced SIRT1 protein levels, while DHA was unable to do so in a1ampk knockdown cells (Fig. 3C), suggesting that the ability of v-3 PUFAs to stimulate SIRT1 expression requires AMPK. SIRT1 is required for v-3 PUFAs to deacetylate NF-kB and antagonize its signaling in macrophages We have previously shown that SIRT1 antagonizes NF-kB (p65) activity by deacetylating its lysine 310 in macrophages [19]. We tested whether v-3 PUFAs are also capable of deacetylating NF-kB in macrophages, which requires SIRT1. Using macrophages with SIRT1 knockdown by lentiviral ShRNA [19], we found that in control cells, treatment of the v-3 PUFA DHA substantially blocked acetylation of p65 at lysine310 induced by p300 (Fig. 4A), an acetyltransferase widely used to acetylate p65 [19]. Quantitation of the blot showed that p300 transfection stimulated a 3.5-fold increase of p65 acetylation, while DHA treatment significantly blocked this stimulation by more than 50% (p,0.05, Fig. 4B). In contrast, DHA failed to fully deacetylate p65 at lysine310 in SIRT1 knockdown cells (Fig. 4A). In SIRT1 knockdown cells, p300 transfection markedly increased p65 acetylation by 5.1 folds. However, DHA treatment failed to prevent the stimulation of p65 acetylation by p300 in knockdown cells (p = 0.2, Fig. 4B). We further determined whether SIRT1 is required for v-3 PUFAs to antagonize NF-kB signaling. DHA treatment substantially blocked LPS-stimulated NF-kB reporter activity in control macrophages, whereas DHA failed to exert the same action in SIRT1 knockdown macrophages (Fig. 5). We finally measured the downstream target genes of NF-kB. In parallel, DHA significantly suppressed LPS-induced expression of pro-inflammatory genes including TNF-a, IL-1b, and inos, in control cells, but not in SIRT1-knockdown cells (Fig. 6). Therefore, these data indicate that SIRT1 mediates the antiinflammatory effects of v-3 PUFAs in macrophages. Discussion This study was designed to test the hypothesis that v-3 PUFAs antagonize macrophage inflammation through activation of AMPK/SIRT1 pathways. The plausibility of this hypothesis was driven by several prior findings on v-3 PUFAs anti-inflammatory effects and AMPK and SIRT1 as novel cellular mediators linking nutrient metabolism and inflammation. First, we and others have previously shown that v-3 PUFAs antagonize macrophage inflammation [20,21,22]. However, the cellular mechanisms PLOS ONE 2 October 2012 Volume 7 Issue 10 e45990
4 Figure 1. v-3 PUFAs suppress LPS-induced pro-inflammatory gene expression. Raw264.7 macrophages were pre-treated with v-3 PUFA mixtures EPA/DHA (50 mm each) for 24 hours and then treated with LPS (100 ng/ml) in the presence or absence of v-3 PUFA for additional 4 hours. The expression of pro-inflammatory genes was measured by real-time RT-PCR and normalized to cyclophilin. All data are expressed as mean 6 SE, n = 6. Statistical significance is indicated by the presence of different superscripts. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. A.U.: Arbitrary Units. doi: /journal.pone g001 underlying the anti-inflammatory effects of v-3 PUFAs are not completely understood. Second, we recently demonstrated that two nutrient sensors AMPK and SIRT1 negatively regulate macrophage inflammation [19]. It would be interesting to know whether these two nutrient sensors also respond to v-3 PUFAs, a group of beneficial nutrients commonly seen in supplementation (fish oil) and dietary sources (e.g. fish), and mediate their antiinflammatory effects. Indeed, we found that v-3 PUFAs activate AMPK and SIRT1 pathways that in turn deacetylate the NF-kB subunit p65 and down-regulate its signaling, leading to suppression of pro-inflammatory gene expression. The most significant finding in our study is that the antiinflammatory effect of v-3 PUFAs may be mediated through activation of AMPK/SIRT1 pathways, which results in downregulation of NF-kB signaling by deacetylating its subunit p65. We previously demonstrated that AMPK and SIRT1 can detect excess nutrients in diet-induced obesity and serve as negative regulators of nutrient stress-induced inflammation [19]. We found that Figure 2. v-3 PUFAs antagonize NF-kB signaling in macrophages. (A) v-3 PUFAs suppress TLR4/NF-kB signaling in transfected 293T cells. 293T cells were transfected with TLR4/MD-2 expression vectors, and NF-kB luciferase reporter constructs. Transfected cells were pre-treated with v-3 PUFA mixtures EPA/DHA (50 mm each) for 24 hours and then stimulated with LPS (100 ng/ml) in the presence or absence of v-3 PUFA for additional 24 hours. NF-kB luciferase activity was measured using a Dual-Luciferase Reporter Assay. A.U.: Arbitrary Units. (B) v-3 PUFAs suppress the NF-kB signaling in macrophages. Raw264.7 macrophages were transfected with NF-kB luciferase reporter constructs alone. The treatment is the same as described in (A). (C) v-3 PUFAs act on the downstream signal(s) of TLR4 to inhibit NF-kB. Raw264.7 macrophages were transfected with CA-MyD88 expression vectors and NF-kB luciferase reporter constructs. (D) v-3 PUFAs blocks NF-kB DNA binding. EMSA was conducted to examine the NF-kB DNA binding and was conducted as described in Materials and Methods. (E) v-3 PUFAs blocks the NF-kB subunit p65 binding to the IL-6 promoter. ChIP assays were conducted to examine p65 DNA binding and were conducted as described in Materials and Methods. SYBR Green quantitative PCR was used to quantitate the immunoprecipitated DNA. For (A) (C), all data are expressed as mean 6 SE, n = 6. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. doi: /journal.pone g002 PLOS ONE 3 October 2012 Volume 7 Issue 10 e45990
5 Figure 3. v-3 PUFAs activate AMPK and enhances SIRT1 expression in macrophages. (A) v-3 PUFAs increasesa1ampk protein levels and phosphorylation. Raw264.7 macrophages were treated with DHA (100 mm) for 24 hours. a1ampk protein and AMPK phosphorylation (Thr172) was measured by immunoblotting with specific antibodies. (B) v-3 PUFAs increase a1ampk activity. Raw264.7 macrophages were treated with DHA (100 mm) for 24 hours. a1ampk activity was measured using an immunocomplex assay with SAMS peptide as described in Materials and Methods. Data are expressed as mean 6 SE, n = 6. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. (C) DHA enhances SIRT1 protein levels in control but not in a1ampk knockdown cells. Macrophages with a1ampk knockdown were treated with DHA (100 mm) for 24 hours. SIRT1 protein was measured by immunoblotting with specific antibody. doi: /journal.pone g003 AMPK signaling in adipose tissue and macrophages are substantially down-regulated by inflammatory stimuli LPS and in dietinduced obesity [19]. To test whether the down-regulation of AMPK signaling might be physiologically significant and contributes to obesity-induced inflammation, we explored the role of AMPK in regulation of macrophage inflammation in both gainand loss- of function studies. We showed that AMPK activates SIRT1 to suppress macrophage inflammation [19]. The underlying mechanism includes the ability of AMPK and SIRT to deacetylate NF-kB, whose acetylation status affect NF-kB activity and signaling [23]. Based on these observations, we determined: 1) whether AMPK/SIRT1 not only detects excess unhealthy nutrients (e.g. saturated lipids) associated with obesity, but also responds to healthy nutrients (e.g. v-3 PUFAs) beneficial for prevention and treatment of obesity-associated metabolic disorders; and 2) whether activation of AMPK/SIRT1 in response to v-3 PUFAs antagonizes macrophage inflammation via antagonism of NF-kB signaling by deacetylating p65. We found that v-3 PUFAs increase expression, phosphorylation and activity of the major isoform a1ampk in macrophages, which further leads to SIRT1 over-expression. Our data suggest that AMPK indeed responds to v-3 PUFAs. It is noteworthy that other antiinflammatory/anti-oxidants such as polyphenols can also activate AMPK [24]. It appears that both inflammatory and antiinflammatory signals converge on AMPK that in turn exerts its actions to regulate inflammation. To study the consequence of AMPK/SIRT1 activation by v-3 PUFAs, we examined the NFkB acetylation and signaling. We found that the v-3 PUFA DHA mimics the effect of SIRT1 to deacetylate NF-kB, and SIRT1 mediates the effect of DHA in deacetylation of NF-kB and inhibition of its signaling. v-3 PUFAs anti-inflammatory functions Figure 4. SIRT1 knockdown reduces the ability of v-3 PUFAs to deacetylate NF-kB in macrophages. A representative blot was shown in (A), and the blots were quantitated with a Li-COR Odyssey Infrared System (B). The SIRT1 knockdown or control macrophages were transfected with expression vectors for p65 or p300, and were then treated with DHA (100 mm) for 24 hours. Acetylation of p65 at lysine310 and SIRT1 protein were measured by immunoblotting with specific antibody. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. doi: /journal.pone g004 have been extensively investigated and a number of potential mechanisms have been proposed. For instance, v-3 PUFAs can competitively inhibit the conversion of arachidonate to proinflammatory lipid intermediates [16,17]. v-3 PUFAs have also been shown to serve as endogenous ligands for PPARc [18], a known signal that has anti-inflammatory function. Serhan s and his colleagues have also identified the anti-inflammatory lipid mediators such as resolvins and protectins that mediate v-3 PUFAs effects [13,14,25]. More recently, Olefsky s group has Figure 5. SIRT1 knockdown reduces the ability of v-3 PUFAs to antagonize NF-kB signaling in macrophages. The SIRT1 knockdown or control macrophages were transfected with NF-kB luciferase reporter constructs. Transfected cells were pre-treated with DHA (100 mm) for 24 hours and then stimulated with LPS (100 ng/ml) in the presence or absence of v-3 PUFA for additional 24 hours. NF-kB luciferase activity was measured using a Dual-Luciferase Reporter Assay. Data are expressed as mean 6 SE, n = 6. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. A.U.: Arbitrary Units. doi: /journal.pone g005 PLOS ONE 4 October 2012 Volume 7 Issue 10 e45990
6 Figure 6. SIRT1 is required for v-3 PUFAs to suppress LPS-induced expression of pro-inflammatory genes in macrophages. The SIRT1 knockdown or control macrophages were treated with DHA (100 mm) for 24 hours and then treated with LPS (100 ng/ml) in the presence or absence of v-3 PUFA for additional 4 hours. The expression of pro-inflammatory genes was measured by real-time RT-PCR and normalized to cyclophilin. All data are expressed as mean 6 SE, n = 6. Statistical significance is indicated by the presence of different superscripts. Groups labeled with the same superscripts are not statistically different from each other. Groups labeled with different superscripts are statistically different from each other. A.U.: Arbitrary Units. doi: /journal.pone g006 reported a novel G-protein coupled receptor GPR120 that mediates the potent anti-inflammatory actions and insulin sensitizing effects of v-3 PUFA. It is not clear how v-3 PUFA regulation of AMPK/SIRT1 would fit and interact with the other pathways to regulate inflammation. It is possible that these pathways may be intertwined. For example, as a ligand, v-3 PUFAs can activate PPARc that has been shown to activate AMPK [26,27]. It is also noteworthy that although we demonstrate that anti-inflammatory effect of v-3 PUFAs is mediated through antagonism of NF-kB signaling, we do not exclude the possibility that v-3 PUFAs may also act on other inflammatory pathways such as JNK and ikk, which appears to be downregulated by v-3 PUFAs in previous reports [9,15]. It is conceivable that all these pathways may not be mutually exclusive, and probably have crosstalk. In summary, we first demonstrate that v-3 PUFAs suppress LPS-induced cytokine expression in macrophages. The antiinflammatory effect of v-3 PUFAs is likely mediated through antagonism of NF-kB signaling. We then demonstrate that AMPK/SIRT1 pathways are downstream signals that mediate v-3 PUFAs anti-inflammatory effects. v-3 PUFAs activates AMPK signaling by increasing its protein levels, which further leads to increased SIRT1 protein expression. More importantly, DHA mimics the effect of SIRT1 on deacetylation of NF-kB, and the full capacity of DHA to deacetylate NF-kB and inhibit its signaling and downstream cytokine expression requires SIRT1. We conclude that v-3 PUFAs negatively regulate macrophage inflammation by deacetylating NF-kB, which acts through activation of AMPK/SIRT1 pathway. AMPK and SIRT1, two classic energy sensors that play key roles in regulating energy metabolism, may serve as novel cellular mediators for the antiinflammatory effects of v-3 PUFAs. Materials and Methods Antibodies and Reagents Phospho-AMPK (Thr172) and acetyl-p65 (lysine-310) were purchased from Cell Signaling (Beverly, MA). Rabbit polyclonal antibodies against SIRT1 and a1ampk were obtained from Upstate (Lake Placid, NY). Rabbit polyclonal antibodies against p65 and goat polyclonal antibodies against actin were from Santa Cruz (Santa Cruz, CA). EPA and DHA were purchased from Sigma-Aldrich (St. Louis, MO). Cell culture Raw264.7 macrophages were purchased from American Type Culture Collection (ATCC, Manassas, VA) and cultured in Dulbecco s Modified Eagle s Medium (DMEM) containing 10% heat-inactivated FBS. AMPK activity Cell lysates (50 mg) were immunoprecipitated with specific antibodies (Upstate) against the a1 subunit bound to protein-g sepharose beads. The kinase activity of the immunoprecipitates was measured using SAMS peptide and [c- 32 P]ATP. Plasmid constructs and transfection Murine TLR4, MD-2, and MyD88-CA expression vectors were described previously [20]. Raw264.7 macrophages or 293T cells were transfected with expression vectors using a SuperFect Transfection Reagent kit (Qiagen, Valencia, CA). Luciferase reporter assay pnfkb-luc and prl-sv40 vectors were purchased from BD Biosciences-Clontech (Mountain View, CA). Luciferase activity was measured using a Dual-Luciferase Reporter Assay kit (Promega, Madison, WI). Lentiviral ShRNA knockdown SIRT1 and a1ampk lentiviral ShRNA knockdown were conducted as we previously described [19]. The SIRT1 and a1ampk lentiviral ShRNA vectors were purchased from Open Biosystems (Huntsville, AL). The ShRNA lentivirus was generated according to the instructions. Briefly, the ShRNA or control lentiviral vectors were co-transfected with the packaging plasmid (pcmv-dr8.91, the Broad Institute, Cambridge, MA) and the envelope plasmid (VSV-G, the Broad Institute) into 293T cells. Medium containing the lentivirus was harvested and filtered, and was used to infect Raw264.7 cells. The infected cells were selected with puromycin (8 mg/ml) for 8 days, and the surviving cells were pooled and used for experiments. Chromatin immunoprecipitation (ChIP) assay ChIP was conducted using a ChIP assay kit (Upstate) as we previously described [19]. Briefly, cells were fixed with 1% of formaldehyde and then harvested in cell lysis buffer (5 mm PIPES, 85 mm KCl, and 0.5% NP-40, supplemented with protease inhibitors, ph 8.0). The lysates were sonicated to shear genomic PLOS ONE 5 October 2012 Volume 7 Issue 10 e45990
7 DNA to an average fragment length of bp. Lysates were centrifuged, and the supernatants were collected. The supernatants underwent overnight immunoprecipitation, elution, reverse cross-link, and protease K digestion. A mock immunoprecipitation with normal serum IgG was also included as a negative control for each sample. The DNA recovered from phenol/chloroform extraction was used for SYBR Green quantitative PCR (Stratagene, Santa Clara, CA), and the DNA quantitation value of each sample was further normalized with the DNA quantitation of individual input control. Electrophoretic mobility shift assays (EMSA) EMSA was conducted as we previously described [20]. The consensus NF-kB oligonucleotides (Promega, Madison, WI) were end-labeled with [c- 32 P]-ATP (Perkin Elmer, Boston, MA) using T4 polynucleotide kinase (Promega). The protein-dna complexes were resolved on a Novex 6% DNA retardation gel (Invitrogen, Carlsbad, CA). Gels were dried and analyzed by a phosphorimaging system (Molecular Dynamics, Sunnyvale, CA). Total RNA extraction and quantitative RT-PCR Macrophage total RNA was extracted using the Tri Reagent kit (Molecular Research Center, Cincinnati, OH), according to manufacturer s protocol. The expression of genes of interest was assessed by quantitative RT-PCR (ABI Universal PCR Master Mix, Applied Biosystems, Foster City, CA) using a Stratagene References 1. Osborn O, Olefsky JM (2012) The cellular and signaling networks linking the immune system and metabolism in disease. Nat Med 18: Xu H, Barnes GT, Yang Q, Tan G, Yang D, et al. (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest 112: Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, et al. (2003) Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest 112: Arkan MC, Hevener AL, Greten FR, Maeda S, Li ZW, et al. (2005) IKK-beta links inflammation to obesity-induced insulin resistance. Nat Med 11: Solinas G, Vilcu C, Neels JG, Bandyopadhyay GK, Luo JL, et al. (2007) JNK1 in hematopoietically derived cells contributes to diet-induced inflammation and insulin resistance without affecting obesity. Cell Metab 6: Hevener AL, Olefsky JM, Reichart D, Nguyen MT, Bandyopadyhay G, et al. (2007) Macrophage PPAR gamma is required for normal skeletal muscle and hepatic insulin sensitivity and full antidiabetic effects of thiazolidinediones. J Clin Invest 117: Odegaard JI, Ricardo-Gonzalez RR, Goforth MH, Morel CR, Subramanian V, et al. (2007) Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature 447: White PJ, Arita M, Taguchi R, Kang JX, Marette A (2010) Transgenic restoration of long-chain n-3 fatty acids in insulin target tissues improves resolution capacity and alleviates obesity-linked inflammation and insulin resistance in high-fat-fed mice. Diabetes 59: Todoric J, Loffler M, Huber J, Bilban M, Reimers M, et al. (2006) Adipose tissue inflammation induced by high-fat diet in obese diabetic mice is prevented by n-3 polyunsaturated fatty acids. Diabetologia 49: Calder PC (2006) n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr 83: 1505S 1519S. 11. White PJ, Marette A (2006) Is omega-3 key to unlocking inflammation in obesity? Diabetologia 49: Browning LM (2003) n-3 Polyunsaturated fatty acids, inflammation and obesityrelated disease. Proc Nutr Soc 62: Serhan CN, Clish CB, Brannon J, Colgan SP, Chiang N, et al. (2000) Novel functional sets of lipid-derived mediators with antiinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing. J Exp Med 192: Serhan CN, Hong S, Gronert K, Colgan SP, Devchand PR, et al. (2002) Resolvins: a family of bioactive products of omega-3 fatty acid transformation circuits initiated by aspirin treatment that counter proinflammation signals. J Exp Med 196: Oh DY, Talukdar S, Bae EJ, Imamura T, Morinaga H, et al. (2010) GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulinsensitizing effects. Cell 142: Mx3000p thermocycler (Stratagene, La Jolla, CA), as we previously described [20]. The primer and probe pairs used in the assays were purchased from Applied Biosystems. Immunoblotting Immunoblotting was conducted as we previously described [19]. Briefly, the transferred membranes were blocked, washed, incubated with various primary antibodies overnight at 4uC, and followed by Alexa Fluor 680-conjugated secondary antibodies (Invitrogen) at room temperature for 2 hrs. The blots were developed with a Li-COR Odyssey Infrared Imager system (Li- COR Biosciences, Lincoln, NE). Statistics All data are expressed as mean6sem. Data were evaluated for statistical significance by one way ANOVA, and statistical significance for comparison of means of different groups was calculated by the least-significant-difference test using the SPSS software package version p,0.05 was considered significant. Author Contributions Conceived and designed the experiments: BX HS. Performed the experiments: BX HS ZY XW. Analyzed the data: BX HS. Wrote the paper: BX HS. 16. Lee TH, Mencia-Huerta JM, Shih C, Corey EJ, Lewis RA, et al. (1984) Effects of exogenous arachidonic, eicosapentaenoic, and docosahexaenoic acids on the generation of 5-lipoxygenase pathway products by ionophore-activated human neutrophils. J Clin Invest 74: Corey EJ, Shih C, Cashman JR (1983) Docosahexaenoic acid is a strong inhibitor of prostaglandin but not leukotriene biosynthesis. Proc Natl Acad Sci U S A 80: Kliewer SA, Sundseth SS, Jones SA, Brown PJ, Wisely GB, et al. (1997) Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci U S A 94: Yang Z, Kahn BB, Shi H, Xue BZ (2010) Macrophage alpha1 AMP-activated protein kinase (alpha1ampk) antagonizes fatty acid-induced inflammation through SIRT1. J Biol Chem 285: Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, et al. (2006) TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 116: Itoh M, Suganami T, Satoh N, Tanimoto-Koyama K, Yuan X, et al. (2007) Increased adiponectin secretion by highly purified eicosapentaenoic acid in rodent models of obesity and human obese subjects. Arterioscler Thromb Vasc Biol 27: Lee JY, Plakidas A, Lee WH, Heikkinen A, Chanmugam P, et al. (2003) Differential modulation of Toll-like receptors by fatty acids: preferential inhibition by n-3 polyunsaturated fatty acids. J Lipid Res 44: Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, et al. (2004) Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. Embo J 23: Zang M, Xu S, Maitland-Toolan KA, Zuccollo A, Hou X, et al. (2006) Polyphenols stimulate AMP-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor-deficient mice. Diabetes 55: Serhan CN, Clish CB, Brannon J, Colgan SP, Gronert K, et al. (2000) Antimicroinflammatory lipid signals generated from dietary N-3 fatty acids via cyclooxygenase-2 and transcellular processing: a novel mechanism for NSAID and N-3 PUFA therapeutic actions. J Physiol Pharmacol 51: Pilon G, Dallaire P, Marette A (2004) Inhibition of inducible nitric-oxide synthase by activators of AMP-activated protein kinase: a new mechanism of action of insulin-sensitizing drugs. J Biol Chem 279: Saha AK, Avilucea PR, Ye JM, Assifi MM, Kraegen EW, et al. (2004) Pioglitazone treatment activates AMP-activated protein kinase in rat liver and adipose tissue in vivo. Biochem Biophys Res Commun 314: PLOS ONE 6 October 2012 Volume 7 Issue 10 e45990
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Anti-Inflammatory Effects of Fish Oils
 Anti-Inflammatory Effects of Fish Oils A. Macrophage Production of Eicosanoids B. w-3 PUFA Supplementation of Macrophages C. w-3 PUFA in IR & diabetes Oswald Quehenberger Departments of Medicine and Pharmacology
Anti-Inflammatory Effects of Fish Oils A. Macrophage Production of Eicosanoids B. w-3 PUFA Supplementation of Macrophages C. w-3 PUFA in IR & diabetes Oswald Quehenberger Departments of Medicine and Pharmacology
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity
 Downloaded from http:// on December 17, 2017. https://doi.org/10.1172/jci.insight.87748 Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity Xianfeng Wang, 1
Downloaded from http:// on December 17, 2017. https://doi.org/10.1172/jci.insight.87748 Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity Xianfeng Wang, 1
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Differentiating omega-3 fatty acids from SPMs (specialized pro-resolving lipid mediators)
 NIHR Southampton Biomedical Research Centre in nutrition Differentiating omega-3 fatty acids from SPMs (specialized pro-resolving lipid mediators) Philip Calder Professor of Nutritional Immunology University
NIHR Southampton Biomedical Research Centre in nutrition Differentiating omega-3 fatty acids from SPMs (specialized pro-resolving lipid mediators) Philip Calder Professor of Nutritional Immunology University
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different?
 Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different? Trevor A Mori, Ph.D., Professor, School of Medicine and Pharmacology, Royal Perth Hospital Unit, University of Western Australia, Perth,
Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different? Trevor A Mori, Ph.D., Professor, School of Medicine and Pharmacology, Royal Perth Hospital Unit, University of Western Australia, Perth,
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Omega-3 Fatty Acids and Athletics. Current Sports Medicine Reports July 2007, 6:
 Omega-3 Fatty Acids and Athletics 1 Current Sports Medicine Reports July 2007, 6:230 236 Artemis P. Simopoulos, MD The Center for Genetics, Nutrition and Health, Washington, DC, USA EIB exercise-induced
Omega-3 Fatty Acids and Athletics 1 Current Sports Medicine Reports July 2007, 6:230 236 Artemis P. Simopoulos, MD The Center for Genetics, Nutrition and Health, Washington, DC, USA EIB exercise-induced
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
SUPPLEMENTAL MATERIALS AND METHODS. Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of
 SUPPLEMENTAL MATERIALS AND METHODS Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of cysteine and methionine and then treated with 10 μm puromycin in depletion medium
SUPPLEMENTAL MATERIALS AND METHODS Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of cysteine and methionine and then treated with 10 μm puromycin in depletion medium
*This statement has not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any
 *This statement has not been evaluated by the Food and Drug Administration. Healthy Fats. Essential for Health. Exceptional Quality. In a nutshell, these are the benefits of Solgar s extensive selection
*This statement has not been evaluated by the Food and Drug Administration. Healthy Fats. Essential for Health. Exceptional Quality. In a nutshell, these are the benefits of Solgar s extensive selection
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Supplementary Material
 Supplementary Material accompanying the manuscript Interleukin 37 is a fundamental inhibitor of innate immunity Marcel F Nold, Claudia A Nold-Petry, Jarod A Zepp, Brent E Palmer, Philip Bufler & Charles
Supplementary Material accompanying the manuscript Interleukin 37 is a fundamental inhibitor of innate immunity Marcel F Nold, Claudia A Nold-Petry, Jarod A Zepp, Brent E Palmer, Philip Bufler & Charles
Modulation of TRP channels by resolvins in mouse and human
 July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
TLR4 links innate immunity and fatty acid induced insulin resistance
 Research article TLR4 links innate immunity and fatty acid induced insulin resistance Hang Shi, Maia V. Kokoeva, Karen Inouye, Iphigenia Tzameli, Huali Yin, and Jeffrey S. Flier Division of Endocrinology,
Research article TLR4 links innate immunity and fatty acid induced insulin resistance Hang Shi, Maia V. Kokoeva, Karen Inouye, Iphigenia Tzameli, Huali Yin, and Jeffrey S. Flier Division of Endocrinology,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Brd4 Coactivates Transcriptional Activation of NF- B via Specific Binding to Acetylated RelA
 MOLECULAR AND CELLULAR BIOLOGY, Mar. 2009, p. 1375 1387 Vol. 29, No. 5 0270-7306/09/$08.00 0 doi:10.1128/mcb.01365-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Brd4 Coactivates
MOLECULAR AND CELLULAR BIOLOGY, Mar. 2009, p. 1375 1387 Vol. 29, No. 5 0270-7306/09/$08.00 0 doi:10.1128/mcb.01365-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Brd4 Coactivates
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis
 Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis via ameliorating lipid homeostasis at adipose tissue-liver axis in mice Meng Wang a, Xiao-Jing Zhang a, Kun Feng
Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis via ameliorating lipid homeostasis at adipose tissue-liver axis in mice Meng Wang a, Xiao-Jing Zhang a, Kun Feng
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Naohito AOKI, Erina ARAKAWA and Miyuki ITO. Department of Life Science, Graduate School of Bioresources, Mie University Tsu ABSTRACT
 Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3
 Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
S1a S1b S1c. S1d. S1f S1g S1h SUPPLEMENTARY FIGURE 1. - si sc Il17rd Il17ra bp. rig/s IL-17RD (ng) -100 IL-17RD
 SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
Supplemental Information. Increased 4E-BP1 Expression Protects. against Diet-Induced Obesity and Insulin. Resistance in Male Mice
 Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplemental Information
 Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
: Overview of EFA metabolism
 Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Supporting Information
 Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Prostaglandins And Other Biologically Active Lipids
 Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Omega-3 supplementation promotes key anti-inflammatory pathways that naturally support back, neck, and joint health and mobility
 n Promotes key anti-inflammatory pathways n Promotes joint mobility and flexibility n Is a natural adjunct to conventional therapies n Is safe for long-term use Omega-3 supplementation promotes key anti-inflammatory
n Promotes key anti-inflammatory pathways n Promotes joint mobility and flexibility n Is a natural adjunct to conventional therapies n Is safe for long-term use Omega-3 supplementation promotes key anti-inflammatory
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS
 Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh-
 1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
Title: Low omega-3 fatty acid levels associate with frequent gout attacks a case
 Title: Low omega-3 fatty acid levels associate with frequent gout attacks a case control study Authors: A Abhishek 1, Ana M Valdes 1, Michael Doherty 1 Affiliation: Academic Rheumatology, University of
Title: Low omega-3 fatty acid levels associate with frequent gout attacks a case control study Authors: A Abhishek 1, Ana M Valdes 1, Michael Doherty 1 Affiliation: Academic Rheumatology, University of
In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described
 Supplemental Methods In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described previously 1 2 using 32P-labeled 211 bp fragment from 3 HS1. Footprinting reaction mixes contained
Supplemental Methods In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described previously 1 2 using 32P-labeled 211 bp fragment from 3 HS1. Footprinting reaction mixes contained
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Optimize Your Omega-3 Status Personalized Blood Test Reveals a Novel Cardiac Risk Factor
 http://www.lef.org/ Life Extension Magazine May 2010 Optimize Your Omega-3 Status Personalized Blood Test Reveals a Novel Cardiac Risk Factor By Julius Goepp, MD Suppose you could assess with precision
http://www.lef.org/ Life Extension Magazine May 2010 Optimize Your Omega-3 Status Personalized Blood Test Reveals a Novel Cardiac Risk Factor By Julius Goepp, MD Suppose you could assess with precision
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Involvement of n-3 PUFA-derived lipid mediators in the resolution of brain inflammation
 The 1 th Nutrition-Neuroscience meeting The 27 th of march 217 Involvement of n-3 PUFA-derived lipid mediators in the resolution of brain inflammation Charlotte Rey- PhD student Acute inflammatory response
The 1 th Nutrition-Neuroscience meeting The 27 th of march 217 Involvement of n-3 PUFA-derived lipid mediators in the resolution of brain inflammation Charlotte Rey- PhD student Acute inflammatory response
Stewart et al. CD36 ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer
 NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
Impact of hyper-o-glcnacylation on apoptosis and NF-κB activity SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS 3D culture and cell proliferation- MiaPaCa-2 cell culture in 3D was performed as described previously (1). Briefly, 8-well glass chamber slides were evenly coated with 50 µl/well
SUPPLEMENTARY METHODS 3D culture and cell proliferation- MiaPaCa-2 cell culture in 3D was performed as described previously (1). Briefly, 8-well glass chamber slides were evenly coated with 50 µl/well
Rabbit Polyclonal antibody to NFkB p65 (v-rel reticuloendotheliosis viral oncogene homolog A (avian))
 Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
Vadim Ivanov, M.D., Ph.D. Micronutrients in controlling INFLAMMATION Webinar June 12, 2012
 Vadim Ivanov, M.D., Ph.D. Micronutrients in controlling INFLAMMATION Webinar June 12, 2012 1. What is Inflammation? 2. Acute and Chronic Inflammation 3. Role of Chronic Inflammation in Modern Human Diseases
Vadim Ivanov, M.D., Ph.D. Micronutrients in controlling INFLAMMATION Webinar June 12, 2012 1. What is Inflammation? 2. Acute and Chronic Inflammation 3. Role of Chronic Inflammation in Modern Human Diseases
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
INFLAMMATION & REPAIR
 INFLAMMATION & REPAIR Lecture 7 Chemical Mediators of Inflammation Winter 2013 Chelsea Martin Special thanks to Drs. Hanna and Forzan Course Outline i. Inflammation: Introduction and generalities (lecture
INFLAMMATION & REPAIR Lecture 7 Chemical Mediators of Inflammation Winter 2013 Chelsea Martin Special thanks to Drs. Hanna and Forzan Course Outline i. Inflammation: Introduction and generalities (lecture
TD-BF01: Innate immunity to microorganisms
 TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
Pair-fed % inkt cells 0.5. EtOH 0.0
 MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Supplementary Figure 1: STAT3 suppresses Kras-induced lung tumorigenesis
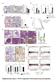 Supplementary Figure 1: STAT3 suppresses Kras-induced lung tumorigenesis (a) Immunohistochemical (IHC) analysis of tyrosine 705 phosphorylation status of STAT3 (P- STAT3) in tumors and stroma (all-time
Supplementary Figure 1: STAT3 suppresses Kras-induced lung tumorigenesis (a) Immunohistochemical (IHC) analysis of tyrosine 705 phosphorylation status of STAT3 (P- STAT3) in tumors and stroma (all-time
Docosahexaenoic Acid Decreases Pro-Inflammatory Mediators in an In Vitro Murine Adipocyte Macrophage Co-Culture Model
 Docosahexaenoic Acid Decreases Pro-Inflammatory Mediators in an In Vitro Murine Adipocyte Macrophage Co-Culture Model Anna A. De Boer, Jennifer M. Monk, Lindsay E. Robinson* Department of Human Health
Docosahexaenoic Acid Decreases Pro-Inflammatory Mediators in an In Vitro Murine Adipocyte Macrophage Co-Culture Model Anna A. De Boer, Jennifer M. Monk, Lindsay E. Robinson* Department of Human Health
Omega 3 and its impact on immune health:
 Omega 3 and its impact on immune health: exploring the potential of omega 3 Gerard Bannenberg November 20 th, 2014 Food Matters Live, London Session: Capitalising on the immunity 'super' trend Objectives
Omega 3 and its impact on immune health: exploring the potential of omega 3 Gerard Bannenberg November 20 th, 2014 Food Matters Live, London Session: Capitalising on the immunity 'super' trend Objectives
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
FROM ABSTRACT Patients with rheumatoid arthritis (RA) improve on a vegetarian diet or supplementation with fish oil.
 Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis Rheumatol Int (2003) 23: 27 36 Olaf Adam, Corinna Beringer, Thomas Kless, Christa Lemmen, Alexander
Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis Rheumatol Int (2003) 23: 27 36 Olaf Adam, Corinna Beringer, Thomas Kless, Christa Lemmen, Alexander
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the
 Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Innate Immunity & Inflammation
 Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
In diabetic patients atherosclerosis and its clinical
 Original Article Polyphenols Stimulate AMP-Activated Protein Kinase, Lower Lipids, and Inhibit Accelerated Atherosclerosis in Diabetic LDL Receptor Deficient Mice Mengwei Zang, 1 Shanqin Xu, 1 Karlene
Original Article Polyphenols Stimulate AMP-Activated Protein Kinase, Lower Lipids, and Inhibit Accelerated Atherosclerosis in Diabetic LDL Receptor Deficient Mice Mengwei Zang, 1 Shanqin Xu, 1 Karlene
Department of Pharmaceutical Sciences, School of Pharmacy, Northeastern University, Boston, MA 02115, USA 2
 Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Outline. Fitting Nutrition into Your Genes. Martha A. Belury, Ph.D., R.D. Martha A. Belury Sept 2018
 Fitting Nutrition into Your Genes, Ph.D., R.D. Human Nutrition The Ohio State University Outline I. History (abridged) of the Study of Genetics and Nutrition II. The Skinny on Fats & Genes III. How Dietary
Fitting Nutrition into Your Genes, Ph.D., R.D. Human Nutrition The Ohio State University Outline I. History (abridged) of the Study of Genetics and Nutrition II. The Skinny on Fats & Genes III. How Dietary
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for use with the PrimePCR Reverse Transcription Control Assay
 Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Taylor Yohe. Project Advisor: Dr. Martha A. Belury. Department of Human Nutrition at the Ohio State University
 Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
SUPPLEMENT. Materials and methods
 SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
University of California, San Diego La Jolla CA 92093
 AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
Supplemental information
 Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
SUPPORTING MATREALS. Methods and Materials
 SUPPORTING MATREALS Methods and Materials Cell Culture MC3T3-E1 (subclone 4) cells were maintained in -MEM with 10% FBS, 1% Pen/Strep at 37ºC in a humidified incubator with 5% CO2. MC3T3 cell differentiation
SUPPORTING MATREALS Methods and Materials Cell Culture MC3T3-E1 (subclone 4) cells were maintained in -MEM with 10% FBS, 1% Pen/Strep at 37ºC in a humidified incubator with 5% CO2. MC3T3 cell differentiation
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by. activating Syk and promoting MyD88 and TRIF degradation via cbl-b
 Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Research shows that the most reliable source of omega-3s is a high-quality fish oil supplement
 n Helps the body naturally address occasional eye irritation n Promotes healthy eye moisture for occasional dry eyes n Promotes normal eye function as we age n Contains the fatty acid DHA, found in greatest
n Helps the body naturally address occasional eye irritation n Promotes healthy eye moisture for occasional dry eyes n Promotes normal eye function as we age n Contains the fatty acid DHA, found in greatest
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author):
 Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Alpha Lipoic Acid Snapshot Monograph
 vitamins minerals nutrients Alpha Lipoic Acid Snapshot Monograph Alpha lipoic Acid Most Frequent Reported Uses: - Antioxidant - Peripheral neuropathy - Improves insulin signaling and regulation of appetite
vitamins minerals nutrients Alpha Lipoic Acid Snapshot Monograph Alpha lipoic Acid Most Frequent Reported Uses: - Antioxidant - Peripheral neuropathy - Improves insulin signaling and regulation of appetite
/06/$15.00/0 Molecular Endocrinology 20(9): Copyright 2006 by The Endocrine Society doi: /me
 0888-8809/06/$15.00/0 Molecular Endocrinology 20(9):2062 2079 Printed in U.S.A. Copyright 2006 by The Endocrine Society doi: 10.1210/me.2005-0316 Androgens, Progestins, and Glucocorticoids Induce Follicle-Stimulating
0888-8809/06/$15.00/0 Molecular Endocrinology 20(9):2062 2079 Printed in U.S.A. Copyright 2006 by The Endocrine Society doi: 10.1210/me.2005-0316 Androgens, Progestins, and Glucocorticoids Induce Follicle-Stimulating
Yiying Zhang, PhD Research Scientist. Research Summary:
 Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Jes S. Lindholt. The role of fish oil in the natural history of abdominal aortic aneurysms. J Am Heart Assoc Jan 26;7(3).
 The role of fish oil in the natural history of abdominal aortic aneurysms J Am Heart Assoc. 2018 Jan 26;7(3). Jes S. Lindholt Professor in Vascular Surgery, DMSci, Ph.D. Odense University Hospital, Denmark.
The role of fish oil in the natural history of abdominal aortic aneurysms J Am Heart Assoc. 2018 Jan 26;7(3). Jes S. Lindholt Professor in Vascular Surgery, DMSci, Ph.D. Odense University Hospital, Denmark.
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
N-3 polyunsaturated fatty acids and allergic disease
 N-3 polyunsaturated fatty acids and allergic disease Current Opinion in Clinical Nutrition and Metabolic Care Volume 7(2) March 2004 pp 123-129 Susan Prescott and Philip Calder Abbreviations DHA: EPA:
N-3 polyunsaturated fatty acids and allergic disease Current Opinion in Clinical Nutrition and Metabolic Care Volume 7(2) March 2004 pp 123-129 Susan Prescott and Philip Calder Abbreviations DHA: EPA:
Antihyperlipidemic Drugs
 Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Certificate of Analysis
 Certificate of Analysis plvx-ef1α-ires-puro Vector Table of Contents Product Information... 1 Description... 2 Location of Features... 3 Additional Information... 3 Quality Control Data... 4 Catalog No.
Certificate of Analysis plvx-ef1α-ires-puro Vector Table of Contents Product Information... 1 Description... 2 Location of Features... 3 Additional Information... 3 Quality Control Data... 4 Catalog No.
Supplementary Fig. 1. Identification of acetylation of K68 of SOD2
 Supplementary Fig. 1. Identification of acetylation of K68 of SOD2 A B H. sapiens 54 KHHAAYVNNLNVTEEKYQEALAK 75 M. musculus 54 KHHAAYVNNLNATEEKYHEALAK 75 X. laevis 55 KHHATYVNNLNITEEKYAEALAK 77 D. rerio
Supplementary Fig. 1. Identification of acetylation of K68 of SOD2 A B H. sapiens 54 KHHAAYVNNLNVTEEKYQEALAK 75 M. musculus 54 KHHAAYVNNLNATEEKYHEALAK 75 X. laevis 55 KHHATYVNNLNITEEKYAEALAK 77 D. rerio
UCLA Nutrition Noteworthy
 UCLA Nutrition Noteworthy Title Effects of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Breast Cancer Permalink https://escholarship.org/uc/item/12g6398b Journal Nutrition Noteworthy, 2(1) ISSN 1556-1895
UCLA Nutrition Noteworthy Title Effects of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Breast Cancer Permalink https://escholarship.org/uc/item/12g6398b Journal Nutrition Noteworthy, 2(1) ISSN 1556-1895
Effect of High-fat or High-glucose Diet on Obesity and Visceral Adipose Tissue in Mice
 ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
Supplementary Figures
 MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
