Phosphorylation of vasodilator-stimulated phosphoprotein Ser239 suppresses filopodia and invadopodia in colon cancer.
|
|
|
- Abel Lloyd
- 5 years ago
- Views:
Transcription
1 Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics Phosphorylation of vasodilator-stimulated phosphoprotein Ser239 suppresses filopodia and invadopodia in colon cancer. David S Zuzga Thomas Jefferson University Joshua Pelta-Heller Thomas Jefferson University, Joshua.Heller@jefferson.edu Peng Li Thomas Jefferson University Alessandro Bombonati Hospital of the University of Pennsylvania Scott A Waldman Thomas Jefferson University, scott.waldman@jefferson.edu See next page for additional authors Let us know how access to this document benefits you Follow this and additional works at: Part of the Medical Pharmacology Commons, and the Pharmacy and Pharmaceutical Sciences Commons Recommended Citation Zuzga, David S; Pelta-Heller, Joshua; Li, Peng; Bombonati, Alessandro; Waldman, Scott A; and Pitari, Giovanni Mario, "Phosphorylation of vasodilator-stimulated phosphoprotein Ser239 suppresses filopodia and invadopodia in colon cancer." (2012). Department of Pharmacology and Experimental Therapeutics Faculty Papers. Paper This Article is brought to you for free and open access by the Jefferson Digital Commons. The Jefferson Digital Commons is a service of Thomas Jefferson University's Center for Teaching and Learning (CTL). The Commons is a showcase for Jefferson books and journals, peer-reviewed scholarly publications, unique historical collections from the University archives, and teaching tools. The Jefferson Digital Commons allows researchers and interested readers anywhere in the world to learn about and keep up to date with Jefferson scholarship. This article has been accepted for inclusion in Department of Pharmacology and Experimental Therapeutics Faculty Papers by an authorized administrator of the Jefferson Digital Commons. For more information, please contact: JeffersonDigitalCommons@jefferson.edu.
2 Authors David S Zuzga, Joshua Pelta-Heller, Peng Li, Alessandro Bombonati, Scott A Waldman, and Giovanni Mario Pitari This article is available at Jefferson Digital Commons:
3 Phosphorylation of Vasodilator-Stimulated Phosphoprotein Ser239 Suppresses Filopodia and Invadopodia in Colon Cancer David S. Zuzga 1,2, Joshua Pelta-Heller 1, Peng Li 1, Alessandro Bombonati 3, Scott A. Waldman 1, and Giovanni M. Pitari 1 1 Department of Pharmacology and Experimental Therapeutics, Thomas Jefferson University, Philadelphia, PA 19107; 2 Department of Biology and Health Sciences, Pace University, New York, NY 10038; 3 Department of Pathology, Hospital of the University of Pennsylvania, Philadelphia, PA Corresponding Author: Giovanni M. Pitari, MD, PhD, Department of Pharmacology and Experimental Therapeutics, Thomas Jefferson University, 1020 Locust St., Suite 348, Philadelphia, PA 19107; Tel.: +(1) ; Fax: +(1) ; Giovanni.Pitari@jefferson.edu Short Title: VASP Ser239 phosphorylation suppresses invadopodia Research Article category: Cancer Cell Biology Key Words: Colorectal cancer; guanylyl cyclase C; cyclic GMP; vasodilator-stimulated phosphoprotein; invadopodia; filopodia. List of Principal Abbreviations: cgmp, cyclic guanosine monophosphate; EVH1, Ena/VASP homology 1; EVH2, Ena/VASP homology 2; F-actin, filamentous actin; G- actin, globular actin; GFP, green fluorescent protein; GCC, guanylyl cyclase C; IHC, immunohistochemistry; MMP, matrix metalloproteinase; PKG, cgmp-dependent protein kinase; VASP, vasodilator-stimulated phosphoprotein. Impact Statement: The manuscript demonstrates VASP Ser239 phosphorylation opposes malignant cell shape promoting invasion in colon cancer cells. Induction of 1
4 VASP Ser239 phosphorylation may represent an innovative intervention to prevent tumor progression and metastasis in patients with colorectal cancer. 2
5 ABSTRACT In colorectal cancer, the antitumorigenic guanylyl cyclase C (GCC) signalome is defective reflecting ligand deprivation from downregulation of endogenous hormone expression. Although the proximal intracellular mediators of that signal transduction system, including cyclic guanosine monophosphate (cgmp) and cgmp-dependent protein kinase (PKG), are well characterized, the functional significance of its distal effectors remain vague. Dysregulation of ligand-dependent GCC signaling through vasodilator-stimulated phosphoprotein (VASP), an actin-binding protein implicated in membrane protrusion dynamics, drastically reduced cgmp-dependent VASP phosphorylation levels in colorectal tumors from patients. Restoration of cgmpdependent VASP phosphorylation by GCC agonists suppressed the number and length of locomotory (filopodia) and invasive (invadopodia) actin-based organelles in human colon cancer cells. Membrane organelle disassembly reflected specific phosphorylation of VASP Ser239, the cgmp/pkg preferred site, and rapid VASP removal from tumor cell protrusions. Importantly, VASP Ser239 phosphorylation inhibited the proteolytic function of invadopodia, reflected by suppression of the cancer cell ability to digest DQcollagen IV embedded in Matrigel. These results demonstrate a previously unrecognized role for VASP Ser239 phosphorylation, a single intracellular biochemical reaction, as an effective mechanism which opposes tumor cell shape promoting colon cancer invasion and metastasis. Reconstitution of physiological cgmp circuitry through VASP, in turn, represents an attractive targeted approach for patients with colorectal cancer. 3
6 INTRODUCTION Despite considerable advancements in elucidating pathognomonic genetic and molecular alterations, the causal mechanisms underlying initiation and progression of colorectal cancer, the third most common and deadly neoplasm in the western world, remain unknown. In the paracrine hormone hypothesis of colon cancer, 1 tumorigenesis is characterized by a state of guanylyl cyclase C (GCC) ligand insufficiency, in which expression of endogenous hormones guanylin and uroguanylin, secreted by and activating GCC receptors on intestinal epithelial cells in an autocrine and paracrine fashion, is dramatically reduced following a yet unidentified event early during transformation. 2-3 Ligand-induced GCC activation physiologically regulates intestinal fluid and electrolyte homeostasis through intracellular accumulation of the second messenger cyclic guanosine monophosphate (cgmp) and cgmp-dependent protein kinase (PKG) phosphorylation of ion channels. 4 Activation of GCC also promotes the transition from the proliferative crypt phenotype to the differentiated colonocyte along intestinal mucosal surfaces by imposing cytostasis 5 and oxidative metabolic reprogramming, 6 effects presumably underlying GCC-mediated colorectal antitumorigenesis. 7 Thus, the GCC ligandopenia associated with neoplastic transformation may promote colorectal carcinogenesis by producing a repressed GCC and cgmp pathway devoid of tumor suppressor activities. However, the exact consequences at the molecular level of the absence of GCC activation in colon cancer cells are unclear. A poorly investigated target for the antitumorigenic GCC pathway is the vasodilatorstimulated phosphoprotein (VASP). 8 A mammalian member of the highly conserved 4
7 Ena/VASP family proteins, VASP acts as a molecular scaffold to bring together growing actin filaments with regulatory proteins at the cell leading edge. 9 VASP controls cell spreading and migration by regulating the formation and stability of protrusive membrane structures driven by actin polymerization, including lamellipodia and filopodia, and the integrity of cell-matrix interactions at adhesion complexes. 9 In that context, VASP exerts a pivotal role in filopodial dynamics, reflected by its localization at the filopodial tip complex where it promotes filopodia formation and elongation by recruiting the initial nanomachinery and imposing anti-capping pressure on actin filament barbed ends. 10 Three critical domains mediates VASP/actin interactions: 9 1) the Ena/VASP homology 1 (EVH1) domain at the N-terminus, which binds to proteins with poly-proline II helix, including vinculin and zyxin; 2) the central prolin-rich region, which binds to proteins containing SH3 and WW domains and the globular (G)-actin binding profilin, and 3) the EVH2 domain at the C-terminus, which binds to both G- and filamentous (F)-actin and mediates VASP oligomerization, thereby promoting F-actin bundling and stabilization. Importantly, VASP harbors 3 phosphorylation sites (Ser157, Ser239 and Thr278) which are targeted with different affinities by PKG, camp-dependent protein kinase and AMPactivated protein kinase, and modulate its localization and function Phosphorylation of VASP Ser239, adjacent to the G-actin binding site in the EVH2 domain, is selectively regulated by cgmp-dependent signaling through PKG, 11 an event that disrupts VASP anti-capping and filament-bundling activities and inhibits membrane protrusion 9, 14 formation. While its engagement in migration and membrane protrusion dynamics in normal cells is well-established, a similar role for VASP Ser phosphorylation in cancer cells has not 5
8 emerged. However, actin cytoskeletal remodeling at dynamic membrane regions determines oncogenic behaviors, from epithelial-to-mesenchymal transition to tissue invasion and metastasis In principle, PKG-mediated VASP phosphorylation could represent one of the biochemical reactions of the antitumorigenic GCC/cGMP signalome. 1 Here, human colon tumors exhibited reduced GCC ligand expression associated with depletion of cgmp-dependent VASP phosphorylation, indicating interruption of downstream signaling by the dysregulated GCC pathway. Restoration of cgmp-mediated phosphorylation of VASP Ser239 by ligand-induced GCC signaling suppressed the invasive cancer cell phenotype, with disassembly of protrusive membrane organelles (filopodia, invadopodia) and inability to promote DQ-collagen IV degradation. Together, these observations reveal a previously unrecognized suppressor of invasive membrane protrusions and therapeutic target, VASP Ser239 phosphorylation, for colorectal cancer. MATERIAL AND METHODS Reagents. ST was prepared as described, 5 while 8-br-cGMP was obtained from Sigma- Aldrich (St. Louis, MO). Antibodies to human VASP, phosphorylated VASP at Ser157, GAPDH, villin and cortactin were from Santa Cruz Biotechnology (Santa Cruz, CA). Two anti-phoshorylated VASP at Ser239 antibodies were purchased, including clone 16C2 from Abcam (Cambridge, MA) and a rabbit antibody from Sigma-Aldrich. The anti-human guanylin antibody was from BioDesign International (Saco, ME), while the anti-human PKG was obtained from Assay Designs (Ann Arbor, MI). The antibody to the human GCC was obtained from Rockland Immunochemicals (Gilbertsville, PA). DQ-collagen IV, Alexa fluor 555 anti-rabbit IgG, Alexa fluor 633 anti-mouse IgG, 6
9 Oregon Green 488 phalloidin and Slowfade Gold with DAPI were obtained from Invitrogen (Carlsbad, CA). Matrigel was from BD Bioscience (Bedford, MA), the general matrix metalloproteinase (MMP) inhibitor GM1006 from Calbiochem (San Diego, CA), and all the reagents for cell culture from Mediatech Inc. (Herndon, VA). Clinical specimens and immunohistochemistry. Paraffin-embedded specimens from 7 patients (Supporting Information Table 1) with histologically-confirmed adenocarcinomas of the colo-rectum were obtained from the Department of Pathology, Anatomy and Cell Biology of Thomas Jefferson University (Philadelphia, PA) under a protocol approved by the Institutional Review Board and subjected to immunohistochemistry (IHC). Briefly, following deparaffinization and rehydratation (with xylene/ethanol/water washes), antigens in tissue sections (5 μm) were unmasked by two consecutive heating cycles (100 C for 5 min in 10 mm citric buffer, ph 6.0). Then, specimens were incubated overnight (4 C) with antibodies (1:100 dilution, unless otherwise indicated) against either human guanylin, GCC, PKG, VASP, and phosphorylated VASP at Ser157 or Ser239 followed by incubation with the respective secondary antibody and DAB substrate (Avidin-biotin kit; Vector Laboratory, Burlingame, CA). Staining intensity was quantified by a blinded pathologist on a 1 to 3 scale (1, low or absent; 2, medium; 3 high). Cell culture. All the human colon carcinoma cell lines investigated were purchased from the American Type Culture Collection (ATCC, Manassas, VA) and used within 6 months from resuscitation. Methods of cell authentication by ATCC include analyses of morphological and growth characteristics, isoenzymology and DNA profiles. T84 and HCT-116 human colon carcinoma cells were cultured (37 C, 5% CO 2 ) with 10% fetal 7
10 bovine serum in DMEM/F12 or DMEM, respectively. Cells (2-20 passages) were fed with fresh medium every third day and split when sub-confluent. Plasmids and cell transductions. Full length VASP cdna (Invitrogen, Carlsbad, CA) was sub-cloned into the Xho1-EcoR1 multiple cloning site of mouse stem cell virus (MSCV)-puro retroviral vector. Point mutations of VASP serines 157 (tcc gcc) and 239 (agc gcc) were performed employing the QuikChange XL Site-Directed Mutagenesis kit (Stratagene, La Jolla, CA) to generate VASP mutants with alanine substitutions at Ser157 (S157A), Ser239 (S239A) or both (AA). Green fluorescent protein (GFP)-VASP constructs were generated by sub-cloning wild type VASP or the VASP mutant S239A in-frame to the C-terminus of GFP using the pretroq-acgfp-cl vector (Stratagene). HEK 293T17 cells (from ATCC) were transfected with 1 µg of each purified MSCV construct plus 1 µg of the packaging vector pcl-ampho employing Fugene transfection reagent (Roche, Basel, Switzerland). T84 cells were transduced (for 72 h) with viral supernatants supplemented with 4 µg/ml polybrene, then selected and maintained in 5 µg/ml puromycin. In this way, the following T84 clones stablyexpressing the MSCV-driven gene were generated: VASP, VASP-S157A, VASP-S239A, VASP-AA, GFP, GFP-VASP and GFP-VASP-S239A. Immunoblot analysis. Protein samples (40 g in SDS loading buffer) were separated by electrophoresis on 10% acrylamide Tris-Glycine gels, transferred to nitrocellulose membranes, and probed (1:1,000 each) with rabbit polyclonal antibodies against VASP, phosphorylated VASP at Ser157, GAPDH, villin or mouse monoclonal antibody against phosphorylated VASP at Ser239 in TBS-Tween (5% milk) overnight at 4 C. After washing the primary antibody, membranes were probed with the appropriate secondary 8
11 antibody (1:2,000) (SantaCruz Biotechnology) for 1 h at room temperature. Immunostained bands were imaged with a Kodak Image Station 4000R and quantified by densitometry. Immunofluorescence and imaging. Filopodia dynamics were studied in cancer cells cultured on Lab-Tek glass microscope-chambered slides and treated for 2 h with PBS (control), the GCC ligand ST (1 μm) or the cgmp analog 8-br-cGMP (5 mm). Invadopodia were examined employing glass coverslips coated with 25 l Matrigel and incubated at 37 C (10 min) to solidify. Here, cancer cells were plated drop-wise directly onto Matrigel, allowed to firmly adhere (1 h) and equilibrate (24 h) in their culturing medium, and finally treated (2 h) with PBS or ST (1 μm). Following treatments, cells were fixed (15 min) with ice-cold paraformaldehyde (4%, in PBS) and permeabilized (10 min) with 0.1% Triton X-100. Following overnight incubations (4 C; in PBS containing 2% bovine serum albumin) with mouse anti-cortactin and/or rabbit anti-vasp antibodies (1:100 each) and 5 units/ml Oregon Green 488 phalloidin, cells were incubated (60 min at room temperature) with Alexa fluor 633 anti-mouse IgG and/or Alexa fluor 555 antirabbit IgG. Specificity of antigen/antibody reactions was assessed employing the secondary antibody alone. Some preparations were counterstained with Slowfade Gold anti-fade reagent with DAPI to identify cell nuclei. Images (63x) were acquired with a confocal laser scanning microscope (LSM 510, Carl Zeiss, Jena, Germany) equipped with a monochrome CCD camera and pseudo-colored with image-analysis software. For filopodia measurements, cells were analyzed by differential interference contrast (DIC) microscopy. Filopodial protrusions ( 5 M) were examined on 8-10 random cell colonies per treatment (~ filopodia/treatment) by quantifying the number (per cell 9
12 colony perimeter) and length (the average distance from the cell base to the tip) of filopodia. The cancer cell colony perimeter (~264 ± 41 M) was determined to ensure that results from colonies with different sizes could be directly compared. Measurements were performed using the Zeiss LSM Image Browser software (Carl Zeiss Microimaging, Thornwood, NY). For invadopodia analyses, the number and length of individual invadopodia (the baso-lateral actin-based membrane projections) per cancer cell were quantified (with the Zeiss LSM Image Browser software) on 8-10 random cells per treatment from 3-dimensional (3-D) reconstructions created from orthogonal X, Y and Z sections of complete Z-stack cell images (at 1 μm increments) of the confocal microscope. Finally, live cell microscopy was performed on culture dishes (fitted on a heated stage) of GFP-VASP (n=6) or GFP-VASP-S239A (n=7) cells by acquiring timelapse images at 0, 1, 5, 10 and 15 min after treatment with the confocal microscope. Analysis of filopodia was performed with Image J (NIH, Bethesda, MD). The GFPbased fluorescent signal was quantified at filopodia tips (the distal ¼ of each filopodia), along the entire length of each filopodium and throughout the cell body, and normalized to the background fluorescence. DQ-collagen IV degradation. Glass coverslips were coated with 20 μl Matrigel containing 25 μg/ml DQ-collagen IV and incubated at 37 C (10 min) to solidify. Cancer cells were plated drop-wise directly onto these Matrigel scaffolds, allowed to firmly adhere (1 h), and treated (24 h) in their culturing medium with PBS or ST (1 μm). Then, cells were fixed in methanol (-20 C for 15 min) and processed by confocal microscopy to generate Z-stack images, as described for invadopodia analyses above. DQ-collagen IV degradation was quantified in 8-10 cells per treatment with the NIH Image J as the mean 10
13 fluorescent intensity of the fluorescent cleavage product (from DQ-collagen IV) over the cell and pericellular area per cell area. Results were normalized to the background fluorescence. Statistical analysis. Unless otherwise indicated, data are mean ± SEM of 3 independent experiments. Statistical analyses were performed with the Student s t test. RESULTS VASP is an intracellular effector of the GCC pathway in colon cancer cells. VASP is expressed in colon cancer cells (46-kDa band; Supporting Information Fig. 1a) and distributes to dynamic membrane domains, including cell leading edges (Fig. 1a) and invadopodia (Fig. 1b), where it colocalizes with cortactin and actin (Fig. 1b, right panels). Invadopodia identity was confirmed by assessing their ability to focally degrade DQ-collagen IV (Fig. 1c), a fluorogenic analog of the basement membrane component collagen IV. In addition, VASP is an intracellular target of ligand-dependent GCC signaling in human colon cancer cells, 8 and the potent GCC agonist ST 4 induced rapid phosphorylation of VASP at both Ser157 and Ser239 (Supporting Information Fig. 1b). ST effects on VASP were mimicked by the membrane-permeant cgmp analog 8-brcGMP (Fig. 1d-e and Supporting Information Fig. 2a) and blocked by the selective PKG peptide inhibitor DT2 17 (Fig. 1e and Supporting Information Fig. 2b), confirming that ST induces VASP Ser phosphorylation by activating the GCC/cGMP/PKG pathway in human colon cancer cells. GCC signaling through VASP is dysregulated in colon cancer. In the human colon, the GCC pathway is constitutively activated by the paracrine hormone guanylin, which induces cgmp- and PKG-dependent phosphorylation of specific substrates. 1, 4 11
14 Accordingly, normal colonic mucosa from patients exhibited epithelial cells with VASP Ser157 and Ser239 phosphorylation in the context of an intact guanylin-vasp signaling axis (Fig. 2a, upper panel). However, following transformation and loss of endogenous ligand expression 2-3 this GCC pathway becomes dysregulated (Fig. 2a, upper panel). While GCC and PKG exhibited mixed trends with no significant changes, expression of guanylin and all the VASP species were significantly reduced in colorectal adenocarcinomas compared to their respective normal counterparts (Fig. 2a). Of significance, VASP phosphorylation at Ser157 and Ser239 were the most compromised signals of the GCC pathway, reflected by their downregulation in all tumors examined (Fig. 2a, middle panel) and maximal loss of expression (Fig. 2a, lower panel). Thus, loss of VASP Ser phosphorylation in colon cancer reflects interruption of effective induction and transmission of GCC-VASP signaling, beyond the mere changes in VASP expression (Supporting Information Fig. 3). VASP Ser239 phosphorylation by GCC activation inhibits cancer cell shape mediating migration and invasion. As VASP regulates membrane protrusion geometry 18 and is present at cell leading edges and invadopodia (Fig. 1a-b), the functional consequences of ligand-dependent GCC signaling through VASP Ser phosphorylation on the membrane architecture of colon cancer cells were examined. GCC signaling through cgmp significantly reduced the number and length of migratory (filopodia; Fig. 2b) and invasive (invadopodia; Fig. 2c) actin-based protrusions. Filopodia identity was confirmed by specific immunostaining with the filopodia marker fascin (data not shown). Importantly, cgmp inhibitory effects were specifically mediated by induction of VASP Ser239 phosphorylation, as demonstrated by employing 12
15 tumor cells stably expressing distinct VASP phosphomutants (Supporting Information Fig. 4a). Thus, cancer cells with VASP-S239A or VASP-AA, which contain point mutations at Ser239 and do not accept phosphorylation at that site (Supporting Information Fig. 4b), were resistant to GCC-mediated inhibition of filopodia (Fig. 3) and invadopodia (Fig. 4). In contrast, control cells expressing GFP or overexpressing VASP and tumor cells with the phosphomutant VASP-S157A, which harbors a point mutation at Ser157 only and does not accept phosphorylation at that site (Supporting Information Fig. 4b), were sensitive to suppression of filopodia (Fig. 3) and invadopodia (Fig. 4) upon ligand-dependent GCC signaling. VASP Ser239 phosphorylation suppresses invasive membrane protrusions by inducing intracellular VASP redistribution. The molecular mechanism mediating inhibition of invasive cell shape by VASP Ser239 phosphorylation was investigated with live imaging microscopy in cells stably expressing GFP-VASP constructs (Fig. 5). Colon cancer cells with GFP-VASP, but not mutant cells expressing GFP-VASP-S239A, exhibited disassembly of filopodia (Fig. 5a) and invadopodia (Fig. 5b) upon liganddependent GCC activation. Protrusion retraction by GCC (Fig. 5c) reflected rapid removal of VASP from membrane projections (Fig. 5d), with a calculated half-life of 8.11 min (Supporting Information Fig. 5), and VASP redistribution to inner cell compartments away from membrane tips (Fig. 5e). However, the GFP-VASP-S239A mutant (resistant to Ser239 phosphorylation), was insensitive to GCC signaling (Fig. 5c), and retained its localization (Fig. 5d) and distribution (Fig. 5e) at locomotory membrane extensions, suggesting that VASP Ser239 phosphorylation represents a disassembly 13
16 signal for invasive migratory organelles by uncoupling VASP from actin-based microregions at colon cancer cell membranes. VASP Ser239 phosphorylation prevents DQ-collagen IV degradation by colon cancer cells. Metastatic cancer dissemination requires degradation and remodeling of the surrounding environment, 19 a malignant attribute conferred to colon cancer cells by their invadopodia (Fig. 1c). Strikingly, activation of GCC signaling through VASP abolished the cancer ability to degrade DQ-collagen IV, with similar magnitude to that of a general inhibitor of MMPs (Fig. 6a), enzymes mediating tumor matrix degradation. 20 However, colon cancer cells with VASP-S239A or VASP-AA, but not VASP-S157A, did continue to proteolytically digest DQ-collagen IV following ligand-dependent GCC signaling (Fig. 6a), indicating that VASP Ser239 phosphorylation mediates arrest of invadopodiamediated proteolysis by the GCC pathway. Thus, VASP Ser239 phosphorylation is a unique biochemical reaction that suppresses membrane protrusions promoting tumor invasion in colorectal cancer (Fig. 6b). DISCUSSION The antitumorigenic GCC signalome is deregulated during colorectal transformation reflecting loss of endogenous ligand expression. 1-3 Present findings reveal that this alteration affects a cgmp-dependent intracellular pathway comprising VASP as the distal effector, with subsequent silencing of an important biochemical reaction, VASP Ser239 phosphorylation, which opposes malignant cell shape. Interruption of GCC-VASP signaling, in turn, results in specific depletion of the VASP phosphospecies, which represent previously undescribed biomarkers for GCC deregulation and tumorigenesis in colon cancer. Conversely, restoration of GCC signaling by ligand replacement therapy 14
17 may provide a rationale strategy for reconstituting cgmp-vasp circuitry and preventing metastasis. Indeed, ligand-dependent GCC activation compromises motility and metastasis of colon cancer cells, 21 presumably by targeting VASP signaling suppressing invasive membrane protrusions (Fig. 6b) described herein. Membrane protrusion extension principally reflects the balance of antagonistic forces at actin filament barbed ends imposing persistence (anti-capping proteins) or arrest (capping proteins) of actin polymerization. 10 VASP is a critical anti-capping protein that initiates and maintains dynamic membrane regions by orchestrating a cytoskeletal assembly-line 10, 22 promoting simultaneous F-actin elongation and bundling. In this way, Ena/VASP family members control migration and membrane protrusion formation underlying important physio(patho)logical processes, from wound-healing, platelet aggregation and neural development 9 9, to inflammation, cancer spreading and metastasis. Accordingly, expression of Ena/VASP proteins positively correlates with disease progression in patients with lung 25 or breast 26 cancer, suggesting their involvement in carcinogenesis and metastasis. The present study underscores the critical role of VASP in organizing cancer functions associated with the malignant membrane shape. In that context, although with unclear activities VASP is a molecular component of invadopodia, 27 cancer cell projections mediating focal matrix proteolysis. Here, colon cancer cells required VASP for invadopodia stability and function, indicating that VASP maintains the structural integrity of these invasive protrusions. Indeed, intracellular VASP redistribution and removal from protrusive tips, upon GCC-dependent phosphorylation, destabilizes actin-driven membrane extensions (Fig. 5) by presumably depleting actin filament plus-ends of VASP-mediated anti-capping forces, 22 resulting in 15
18 protrusion retraction from the persistent actin retrograde flow. Importantly, invadopodia represent attractive antimetastatic targets since they function as critical hubs of invasive cell behavior, spatially and temporally organizing the activities of actin nucleators and regulators (Arp2/3, N-WASP, cortactin), signaling enzymes (Src, Erk1/Erk2, FAK), adhesive receptors (integrins β1/β3, CD44), and proteases (MT1-MMP, MMP2, MMP9) Thus, the ability of cgmp-dependent VASP phosphorylation to dismantle invadopodia in colon cancer cells (Fig. 4) offers an innovative antiinvadopodial strategy to be exploited against tumor metastasis in patients. Tumor suppressors inhibiting metastatic cell dissemination are scarce. Their critical value resides in the potential to serve as biomarkers to improve clinical staging and targets to reduce cancer mortality. VASP Ser239 phosphorylation, a simple intracellular biochemical reaction, could represent a unique invasion suppressor with sophisticated regulatory dynamics, an inducible mechanism embedded into signal transduction networks shaping tumor cell metastasis. Further, inhibition of metastasis by VASP Ser239 phosphorylation could be a general strategy, as VASP is ubiquitously expressed across human tissues. 9 Of significance, cgmp-dependent VASP Ser157 phosphorylation does not appear to affect the invasive phenotype of colon cancer cells (Figs. 3, 4 and 6). 9, 14 Since VASP Ser239 is selectively regulated by PKG-dependent phosphorylation, while VASP Ser157 is a more promiscuous site phosphorylated by PKG, campdependent protein kinase, and protein kinase C-dependent and -independent mechanisms, 11, 29 present observations suggest a unique role of the GCC/cGMP/PKG pathway in suppressing colorectal cancer invasion through VASP Ser239. Reduced levels of VASP Ser157 phosphorylation in colon cancer (Fig. 2a), in turn, may reflect 16
19 more complex deregulation of tumor intracellular networks, including cgmp-dependent and -independent pathways, probably affecting carcinogenetic processes beyond GCCregulated invasive cell shape, a model currently being explored in this laboratory. Present findings also support the existence of a biological divergence between cgmp and camp signaling on Ena/VASP activities, an established paradigm in neurons where the camp-vasp axis mediates attractive guidance cues by increasing the number and length of filopodia, while the cgmp/pkg/vasp pathway promotes repulsive guidance cues However, the precise consequences of cyclic nucleotide signaling through VASP on the actin cytoskeleton appear to be cell and tissue specific, as demonstrated by identical cgmp and camp inhibitory actions on platelet aggregation, reflecting respective VASP phosphorylation mechanisms. 32 Regardless, VASP Ser239 phosphorylation induced by cgmp is emerging as a pivotal mechanism mediating VASP redistribution and disassembly of VASP-regulated membrane protrusions, including filopodia and 14, lamellipodia. In that context, Ser239 is strategically located within the EVH2 domain of VASP, immediately adjacent to the G-actin binding site, which localizes VASP at filopodial tips and coordinates the transfer of profilin-bound G-actin to the barbed end of the elongating F-actin filament. 33 Phosphorylation of VASP Ser239 interferes with EVH2-dependent processes at membrane leading edges, destabilizing 14, 33 VASP-actin interactions and organelle protrusive dynamics. Moreover, VASP Ser239, but not VASP Ser157, phosphorylation inhibits VASP-driven F-actin polymerization. 13 This study demonstrates VASP Ser239 phosphorylation suppresses the invasive actin cytoskeleton of tumor cells and represents an unappreciated prototype to be exploited in innovative diagnostic and therapeutic approaches for colorectal cancer. 17
20 Specifically, ligand-dependent GCC signaling may prevent colon cancer metastasis by neutralizing oncogenic VASP functions at the invasive cancer front. 18
21 ACKNOWLEDGMENTS This work was supported by grants to GMP from NIH (R03CA133950) and Elsa U. Pardee Foundation, and to SAW from NIH (CA75123, CA95026, RC1CA112147). The National Institute of Health specifically disclaims responsibility for any analyses, interpretations or conclusions. SAW is the Samuel M.V. Hamilton Professor of Medicine, and a paid consultant to Merck and the Chair (uncompensated) of the Scientific Advisory Board of Targeted Diagnostics and Therapeutics, Inc. GMP receives research salary support from Merck. All the other authors have no conflict of interest to disclose. 19
22 REFERENCES 1. Pitari GM, Li P, Lin JE, Zuzga D, Gibbons AV, Snook AE, Schulz S, Waldman SA. The paracrine hormone hypothesis of colorectal cancer. Clin Pharmacol Ther 2007;82: Steinbrecher KA, Tuohy TM, Heppner Goss K, Scott MC, Witte DP, Groden J, Cohen MB. Expression of guanylin is downregulated in mouse and human intestinal adenomas. Biochem Biophys Res Commun 2000;273: Notterman DA, Alon U, Sierk AJ, Levine AJ. Transcriptional gene expression profiles of colorectal adenoma, adenocarcinoma, and normal tissue examined by oligonucleotide arrays. Cancer Res 2001;61: Lucas KA, Pitari GM, Kazerounian S, Ruiz-Stewart I, Park J, Schulz S, Chepenik KP, Waldman SA. Guanylyl cyclases and signaling by cyclic GMP. Pharmacol Rev 2000;52: Pitari GM, Di Guglielmo MD, Park J, Schulz S, Waldman SA. Guanylyl cyclase C agonists regulate progression through the cell cycle of human colon carcinoma cells. Proc Natl Acad Sci U S A 2001;98: Lin JE, Li P, Snook AE, Schulz S, Dasgupta A, Hyslop TM, Gibbons AV, Marszlowicz G, Pitari GM, Waldman SA. The hormone receptor GUCY2C suppresses intestinal tumor formation by inhibiting AKT signaling. Gastroenterology 2010;138: Li P, Schulz S, Bombonati A, Palazzo JP, Hyslop TM, Xu Y, Barab AA, Siracusa LD, Pitari GM, Waldman SA. Guanylyl cyclase C suppresses intestinal tumorigenesis by 20
23 restricting proliferation and maintaining genomic integrity. Gastroenterology 2007;133: Deguchi A, Soh JW, Li H, Pamukcu R, Thompson WJ, Weinstein IB. Vasodilatorstimulated phosphoprotein (VASP) phosphorylation provides a biomarker for the action of exisulind and related agents that activate protein kinase G. Mol Cancer Ther 2002;1: Krause M, Dent EW, Bear JE, Loureiro JJ, Gertler FB. Ena/VASP proteins: regulators of the actin cytoskeleton and cell migration. Annu Rev Cell Dev Biol 2003;19: Mejillano MR, Kojima S, Applewhite DA, Gertler FB, Svitkina TM, Borisy GG. Lamellipodial versus filopodial mode of the actin nanomachinery: pivotal role of the filament barbed end. Cell 2004;118: Butt E, Abel K, Krieger M, Palm D, Hoppe V, Hoppe J, Walter U. camp- and cgmp-dependent protein kinase phosphorylation sites of the focal adhesion vasodilatorstimulated phosphoprotein (VASP) in vitro and in intact human platelets. J Biol Chem 1994;269: Blume C, Benz PM, Walter U, Ha J, Kemp BE, Renne T. AMP-activated protein kinase impairs endothelial actin cytoskeleton assembly by phosphorylating vasodilatorstimulated phosphoprotein. J Biol Chem 2007;282: Benz PM, Blume C, Seifert S, Wilhelm S, Waschke J, Schuh K, Gertler F, Munzel T, Renne T. Differential VASP phosphorylation controls remodeling of the actin cytoskeleton. J Cell Sci 2009;122:
24 14. Lindsay SL, Ramsey S, Aitchison M, Renne T, Evans TJ. Modulation of lamellipodial structure and dynamics by NO-dependent phosphorylation of VASP Ser239. J Cell Sci 2007;120: Yilmaz M, Christofori G. EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev 2009;28: Yamaguchi H, Condeelis J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochim Biophys Acta 2007;1773: Dostmann WR, Taylor MS, Nickl CK, Brayden JE, Frank R, Tegge WJ. Highly specific, membrane-permeant peptide blockers of cgmp-dependent protein kinase Ialpha inhibit NO-induced cerebral dilation. Proc Natl Acad Sci U S A 2000;97: Bear JE, Gertler FB. Ena/VASP: towards resolving a pointed controversy at the barbed end. J Cell Sci 2009;122: Steeg PS. Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 2006;12: Linder S. The matrix corroded: podosomes and invadopodia in extracellular matrix degradation. Trends Cell Biol 2007;17: Lubbe WJ, Zuzga DS, Zhou Z, Fu W, Pelta-Heller J, Muschel RJ, Waldman SA, Pitari GM. Guanylyl cyclase C prevents colon cancer metastasis by regulating tumor epithelial cell matrix metalloproteinase-9. Cancer Res 2009;69: Applewhite DA, Barzik M, Kojima S, Svitkina TM, Gertler FB, Borisy GG. Ena/VASP proteins have an anti-capping independent function in filopodia formation. Mol Biol Cell 2007;18:
25 23. Philippar U, Roussos ET, Oser M, Yamaguchi H, Kim HD, Giampieri S, Wang Y, Goswami S, Wyckoff JB, Lauffenburger DA, Sahai E, Condeelis JS, et al. A Mena invasion isoform potentiates EGF-induced carcinoma cell invasion and metastasis. Dev Cell 2008;15: Han G, Fan B, Zhang Y, Zhou X, Wang Y, Dong H, Wei Y, Sun S, Hu M, Zhang J, Wei L. Positive regulation of migration and invasion by vasodilator-stimulated phosphoprotein via Rac1 pathway in human breast cancer cells. Oncol Rep 2008;20: Dertsiz L, Ozbilim G, Kayisli Y, Gokhan GA, Demircan A, Kayisli UA. Differential expression of VASP in normal lung tissue and lung adenocarcinomas. Thorax 2005;60: Hu LD, Zou HF, Zhan SX, Cao KM. EVL (Ena/VASP-like) expression is upregulated in human breast cancer and its relative expression level is correlated with clinical stages. Oncol Rep 2008;19: Weaver AM. Invadopodia: specialized cell structures for cancer invasion. Clin Exp Metastasis 2006;23: Stylli SS, Kaye AH, Lock P. Invadopodia: at the cutting edge of tumour invasion. J Clin Neurosci 2008;15: Wentworth JK, Pula G, Poole AW. Vasodilator-stimulated phosphoprotein (VASP) is phosphorylated on Ser157 by protein kinase C-dependent and -independent mechanisms in thrombin-stimulated human platelets. Biochem J 2006;393:
26 30. Lebrand C, Dent EW, Strasser GA, Lanier LM, Krause M, Svitkina TM, Borisy GG, Gertler FB. Critical role of Ena/VASP proteins for filopodia formation in neurons and in function downstream of netrin-1. Neuron 2004;42: Gomez TM, Robles E. The great escape; phosphorylation of Ena/VASP by PKA promotes filopodial formation. Neuron 2004;42: Aszodi A, Pfeifer A, Ahmad M, Glauner M, Zhou XH, Ny L, Andersson KE, Kehrel B, Offermanns S, Fassler R. The vasodilator-stimulated phosphoprotein (VASP) is involved in cgmp- and camp-mediated inhibition of agonist-induced platelet aggregation, but is dispensable for smooth muscle function. EMBO J 1999;18: Barzik M, Kotova TI, Higgs HN, Hazelwood L, Hanein D, Gertler FB, Schafer DA. Ena/VASP proteins enhance actin polymerization in the presence of barbed end capping proteins. J Biol Chem 2005;280: Yaroslavskiy BB, Zhang Y, Kalla SE, Garcia Palacios V, Sharrow AC, Li Y, Zaidi M, Wu C, Blair HC. NO-dependent osteoclast motility: reliance on cgmp-dependent protein kinase I and VASP. J Cell Sci 2005;118:
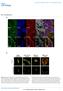
27 FIGURE LEGENDS Fig. 1. VASP Ser phosphorylation is induced by GCC activation in colon cancer cells. (a) Cell leading edges (multi-arrows in upper-left panel) of T84 human colon carcinoma cells (on 2-D surfaces) were imaged (magnification, 63x) by DIC and confocal microscopy following immunofluorescent staining with phalloidin (green) and anti- VASP antibody (red). Yellow in merged image indicates F-actin and VASP colocalization at leading tips. (b) Representative confocal microscopy images of invadopodia from T84 cells grown on 3-D Matrigel scaffolds (Matrigel/air interfaces, dotted lines in top panels). Boxes in cartoons on left indicate visual planes in vertical (top panels) and horizontal (middle panels) cross-sections from Z-stack images. Cells were immunofluorescently stained for invadopodial marker cortactin (ref. 27) (blue), VASP (red) and F-actin (green). In merged images, white (upper panels) and coincidence of picks (bottom panels) from distinct fluorescent signals along lines depicted in middle panels indicate cortactin, VASP and F-actin colocalization. (c) Vertical or horizontal (bottom-right) T84 cell cross-sections of confocal microscopy images obtained as in b. Here, Matrigel contained DQ-collagen IV, which releases a fluorescent product (green) upon cleavage by extracellular proteases. Application of 60 nm of the broad MMP inhibitor GM6001 (immp) was employed to prevent DQ-collagen IV degradation by tumor cells (ref. 20) and serve as the negative control condition. Red in right panels reflects F-actin staining. (d) Immunoblots of a representative experiment with T84 cells, treated with the GCC ligand ST (1 μm, 5 min) or the cgmp analog 8-brcGMP (5 mm, 30 min). ps157-vasp and ps239-vasp, VASP phosphorylated at Ser157 and Ser239, respectively; GAPDH, the loading control. (e) Bar graphs reflect 25
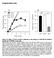
28 densitometric quantification of immunobands, normalized to respective loading (GAPDH) and vehicle (PBS) controls (CTR), from experiments with T84 and HCT116 colon cancer cells treated as in d. Some conditions also received the peptide DT2 (5 μm, 30 min) to selectively and completely inhibit PKG-Iα activity (K i, 12.5 nm; ref. 17)., P < 0.05, versus respective PBS control. Fig. 2. GCC signaling through VASP is disrupted in colon cancer and its reconstitution in vitro suppresses dynamic membrane regions. (a, upper panel) Representative IHC images (magnification, 20x) of primary tumor (Tumor) and matched normal adjacent tissue (NAT) from 7 patients with colorectal cancer. Tissues were stained with the specific primary antibody (brown) and hematoxylin (blue, nuclei). The primary antibody for VASP phosphorylated at Ser239 was clone 16C2 from Abcam. (a, middle panel) Distribution (percentages indicated in boxes) of patients with different expression levels (by staining intensity, as per Material and Methods) of the GCC pathway components. The fraction of patients with upregulation ( ) or downregulation ( ) of the indicated protein in tumors compared to respective NATs is provided in bottom. (a, lower panel) Differential protein expression (the delta of staining intensity scores) in colorectal tumors compared to matched NATs., P < 0.05;, P < of tumors versus NATs. ps157-vasp and ps239-vasp, VASP phosphorylated at Ser157 and Ser239, respectively. (b) Filopodia (arrows in upper-left slide) were imaged in cancer cell colonies by DIC. (c) Invadopodia were imaged by confocal microscopy in single T84 cells, immunofluorescently stained with 4',6-diamidino-2-phenylindole (DAPI, for nuclei in blue) and phalloidin (for F-actin in green). Boxes in cartoons on top indicate visual planes in vertical (left panels) and horizontal (right panels) cross-sections from Z-stack 26
29 images. In b-c, number and length of filopodia and invadopodia were quantified as described in Material and Methods, and results are shown in bar graphs. Treatments (2 h) were PBS (the vehicle control), ST (1 μm), or 8-bromo-cGMP (5 mm)., P < 0.05, versus respective PBS control. Fig. 3. VASP Ser239 phosphorylation by GCC disrupts colon cancer cell filopodia. T84 cells were treated (with the PBS control or 1 μm ST to induce GCC signaling), imaged and analyzed for quantification of filopodia as in Fig. 2b. Cancer cells stably expressed the indicated MSCV-driven genes (see Material and Methods for details), including the controls GFP and VASP, and VASP-S157A (S157A), VASP-S239A (S239A) and VASP-AA (AA). Arrows in upper panels indicate representative filopodia. Bars, 10 µm., P < 0.05 versus respective control. Fig. 4. VASP Ser239 phosphorylation by GCC disrupts colon cancer cell invadopodia. T84 cells were treated (with the PBS control or 1 μm ST to induce GCC signaling), imaged and analyzed for quantification of invadopodia as in Fig. 2c. Cancer cells stably expressed the indicated MSCV-driven genes (see Material and Methods for details), including the control VASP, and VASP-S157A (S157A), VASP-S239A (S239A) and VASP-AA (AA). Boxes in cartoons on left of top images indicate visual planes in vertical (upper panels) and horizontal (bottom panels) cross-sections from Z-stacks., P < 0.05 versus respective control. Fig. 5. VASP Ser239 phosphorylation induces membrane protrusion retraction in colon cancer cells. Fluorescently-tagged VASP constructs, GFP-VASP (VASP) and GFP- VASP-S239A (S239A), were stably expressed in T84 cells as described in Material and Methods. GCC signaling was induced with ST (1 μm), while PBS was the vehicle 27
30 control. (a) Representative time course of a live cell experiment analyzing filopodial kinetics by confocal microscopy, performed as detailed in Material and Methods. Photographs reflect merged DIC and confocal microscopy images. (b) Representative 3- D projections from live imaging (confocal microscopy) analyzing invadopodia in single tumor cells plated on Matrigel scaffolds. Cartoons on top indicate visual vertical (left panels) and basolateral en face (right panels) perspectives of 3-D reconstructions from Z- stack images. (c) The length of filopodia following treatments for 15 min was quantified as in Fig. 2b and expressed as percentages of respective PBS controls. (d) Quantification of GFP-tagged VASP signals at filopodia tips from experiments shown in a. Results are percentages of respective time 0 (before ST administration) controls. (e) Quantification of GFP-tagged VASP signals in cell bodies (CB) and filopodia (F) before (CTR, time 0 control) and after ST (15 min) treatments, as shown in a. AU, arbitrary units., P < 0.05 versus respective control. Fig. 6. VASP Ser239 phosphorylation inhibits the cleavage of DQ-collagen IV by colon cancer cells. (a) T84 cells stably-expressing the indicated MSCV-driven genes (VASP, the control; VASP-S157A, S157A; VASP-S239A, S239A; VASP-AA, AA) were treated for 24 h with PBS, 1 μm ST or 60 nm of the broad MMP inhibitor GM6001 (immp), imaged and analyzed by confocal microscopy for quantification of DQ-collagen IV degradation as detailed in Material and Methods. Photographs (top) are merged DIC and confocal microscopy images. Results (bottom) are expressed as arbitrary units (AU) of the fluorescent signal from the cleaved collagen product. WT, wild-type T84 cells., P < 0.05, versus respective PBS control. (b) Proposed role of VASP in colorectal carcinogenesis. During tumor progression, VASP phosphorylation at Ser239 (p-ser239-28
31 VASP) is reduced, reflecting a dysregulated GCC pathway with interruption of cgmpmediated signaling, which promotes the formation of migratory membrane protrusions and cancer cell invasion. Reconstitution of GCC and cgmp signaling, in turn, provides a unique therapeutic opportunity to restrain the invasive tumor cell shape and prevent metastasis. 29
32 Figure 1 a b Cortactin VASP F-actin Merged DIC β-actin VASP Merged c immp DQ-collagen IV F-actin d VASP (46-kDa) GAPDH ps157-vasp GAPDH ps239-vasp GAPDH - ST - cgmp e pvasp Levels (fold over Control) ps157-vasp ps239-vasp CTR ST cgmp ST + cgmp DT2 T84 HCT116
33 Figure 2 a b Guanylin GCC PKG VASP ps157-vasp ps239-vasp T84 HCT116 - ST cgmp cgmp Filopodia (length, m) Filopodia (number/mm) c 0 PBS ST cgmp NAT High 29 Medium Absent/Low Tumor NAT Tumor NAT Tumor NAT Tumor NAT Tumor NAT Tumor Guanylin GCC PKG VASP ps157-vasp ps239-vasp 0/7; 6/7 2/7; 3/7 4/7; 3/7 0/7; 5/7 0/7; 7/7 0/7; 7/ T84 HCT116 T84 HCT116 Patients, % Guanylin GCC 29 PKG VASP ps157-vasp ps239-vasp Δ-Expression (Tumor-NAT) GCC Signaling Invadopodia (length, m) Invadopodia (number/cell) Tumor NAT cgmp Signaling c ST ST ST - + ST
34 Figure 3 GFP p=0.06 PBS ST GFP VASP S157A S239A AA Filopodia (length, m) Filopodia (number/mm) 0 VASP S157A S239A AA GFP VASP S157A S239A AA GCC Signaling
35 VASP S157A S239A AA - + GCC Signaling GCC Signaling VASP S157A S239A AA Invadopodia (length, m) VASP S157A S239A AA PBS ST Invadopodia (number/cell) Figure 4
36 VASP S239A 0 Minutes Figure 5 b - + c d VASP S239A ST Minutes GCC Signaling Fluorescence Intensity at Filopodia, % 700 p= Fluorescence Intensity, AU Filopodia (length, %) GCC Signaling + a ST Minutes - PBS 0 PBS ST e VASP S239A 0 CTR ST 15 min CB F CB F CB F CB F VASP S239A S239A VASP
37 a Figure 6 WT VASP S157A S239 GCC Signaling - + PBS ST A AA Collagen Degradation (AU) immp PBS ST immp WT VASP S157A S239A AA b GCC Signaling - + Loss of p-ser239-vasp EVH1 PPR EVH2 Reconstitution of p-ser239-vasp P P EVH1 PPR EVH2 BM Tumor Invasion BM Invasion Suppression
Targeting the cgmp Pathway to Treat Colorectal Cancer
 Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 29 Targeting the cgmp
Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 29 Targeting the cgmp
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
SUPPLEMENT. Materials and methods
 SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION
 Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
Figure S1. Loss of Ena/VASP proteins inhibits filopodia and neuritogenesis. (a) Bar graph of filopodia number per stage 1 control and mmvvee (Mena/ VASP/EVL-null) neurons at 40hrs in culture. Loss of all
A Mena Invasion Isoform Potentiates EGF-Induced Carcinoma Cell Invasion and Metastasis
 A Mena Invasion Isoform Potentiates EGF-Induced Carcinoma Cell Invasion and Metastasis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters.
A Mena Invasion Isoform Potentiates EGF-Induced Carcinoma Cell Invasion and Metastasis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters.
supplementary information
 DOI: 10.1038/ncb2133 Figure S1 Actomyosin organisation in human squamous cell carcinoma. (a) Three examples of actomyosin organisation around the edges of squamous cell carcinoma biopsies are shown. Myosin
DOI: 10.1038/ncb2133 Figure S1 Actomyosin organisation in human squamous cell carcinoma. (a) Three examples of actomyosin organisation around the edges of squamous cell carcinoma biopsies are shown. Myosin
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Material and Methods
 Online Supplement Kockx et al, Secretion of Apolipoprotein E from Macrophages 1 Supplementary Material and Methods Cloning of ApoE-GFP Full-length human apoe3 cdna (pcdna3.1/zeo + -apoe) was kindly provided
Online Supplement Kockx et al, Secretion of Apolipoprotein E from Macrophages 1 Supplementary Material and Methods Cloning of ApoE-GFP Full-length human apoe3 cdna (pcdna3.1/zeo + -apoe) was kindly provided
Supplemental Information. Otic Mesenchyme Cells Regulate. Spiral Ganglion Axon Fasciculation. through a Pou3f4/EphA4 Signaling Pathway
 Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Neuron, Volume 73 Supplemental Information Otic Mesenchyme Cells Regulate Spiral Ganglion Axon Fasciculation through a Pou3f4/EphA4 Signaling Pathway Thomas M. Coate, Steven Raft, Xiumei Zhao, Aimee K.
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC
 Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
Supplementary Figure 1. Characterization of NMuMG-ErbB2 and NIC breast cancer cells expressing shrnas targeting LPP. NMuMG-ErbB2 cells (a) and NIC cells (b) were engineered to stably express either a LucA-shRNA
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG)
 In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG) 1 Dr Saeb Aliwaini 13/11/2015 Migration in vivo Primary tumors are responsible for only about 10%
In vitro scratch assay: method for analysis of cell migration in vitro labeled fluorodeoxyglucose (FDG) 1 Dr Saeb Aliwaini 13/11/2015 Migration in vivo Primary tumors are responsible for only about 10%
Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures
 Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures Supplementary Figure 1 mir-128-3p is highly expressed in chemoresistant, metastatic
Type of file: PDF Size of file: 0 KB Title of file for HTML: Supplementary Information Description: Supplementary Figures Supplementary Figure 1 mir-128-3p is highly expressed in chemoresistant, metastatic
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the
 Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Impact of hyper-o-glcnacylation on apoptosis and NF-κB activity SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS 3D culture and cell proliferation- MiaPaCa-2 cell culture in 3D was performed as described previously (1). Briefly, 8-well glass chamber slides were evenly coated with 50 µl/well
SUPPLEMENTARY METHODS 3D culture and cell proliferation- MiaPaCa-2 cell culture in 3D was performed as described previously (1). Briefly, 8-well glass chamber slides were evenly coated with 50 µl/well
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Influenza virus exploits tunneling nanotubes for cell-to-cell spread
 Supplementary Information Influenza virus exploits tunneling nanotubes for cell-to-cell spread Amrita Kumar 1, Jin Hyang Kim 1, Priya Ranjan 1, Maureen G. Metcalfe 2, Weiping Cao 1, Margarita Mishina 1,
Supplementary Information Influenza virus exploits tunneling nanotubes for cell-to-cell spread Amrita Kumar 1, Jin Hyang Kim 1, Priya Ranjan 1, Maureen G. Metcalfe 2, Weiping Cao 1, Margarita Mishina 1,
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY
 Nagoya J. Med. Sci. 72. 139 ~ 144, 1 ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY FENG XU, SATOKO ITO, MICHINARI HAMAGUCHI and TAKESHI
Nagoya J. Med. Sci. 72. 139 ~ 144, 1 ORIGINAL PAPER DISRUPTION OF CELL SPREADING BY THE ACTIVATION OF MEK/ERK PATHWAY IS DEPENDENT ON AP-1 ACTIVITY FENG XU, SATOKO ITO, MICHINARI HAMAGUCHI and TAKESHI
Supplemental information
 Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
ZYXIN-VASP INTERACTIONS ALTER ACTIN REGULATORY ACTIVITY IN ZYXIN-VASP COMPLEXES
 CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 20 June 2012 Volume 18 (2013) pp 1-10 Final form accepted: 12 October 2012 DOI: 10.2478/s11658-012-0035-2 Published online: 17 October
CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 20 June 2012 Volume 18 (2013) pp 1-10 Final form accepted: 12 October 2012 DOI: 10.2478/s11658-012-0035-2 Published online: 17 October
El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb /DC1
 Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
Supplemental material JCB El Azzouzi et al., http ://www.jcb.org /cgi /content /full /jcb.201510043 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Acquisition of -phluorin correlates negatively with podosome
TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
cell movement and neuronal migration
 cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
DNA Microarray Analysis of the Epithelial Mesenchymal Transition of Mesothelial Cells in a Rat Model of Peritoneal Dialysis
 Advances in Peritoneal Dialysis, Vol. 27, 2011 Toshimi Imai, Ichiro Hirahara, Yoshiyuki Morishita, Akira Onishi, Makoto Inoue, Shigeaki Muto, Eiji Kusano DNA Microarray Analysis of the Epithelial Mesenchymal
Advances in Peritoneal Dialysis, Vol. 27, 2011 Toshimi Imai, Ichiro Hirahara, Yoshiyuki Morishita, Akira Onishi, Makoto Inoue, Shigeaki Muto, Eiji Kusano DNA Microarray Analysis of the Epithelial Mesenchymal
High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer.
 Biomedical Research 2017; 28 (18): 7779-7783 ISSN 0970-938X www.biomedres.info High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer. Hu Song 1, Qi-yu Liu 2, Zhi-wei
Biomedical Research 2017; 28 (18): 7779-7783 ISSN 0970-938X www.biomedres.info High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer. Hu Song 1, Qi-yu Liu 2, Zhi-wei
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Figure S1: Effects on haptotaxis are independent of effects on cell velocity A)
 Supplemental Figures Figure S1: Effects on haptotaxis are independent of effects on cell velocity A) Velocity of MV D7 fibroblasts expressing different GFP-tagged Ena/VASP family proteins in the haptotaxis
Supplemental Figures Figure S1: Effects on haptotaxis are independent of effects on cell velocity A) Velocity of MV D7 fibroblasts expressing different GFP-tagged Ena/VASP family proteins in the haptotaxis
Human Urokinase / PLAU / UPA ELISA Pair Set
 Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
An epithelial-to-mesenchymal transition-inducing potential of. granulocyte macrophage colony-stimulating factor in colon. cancer
 An epithelial-to-mesenchymal transition-inducing potential of granulocyte macrophage colony-stimulating factor in colon cancer Yaqiong Chen, Zhi Zhao, Yu Chen, Zhonglin Lv, Xin Ding, Renxi Wang, He Xiao,
An epithelial-to-mesenchymal transition-inducing potential of granulocyte macrophage colony-stimulating factor in colon cancer Yaqiong Chen, Zhi Zhao, Yu Chen, Zhonglin Lv, Xin Ding, Renxi Wang, He Xiao,
Fang et al. NMuMG. PyVmT unstained Anti-CCR2-PE MDA-MB MCF MCF10A
 A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression. through inhibition of Hsp90
 Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplemental Data Figure S1 Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells.
 Supplemental Data Figure S1. Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells. (A) Specific lysis of IFN-γ-treated SKBR-3 cells in the absence
Supplemental Data Figure S1. Effect of TS2/4 and R6.5 antibodies on the kinetics of CD16.NK-92-mediated specific lysis of SKBR-3 target cells. (A) Specific lysis of IFN-γ-treated SKBR-3 cells in the absence
Supplemental Data. TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim.
 Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Nature Methods: doi: /nmeth.4257
 Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Cytoskeleton and cell communication
 Cytoskeleton and cell communication Actin monomer has subdomains 1-4. A simplified cartoon is at right. ATP binds, along with Mg ++, within a deep cleft between subdomains 2 & 4. G-actin (globular actin),
Cytoskeleton and cell communication Actin monomer has subdomains 1-4. A simplified cartoon is at right. ATP binds, along with Mg ++, within a deep cleft between subdomains 2 & 4. G-actin (globular actin),
TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer
 AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
AD Award Number: TITLE: The Role of hcdc4 as a Tumor Suppressor Gene in Genomic Instability Underlying Prostate Cancer PRINCIPAL INVESTIGATOR: Audrey van Drogen, Ph.D. CONTRACTING ORGANIZATION: Sidney
Immature organoids appear after hours.
 THE ESSENTIALS OF LIFE SCIENCE RESEARCH GLOBALLY DELIVERED Allison Ruchinskas, B.S., and James Clinton, Ph.D. ATCC Cell Systems, Gaithersburg, MD INTRODUCTION Figure 1. Mouse small intestinal organoid
THE ESSENTIALS OF LIFE SCIENCE RESEARCH GLOBALLY DELIVERED Allison Ruchinskas, B.S., and James Clinton, Ph.D. ATCC Cell Systems, Gaithersburg, MD INTRODUCTION Figure 1. Mouse small intestinal organoid
Focus Application. Compound-Induced Cytotoxicity
 xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity Featured Study: Using the Time Resolving Function of the xcelligence System to Optimize Endpoint Viability and
xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity Featured Study: Using the Time Resolving Function of the xcelligence System to Optimize Endpoint Viability and
Invasive cancer cells form dynamic adhesive structures
 Published Online: 10 March, 2008 Supp Info: http://doi.org/10.1083/jcb.200708048 Downloaded from jcb.rupress.org on November 7, 2018 JCB: ARTICLE Calpain 2 and PTP1B function in a novel pathway with Src
Published Online: 10 March, 2008 Supp Info: http://doi.org/10.1083/jcb.200708048 Downloaded from jcb.rupress.org on November 7, 2018 JCB: ARTICLE Calpain 2 and PTP1B function in a novel pathway with Src
Tumor microenvironment Interactions and Lung Cancer Invasiveness. Pulmonary Grand Rounds Philippe Montgrain, M.D.
 Tumor microenvironment Interactions and Lung Cancer Invasiveness Pulmonary Grand Rounds Philippe Montgrain, M.D. February 26, 2009 Objectives Review epithelial mesenchymal transition (EMT), and its implications
Tumor microenvironment Interactions and Lung Cancer Invasiveness Pulmonary Grand Rounds Philippe Montgrain, M.D. February 26, 2009 Objectives Review epithelial mesenchymal transition (EMT), and its implications
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
Human SH-SY5Y neuroblastoma cells (A.T.C.C., Manassas, VA) were cultured in DMEM, F-12
 SUPPLEMENTARY METHODS Cell cultures Human SH-SY5Y neuroblastoma cells (A.T.C.C., Manassas, VA) were cultured in DMEM, F-12 Ham with 25 mm HEPES and NaHCO 3 (1:1) and supplemented with 10% (v/v) FBS, 1.0
SUPPLEMENTARY METHODS Cell cultures Human SH-SY5Y neuroblastoma cells (A.T.C.C., Manassas, VA) were cultured in DMEM, F-12 Ham with 25 mm HEPES and NaHCO 3 (1:1) and supplemented with 10% (v/v) FBS, 1.0
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth.
 Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Cancer Biology Course. Invasion and Metastasis
 Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
Cancer Biology Course Invasion and Metastasis 2016 Lu-Hai Wang NHRI Cancer metastasis Major problem: main reason for killing cancer patients, without it cancer can be cured or controlled. Challenging questions:
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
Enterotoxin preconditioning restores calciumsensing receptor-mediated cytostasis in colon cancer cells
 Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 6-19-2008 Enterotoxin
Thomas Jefferson University Jefferson Digital Commons Department of Pharmacology and Experimental Therapeutics Faculty Papers Department of Pharmacology and Experimental Therapeutics 6-19-2008 Enterotoxin
Focus Application. Compound-Induced Cytotoxicity
 xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity For life science research only. Not for use in diagnostic procedures. Featured Study: Using the Time Resolving
xcelligence System Real-Time Cell Analyzer Focus Application Compound-Induced Cytotoxicity For life science research only. Not for use in diagnostic procedures. Featured Study: Using the Time Resolving
NF-κB p65 (Phospho-Thr254)
 Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Quantification of early stage lesions for loss of p53 should be shown in the main figures.
 Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538
 Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
Quantitative Data Analysis Assignment Sample Newessays.co.uk
 Absorbance Quantitative Data Analysis Assignment Sample Newessays.co.uk Part A: Results of the Study Is there a difference of curve profile between the MTT assay and the cell number? What do the different
Absorbance Quantitative Data Analysis Assignment Sample Newessays.co.uk Part A: Results of the Study Is there a difference of curve profile between the MTT assay and the cell number? What do the different
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
genome edited transient transfection, CMV promoter
 Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Cancer Biology Dynamical Cell Systems
 The Institute of Cancer Research PHD STUDENTSHIP PROJECT PROPOSAL PROJECT DETAILS Project Title: SUPERVISORY TEAM Primary Supervisor: The forces behind pancreatic cancer; and changing them as a therapeutic
The Institute of Cancer Research PHD STUDENTSHIP PROJECT PROPOSAL PROJECT DETAILS Project Title: SUPERVISORY TEAM Primary Supervisor: The forces behind pancreatic cancer; and changing them as a therapeutic
Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF
 Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF- 02341066 Assay IC 50 nm Selectivity Ratio d Biochemical Activity In Vitro c-met/hgfr enzyme (Ki, nm) a 4 NA Cellular Activity
Supplemental Table 1. Biochemical and Cellular Potency and Selectivity of PF- 02341066 Assay IC 50 nm Selectivity Ratio d Biochemical Activity In Vitro c-met/hgfr enzyme (Ki, nm) a 4 NA Cellular Activity
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION 60611, USA
 Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
Epithelial cell death is an important contributor to oxidant-mediated acute lung injury SUPPORTING INFORMATION G.R. Scott Budinger 1,2 *, Gökhan M. Mutlu 1 *, Daniela Urich 2, Saul Soberanes 1, Leonard
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function
 ONLINE DATA SUPPLEMENTS Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function Supplementary Figures Figure S1 Effect of Ad-p27-126TS on the expression
ONLINE DATA SUPPLEMENTS Santulli G. et al. A microrna-based strategy to suppress restenosis while preserving endothelial function Supplementary Figures Figure S1 Effect of Ad-p27-126TS on the expression
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
SUPPLEMENTARY INFORMATION Advances in pancreatic islet monolayer culture on glass surfaces enable superresolution microscopy and insights into beta cell ciliogenesis and proliferation Edward A. Phelps,
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
F-actin VWF Vinculin. F-actin. Vinculin VWF
 a F-actin VWF Vinculin b F-actin VWF Vinculin Supplementary Fig. 1. WPBs in HUVECs are located along stress fibers and at focal adhesions. (a) Immunofluorescence images of f-actin (cyan), VWF (yellow),
a F-actin VWF Vinculin b F-actin VWF Vinculin Supplementary Fig. 1. WPBs in HUVECs are located along stress fibers and at focal adhesions. (a) Immunofluorescence images of f-actin (cyan), VWF (yellow),
SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE
 SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE 45 CLINICAL CASES SERIAL DILUTION MULTIPLE PROTEIN CONCENTRATION QUANTITATIVE ASSAY PRODUCT NUMBER: PM1-001-N SOMAPLEX REVERSE
SOMAPLEX REVERSE PHASE PROTEIN MICROARRAY HUMAN KIDNEY TUMOR & NORMAL TISSUE 45 CLINICAL CASES SERIAL DILUTION MULTIPLE PROTEIN CONCENTRATION QUANTITATIVE ASSAY PRODUCT NUMBER: PM1-001-N SOMAPLEX REVERSE
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Production of Exosomes in a Hollow Fiber Bioreactor
 Production of Exosomes in a Hollow Fiber Bioreactor John J S Cadwell, President and CEO, FiberCell Systems Inc INTRODUCTION Exosomes are small lipid membrane vesicles (80-120 nm) of endocytic origin generated
Production of Exosomes in a Hollow Fiber Bioreactor John J S Cadwell, President and CEO, FiberCell Systems Inc INTRODUCTION Exosomes are small lipid membrane vesicles (80-120 nm) of endocytic origin generated
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
DOI: 10.1038/ncb2988 Supplementary Figure 1 Kif7 L130P encodes a stable protein that does not localize to cilia tips. (a) Immunoblot with KIF7 antibody in cell lysates of wild-type, Kif7 L130P and Kif7
Downregulation of angiotensin type 1 receptor and nuclear factor-κb. by sirtuin 1 contributes to renoprotection in unilateral ureteral
 Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Supplementary Information Downregulation of angiotensin type 1 receptor and nuclear factor-κb by sirtuin 1 contributes to renoprotection in unilateral ureteral obstruction Shao-Yu Yang 1,2, Shuei-Liong
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest.
 Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Selective protection of an ARF1-GTP signaling axis by a bacterial scaffold induces bidirectional trafficking arrest. Andrey S. Selyunin, L. Evan Reddick, Bethany A. Weigele, and Neal M. Alto Supplemental
Rabbit Polyclonal antibody to NFkB p65 (v-rel reticuloendotheliosis viral oncogene homolog A (avian))
 Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
Datasheet GeneTex, Inc : Toll Free 1-877-GeneTex (1-877-436-3839) Fax:1-949-309-2888 info@genetex.com GeneTex International Corporation : Tel:886-3-6208988 Fax:886-3-6208989 infoasia@genetex.com Date :
B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small cell lung cancer
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Experimental Methods Cell culture B16-F10 (Mus musculus skin melanoma), NCI-H460 (human non-small
were isolated from the freshly drawn blood of healthy donors and ACS patients using the
 Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Supplemental Figure 1. Quality control of CD4 + T-cell purification. CD4 + T cells were isolated from the freshly drawn blood of healthy donors and ACS patients using the RosetteSep CD4 + T Cell Enrichment
Nature Immunology: doi: /ni.3631
 Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1
 CD31 FN Supplementary Figure 1 a Multivariate Cox regression analysis of predicting factors for disease-free and overall survival in 435 HNSCC patients b FN staining in whole sections of HNSCC c FN expression
CD31 FN Supplementary Figure 1 a Multivariate Cox regression analysis of predicting factors for disease-free and overall survival in 435 HNSCC patients b FN staining in whole sections of HNSCC c FN expression
Impact of NM23-M1 "knock-out" on metastatic potential of hepatocellular carcinoma: a transgenic approach
 Impact of NM23-M1 "knock-out" on metastatic potential of hepatocellular carcinoma: a transgenic approach NM23-M1 "knock- out" mice (without NDPK A) Experimental models of hepatocarcinogenesis Chemical
Impact of NM23-M1 "knock-out" on metastatic potential of hepatocellular carcinoma: a transgenic approach NM23-M1 "knock- out" mice (without NDPK A) Experimental models of hepatocarcinogenesis Chemical
Supplementary Figure (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were
 Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
Supplementary Figure 1. Gd@C 82 (OH) 22 nanoparticles did not affect cell viability and apoposis. MDA-MB-231, MCF-7, MCF-10A and BT549 cells were treated with PBS, Gd@C 82 (OH) 22, C 60 (OH) 22 or GdCl
TITLE: Autocrine and Paracrine Control of Breast Cancer Growth by Sex Hormone-Binding Globulin
 AD Award Number: DAMD17-02-1-0572 TITLE: Autocrine and Paracrine Control of Breast Cancer Growth by Sex Hormone-Binding Globulin PRINCIPAL INVESTIGATOR: William Rosner, M.D. Scott M. Kahn, Ph.D. CONTRACTING
AD Award Number: DAMD17-02-1-0572 TITLE: Autocrine and Paracrine Control of Breast Cancer Growth by Sex Hormone-Binding Globulin PRINCIPAL INVESTIGATOR: William Rosner, M.D. Scott M. Kahn, Ph.D. CONTRACTING
