Supplementary Discussion
|
|
|
- Dominick Boone
- 5 years ago
- Views:
Transcription
1 doi: /nature09513 Supplementary Discussion Time course of CNS vascular development The timing of endothelial cell entry into the rat cerebral cortex and pericyte recruitment are further demonstrated in Supplementary Figure 1. In this figure pericytes are labeled with an antibody directed against Ng2. Ng2 is expressed both in pericytes and OPCs, but here we analyze early embryonic time points from E11-E17 prior to OPC generation at E19 (Fig. 1C), therefore all Ng2 + cells are pericytes. Endothelial cells are first observed on the cortical surface at E11 in a caudal to rostral gradient (Supplementary Fig. 1A-B). Ng2 + cells are also observed on the cortical surface in a similar gradient (Supplementary Fig. 1A-B). Endothelial cells also enter the cortex in a caudal-rostral gradient starting at E12, and these initial sprouts into the CNS are not covered by pericytes (Supplementary Fig. 1C-D). Pericytes are soon recruited to the nascent vessels and are observed to be coating the vessels by E13 and this process continues as vessels continue to be generated (Supplementary Fig. 1E-F). Pericyte cell bodies are closely associated with the endothelial cells, and have processes forming all around the endothelial cells (Supplementary Fig. 1G) Time course of BBB development We further examined the time course of Glut1 and ZO-1 expression in the developing rat cerebral cortex (Supplementary Fig. 3, 4). At E11-12, when endothelial cells are associated with the cortical surface, but have yet to dive into the cortex, the developing neural tissue expresses Glut1 throughout all cells (Supplementary Fig. 3A-C). As endothelial cells enter the cortex, the expression of Glut1 is quickly restricted to the endothelial cells, and lost from the developing neural tissue (Supplementary Fig. 3D-F). Glut1 is expressed in the earliest cells that invade the CNS (Supplementary Fig. 3D). At E11-E12 the tight junction protein ZO-1 is expressed in the epithelial cells in the ependymal layer of the developing cortex (Supplementary Fig. 4A-B). At E12, endothelial cells express ZO-1 as they begin to invade the cortex (Supplementary Fig. 4B). At this early developmental time point ZO-1 staining is patchy throughout the endothelial cell, and then is observed to form complete rings around the cell borders at E15 (Supplementary Fig. 4D). This is consistent with electron microscopy studies which suggest that ultrastructural tight junctions in CNS endothelial cells gain complexity through development 1. Our data also demonstrated that genes associated with increased permeability, including Plvap (transcytosis) and Icam1 (leukocyte adhesion) are initially expressed in endothelial cells as they enter the cerebral cortex, but are down-regulated during development (Supplementary Fig. 7). This is consistent with ultrastructural work which has demonstrated that pinocytotic vesicles are significantly down-regulated during embryonic mouse development 2, and correlates with the pericyte coverage of the vessels. Interestingly, several studies suggest that there may be species specific differences in BBB development. For example, elegant studies in the opossum, a marsupial which is born prior to vascularization of the cerebral cortex, have demonstrated a tight barrier as soon as vessels enter the cortex 3. Furthermore, studies of plasma protein localization in human brain samples also have suggested that the BBB is present at early stages of brain development 4. This is in contrast with data which suggests that fenestrated vessels are present in the developing rodent brain. Our studies, however, suggest that barrier properties are present in rat and mouse vessels as soon as they enter the cerebral cortex, similar to that of humans and opossums. Perhaps a leaky BBB is not conducive to neural development. 1
2 Time Course of Glial Cell Development We have demonstrated that in the rat cerebral cortex OPC generation begins at E18 and astrocyte generation starts at birth. This corresponds with existing literature in which it has been observed that neural stem cells produce neurons and glia in a defined order: first neurons, then oligodendrocyte lineage cells, and then astrocytes 5,6. Furthermore, our work corresponds with existing literature which suggests that astrocytes extend elaborate cellular processes during the first few postnatal weeks that contact both neurons and vessels 5. The fact that we observe BBB properties prior to astrocyte generation suggests that astrocytes cannot be the cell type that induces BBB properties during embryogenesis. Interestingly, there is a striking up-regulation of Pgp that corresponds with astrocyte generation and ensheathment of vessels. Indeed, astrocytes have been demonstrated to induce Pgp expression in endothelial cells 7. Furthermore, there is an increase in trans-endothelial cell electrical resistance during postnatal development in vivo, suggesting that astrocytes may be key to the maturation of the BBB during postnatal development 8. In addition, although we observed tracer leakage in Pdgfrb F7/- and Pdgfrb F7/F7 neonatal mice, no tracer leakage was observed in the adults of these genotypes (data not shown), suggesting that astrocytes can further strengthen the barrier after they ensheath the vessels. A role for astrocytes dynamically regulating the BBB in response to blood contents, neuronal activity and disease remains very likely. Pericyte Coverage in Pdgfrb +/+ and Pdgfrb +/- mice We examined the pericyte coverage of CNS vessels in the cortex of Pdgfrb +/+ and Pdgfrb +/- mice (Supplementary Fig. 8) by Zic1 staining for pericyte nuclei, desmin staining for pericyte cytoskeleton, and Ng2 staining for pericyte cell surface. Tallquist et al 9 have identified that there was a decrease in pericyte number in Pdgfrb +/- mice. By Zic1 staining we also identified that there was a small decrease in pericyte number, however, we have identified that there is comparable coverage of the vascular tubes by Ng2+,desmin+ pericyte processes in both Pdgfrb +/+ and Pdgfrb +/- mice. This suggests that with a small decrease in pericyte number, the remaining pericytes can cover the vasculature to comparable levels. We further demonstrated that there was no difference in the vascular permeability of Pdgfrb +/+ and Pdgfrb +/- mice (Supplementary Fig. 8). Due to the comparable pericyte coverage and vascular permeability, we have combined both Pdgfrb +/+ and Pdgfrb +/- mice as littermate controls for Pdgfrb -/- mice Vascular Permeability in Pdgfrb -/- mice Here we have identified an increase in vascular permeability in Pdgfrb -/- mice that lack CNS pericytes. Hellstrom et al identified that pericyte-deficient mice have increased VEGFA expression, which suggests that the increased permeability may be due to systemic effects of VEGF or other inflammatory molecules including cytokines, and not direct signaling between endothelial cells and pericytes 10. On the other hand, our data demonstrates that co-culture of CNS pericytes with endothelial cells is sufficient to induce BBB properties in CNS endothelial cells, including increased tight junction resistance and inhibition of Icam1 and Angpt2 expression. Therefore, the increased vascular permeability observed in 2
3 the pericyte-deficient mice may be due to a combination of systemic metabolic differences and direct pericyte-endothelial cell signaling. Whereas Hellstrom et al did not analyze BBB function in the Pdgfrb -/- mice, they did demonstrate an increase in membrane folding and vesicular trafficking that was similar to our results. They further demonstrated an increase in caveolin-1 and a redistribution of occludin strands in the Pdgfrb -/- mice 10. We did not observe these defects in our analysis of Pdgfrb -/- mice, which suggests that other factors may influence vascular permeability in these mice including strain and environmental conditions. Interestingly, we observed heterogeneity in Gr1+ leukocyte infiltration in different Pdgfrb F7/F7 mice suggesting that additional environmental factors may be important for leukocyte infiltration, in addition to the increase in Icam1 expression in endothelial cells. Hellstrom et al also identified an increase in Glut1 and Pgp expression in the Pdgfb -/- mice, which lack the PDGF-B ligand and are also pericyte-deficient. They suggest that this may indicate metabolic stress 10. This paper did not analyze these genes in Pdgfrb -/- mice, and we did not observe an increase in either Glut1 or Pgp. This may also indicate strain or environmental differences, or perhaps that PDGF-B has more effects than just signaling through PDGFR-β. Identification of Pericyte-Derived Factors To determine the pericyte transcriptome we performed microarray analysis on mrna isolated from acutely purified CNS pericytes and compared these values to the microarray analysis performed on purified endothelial cells from the Pdgfrb -/- mice which completely lack pericytes. This analysis produced a dataset of pericyte-expressed molecules (Supplementary Table 2). Interestingly, pericytes express genes that are known to induce BBB properties including Angpt1 (as well as the homologous gene Angptl2), and Ace2 11,12. Ace2 is an enzyme involved in the processing of Agt to smaller peptides, which regulate BBB permeability 12. Several other interesting secreted molecules are expressed by endothelial cells, including Bmp5, nodal, netrin1, sema5a, as well as integrins and plexins. Furthermore, we have identified that pericytes express a number of extracellular matrix molecules, including collagens, laminins, vitronectin, asporin and others. We have further analyzed the protein expression of these ECM molecules in Pdgfrb -/- mice and littermate controls (Supplementary Fig. 11) and have identified that there are lower levels of collagens in Pdgfrb -/- mice, but strikingly an increase in vitronectin and MMP9. The altered composition of the endothelial ECM may be important in regulating BBB properties. Specifically, expression of MMP9 has been shown to lead to BBB breakdown in pathological conditions 13,
4 Supplementary References 1. Kniesel, U., Risau, W. & Wolburg, H. Development of blood-brain barrier tight junctions in the rat cortex. Brain Res Dev Brain Res 96, (1996). 2. Bauer, H. C. et al. Neovascularization and the appearance of morphological characteristics of the blood-brain barrier in the embryonic mouse central nervous system. Brain Res Dev Brain Res 75, (1993). 3. Ek, C. J., Dziegielewska, K. M., Stolp, H. & Saunders, N. R. Functional effectiveness of the bloodbrain barrier to small water-soluble molecules in developing and adult opossum (Monodelphis domestica). J Comp Neurol 496, (2006). 4. Mollgard, K., Dziegielewska, K. M., Saunders, N. R., Zakut, H. & Soreq, H. Synthesis and localization of plasma proteins in the developing human brain. Integrity of the fetal blood-brain barrier to endogenous proteins of hepatic origin. Dev Biol 128, (1988). 5. Bushong, E. A., Martone, M. E. & Ellisman, M. H. Maturation of astrocyte morphology and the establishment of astrocyte domains during postnatal hippocampal development. Int J Dev Neurosci 22, (2004). 6. Cahoy, J. D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J Neurosci 28, (2008). 7. Gaillard, P. J. et al. Astrocytes increase the functional expression of P-glycoprotein in an in vitro model of the blood-brain barrier. Pharm Res 17, (2000). 8. Butt, A. M., Jones, H. C. & Abbott, N. J. Electrical resistance across the blood-brain barrier in anaesthetized rats: a developmental study. J Physiol 429, (1990). 9. Tallquist, M. D., French, W. J. & Soriano, P. Additive effects of PDGF receptor beta signaling pathways in vascular smooth muscle cell development. PLoS Biol 1, E52 (2003). 10. Hellstrom, M. et al. Lack of pericytes leads to endothelial hyperplasia and abnormal vascular morphogenesis. J Cell Biol 153, (2001). 11. Hori, S., Ohtsuki, S., Hosoya, K., Nakashima, E. & Terasaki, T. A pericyte-derived angiopoietin-1 multimeric complex induces occludin gene expression in brain capillary endothelial cells through Tie-2 activation in vitro. J Neurochem 89, (2004). 12. Wosik, K. et al. Angiotensin II controls occludin function and is required for blood brain barrier maintenance: relevance to multiple sclerosis. J Neurosci 27, (2007). 13. Gidday, J. M. et al. Leukocyte-derived matrix metalloproteinase-9 mediates blood-brain barrier breakdown and is proinflammatory after transient focal cerebral ischemia. Am J Physiol Heart Circ Physiol 289, H (2005). 14. He, Z. J., Huang, Z. T., Chen, X. T. & Zou, Z. J. Effects of matrix metalloproteinase 9 inhibition on the blood brain barrier and inflammation in rats following cardiopulmonary resuscitation. Chin Med J (Engl) 122, (2009). 4
5 Gene Symbol Probe Set Control PDGFRβ -/- Fold Change Pan-Endothelial cadherin 5 (VE-Cadherin) Cdh _at endothelial-specific receptor tyrosine kinase (Tie2) Tek _at Tight Junction claudin 5 Cldn _at occludin Ocln _at tight junction protein 1 (ZO-1) Tjp _a_at Influx Transporters solute carrier family 2, member 1 (Glut-1) Slc2a _a_at solute carrier family 7, member 1 (Cat1) Slc7a _at solute carrier family 7, member 5 (TA1) Slc7a _at solute carrier family 1, member 1 (EAAC1) Slc1a _a_at solute carrier family 38, member 5 (JM24) Slc38a _at solute carrier family 16, member 1 (MCT1) Slc16a _at Efflux Transporters ATP-binding cassette, sub-family B, member 1A (Pgp) Abcb1a _at ATP-binding cassette, sub-family G, member 2 Abcg _at Permeability Modulators angiopoietin 1 Angpt _at angiopoietin 2 Angpt _at plasmalemma vesicle associated protein Plvap _at Leukocyte Adhesion mucosal vascular addressin cell adhesion molecule 1 Madcam _a_at lectin, galactose binding, soluble 3 Lgals _at intercellular adhesion molecule Icam _at activated leukocyte cell adhesion molecule Alcam _at Supplementary Table 1: Gene expression in Pdgfrb -/- mice Affymetrix microarrays were used to analyze mrna expression levels in purified CD31 + vascular cells from Pdgfrb -/- mice and litter mate controls. Selected genes are listed with gene expression data from Pdgfrb -/- mice and litter-mate controls. Direction of fold change are given if >2. Pan-endothelial genes, tight junction molecules, influx transporters, efflux transporters displayed unaltered gene expression, whereas the vasculature of Pdgfrb -/- mice displayed an increase in factors that increase vascular permeability and leukocyte adhesion. 5
6 Gene Symbol Probe set Pericytes Endothelial cells Pericyte/ Endothelial Cells Pericyte Markers platelet derived growth factor receptor beta Pdgfrb _a_at chondroitin sulfate proteoglycan 4 Cspg _at desmin Des _at regulator of G-protein signaling 5 Rgs _at K+ inwardly-rectifying channel, subfamily J, member 8 Kcnj _at ATP-binding cassette, sub-family C, member 9 Abcc _s_at Extracellular Matrix procollagen, type VI, alpha 2 Col6a _a_at procollagen, type III, alpha 1 Col3a _a_at procollagen, type I, alpha 2 Col1a _a_at procollagen C-endopeptidase enhancer protein Pcolce _a_at laminin gamma 3 Lamc _at laminin, alpha 2 Lama _at vitronectin Vtn _a_at extracellular matrix protein 2 Ecm _at dentin matrix protein 1 Dmp _s_at asporin Aspn _s_at Signaling Factors angiotensin I converting enzyme 2 Ace _at angiopoietin 1 Angpt _at angiopoietin-like 2 Angptl _at bone morphogenetic protein 5 Bmp _at integrin alpha 8 Itga _at integrin beta 5 Itgb _a_at leucine rich repeat containing 16 Lrrc16a _a_at leucine rich repeat containing 4C Lrrc4c _at nodal Nodal _at Notch gene homolog 3 Notch _s_at netrin 1 Ntn _at plexin domain containing 1 Plxdc _at plexin domain containing 2 Plxdc _at retinoic acid receptor responder 2 Rarres _s_at syndecan 2 Sdc _at semaphorin 5A Sema5a _at superoxide dismutase 3, extracellular Sod _at Supplementary Table 2: Gene expression of acutely purified CNS pericytes Affymetrix microarrays were used to analyze mrna expression levels in purified PDGFR-β + pericytes from P7 mice. Selected genes are listed with gene expression data from acutely purified pericytes, and endothelial cells from Pdgfrb -/- mice for a negative control. Molecules were selected due to high ratio of expression in acutely purified pericytes to endothelial cells. Known pericyte markers, extracellular matrix proteins and signaling factors are listed. 6
7 E17 E13 E12-caudal E12-rostral E11-caudal E11-rostral Ng2 BSL Merge Ai i Bi i Ci i Di i Ei i Fi i Supplementary Figure 1 7
8 Gi Ng2 BSL E17 i Ng2 BSL Supplementary Figure 1 Cont d Supplementary Figure 1: Time course of vascular cell generation in the rat cerebral cortex A-F) Tissue sections of rat cerebral cortex at indicated ages were stained for endothelial cells with BSL (green A-F, i) and pericytes with anti-ng2 (red, A-Fi, i). Endothelial cells and pericytes are first observed at the cortical surface in a caudal to rostral gradient starting at E11, and the first endothelial cells enter the neural tissue at E12, also in a caudal to rostral gradient. Although initial endothelial sprouts entering the CNS do not have pericytes, pericytes are quickly recruited and are observed covering the endothelial cells by E13. The endothelial cells continue to vascularize the brain as the brain grows. Scale bar represents 100 µm. G) High magnification image of E17 rat CNS vessel stained for endothelial cells with BSL (green G, i) and pericytes with anti-ng2 (red, Gi, i). Pericytes extend cellular processes that cover the endothelial cells. Scale bars represent 50 µm. 8
9 P5 P1 E21 A B C BSL / Aquaporin 4 Supplementary Figure 2: Astrocyte endfeet associate with endothelial cells during postnatal development High magnification images of tissue sections of rat cerebral cortex at E21 (A), P1 (B) and P5 (C) were stained for endothelial cells with BSL (green A-C) and astrocytes with anti-aquaporin 4 (red, A-C). Astrocytes are not observed during embryogenesis, but extend processes which touch endothelial cells at P1 and start to wrap around the endothelial cells by P5. Arrows indicate astrocyte processes contacting endothelial cells. Scale bar represents 100 µm. 9
10 E17 E13 E12-caudal E12-rostral E11-caudal E11-rostral Glut-1 DAPI / BSL Merge Ai i Bi i Ci i Di i Ei i Fi i Supplementary Figure 3: Time course of Glut1 expression in the rat cerebral cortex A-F) Tissue sections of rat cerebral cortex at indicated ages were stained for endothelial cells with BSL (green A-F, i) and with anti-glut1 (red, A-Fi, i). Prior to endothelial cell invasion into the cortex, Glut1 is expressed throughout the developing neural tissue. After endothelial cells invade the cortex, Glut1 expression is quickly restricted to the endothelial cells. Scale bar represents 100 µm. 10
11 E15 E13 E12-caudal E12-rostral zo-1 BSL Merge Ai i Bi i Ci i Di i Supplementary Figure 4: Time course of ZO-1 expression in the rat cerebral cortex A-D) Tissue sections of rat cerebral cortex at indicated ages were stained for endothelial cells with BSL (green A-F, i) and with anti-zo-1 (red, A-Fi, i). Prior to endothelial cell invasion into the cortex, ZO-1 is expressed specifically in epithelial cells in the ependymal cell layer (yellow arrows), and continues to be expressed in these cells throughout development and maturity (data not shown). As endothelial cells invade the cortex at E12, ZO-1 expression is patchy within endothelial cells (B -white arrows), and then is observed to form complete junctions by E15 (D -white arrows). Scale bar represents 50 µm. 11
12 A Cereberal Cortex B Sub-cortical Forebrain e15 Supplementary Figure 5: Spatial distribution of tracer leakage in embryos E15 rats were given a transcardiac perfusion of a biotin tracer, and tissue sections were stained with streptavidin (green) and the nuclear marker DAPI (blue). Vessels restrict the movement of the biotin tracers into the CNS parenchyma (B), however leakage is observed near the pial surface (A). Scale bar represents 100 µm. 12
13 BSL DAPI Ai E10 E11 i E13 BSL PDGFRβ Bi E10 E11 i E13 BSL AQ4 Ci E18 P2 i P7 BSL zo-1 Di E11 E13 i E18 Ei E10 E11 i i E13 Glut-1 Supplementary Figure 6: Time course of cell generation and BBB properties during mouse development A-E) Tissue sections of mouse cerebral cortex at indicated ages were stained for endothelial cells with BSL (green A-D) and with DAPI (blue, A), anti-pdgfr-β (red, B), anti-aquaporin 4 (red, C), anti-zo-1 (red, D), or anti-glut1 (red, E). Endothelial cells enter the cortex starting at E11, and continue to vascularize the CNS during development. PDGFR-β + pericytes cover endothelial cells as soon as they enter the cortex, whereas astrocytes are only generated after birth. CNS endothelial cells express ZO-1 and Glut1 as they enter the cortex. Scale bars represent 100 µm (A,C,E), 50 µm (B) and 20 µm (D) 13
14 E11 E12 E18 Ai Bi Ci Icam1 BSL PLVAP Icam1 Di Ei Fi PLVAP BSL Supplementary Figure 7: Time course of Icam1 and Plvap expression during mouse development A-C) Tissue sections of mouse cerebral cortex at indicated ages were stained for endothelial cells with BSL (green A-F) and with anti-icam1 (red, A-C) or anti-plvap (red, D-F). Endothelial cells express both Icam1 and Plvap at early embryonic timepoints, but then down-regulate both during embryonic development. Scale bars represent 100 µm. 14
15 PDGFRβ +/+ PDGFRβ +/- Ai Bi Ci PDGFRβ -/- Zic1 BSL Di Ei Fi Ng2 BSL Desmin BSL Gi Hi Ii Supplementary Figure 8 Desmin Ji PDGFRβ +/+ PDGFRβ +/- Ki Ng2 PDGFRβ +/+ PDGFRβ +/- Li Mi Desmin BSL Ng2 BSL N Pericyte Number (% wild type) O Pericyte Coverage (% wild type) Streptavidin P PDGFRβ +/+ PDGFRβ +/- Q R Permeability (% wild type) Supplementary Figure 8 Cont d 15
16 Supplementary Figure 8: Pericyte coverage of CNS vessels in Pdgfrb +/+ and Pdgfrb +/- mice A-I) E15 (G-I) and E18 (A-F) mouse cerebral cortex from Pdgfrb +/+ (A,D,G), Pdgfrb +/- (B,E,H) and Pdgfrb -/- (C,F,I) mice were stained for endothelial cells with BSL (green A-I,) and pericytes with anti-zic (red, A- C), anti-desmin (red D-F), and anti-ng2 (red, G-I). Pericytes are present in the Pdgfrb +/+ and Pdgfrb +/- mice but not the Pdgfrb -/-. Note that the intense desmin staining in the cortex of Pdgfrb -/- mice is due to the fact that the anti-desmin primary antibody is a mouse monoclonal, and therefore the staining is due to the fluorescently labeled goat anti-mouse secondary antibody binding to endogenous mouse antibody that has leaked into the brain. Scale bars represent 100 µm (A-F) and 200 µm (G-I). J-M) High power magnification of pericyte coverage in mouse cerebral cortex from Pdgfrb +/+ and Pdgfrb +/- mice. E15 (L,M) and neonatal (J,K) mouse cerebral cortex from Pdgfrb +/+ (J,L), and Pdgfrb +/- (K,M) mice were stained for endothelial cells with BSL (green J-M ) and pericytes with anti-desmin (red, J,K), and anti-ng2 (red L,M). Scale bars represent 50 µm. N) Pericyte number in Pdgfrb +/+ and Pdgfrb +/- mice was quantified by analyzing number of Zic1+ nculei per length of BSL + vessels. All error bars represent s.e.m. O) Pericyte coverage of CNS vessels in Pdgfrb +/+ and Pdgfrb +/- mice was quantified by analyzing percent length of BSL + vessels opposed to desmin + pericytes. Comparable pericyte coverage was observed in both Pdgfrb +/+ and Pdgfrb +/- mice. All error bars represent s.e.m. P-R) Neonatal Pdgfrb +/+ (P) and Pdgfrb +/- (Q) mice, were given a transcardiac perfusion of a biotin tracer, and tissue sections were stained with streptavidin (green). Scale bar represents 200 µm. R) Fluorescence was quantified in Image J, and permeability relative to control was graphed. All error bars represent s.e.m. No increase in staining for tracer was observed in CNS parenchyma in the Pdgfrb +/- mice. 16
17 Ai Control PDGFRβ -/- Bi Merge BSL Glut-1 i i Ci Di Glut-1 CD31 i i Supplementary Figure 9: Glut1 Expression and Localization in Pdgfrb -/- mice E18 Pdgfrb -/- mice (B, D), and litter mate controls (A,C) were stained with endothelial marker BSL (green A,B,,i,) or anti-cd31 (C,D i,i) and anti-glut1 (red A-Di,i, E,F). A-B) low magnification images depicting overall expression of Glut1 is not affected in the vessels throughout the Pdgfrb -/- mice cerebral cortex. Scale bars represent 100 µm (A,B). (C-D) High magnification images demonstrating that the polarized abluminal localization of Glut1 is not affected in CNS vessels in Pdgfrb -/- mice. Scale bars represent 20 µm. 17
18 Plvap Plvap / BSL Ai PDGFR -/- Control Bi Ci Di Supplementary Figure 10: Plvap expression in Pdgfrb -/- mice E18 Pdgfrb -/- mice (C,D), and litter mate controls (A,B) were stained with anti-plvap (red in i,) and the endothelial marker BSL (green, ). An increase in Plvap staining intensity and the number of positive vessels was observed in the Pdgfrb -/- mice. Scale bar represents 50 µm. 18
19 A PDGFRβ N/N N/+ N/N N/+ Col III Col I Vitronectin MMP-9 PDGFRb B-actin B Relative Expression (AU) * * * * * Control Mutant Col3 Col1 Vitronectin MMP9 PDGFRb Beta actin Supplementary Figure 11: Extracellular matrix in Pdgfrb -/- mice A) Expression levels of extracellular molecules in Pdgfrb -/- mice. Western blots on brain lysates from E18 Pdgfrb -/- and litter mate heterozygote controls, probing for extracellular matrix molecules Col III, Col I, Vitronectin, MMP-9, as well as PDGFR-β and beta-actin. Collagens showed decreased protein expression whereas vitronectin and MMP-9 showed an increase in protein expression in the Pdgfrb -/- brain. B) Quantification of extracellular matrix molecules in Pdgfrb -/- mice and littermate controls. * P<0.05 by Students t-test. All error bars represent s.e.m. 19
20 Ai Endothelial cells Endothelial cells + Pericytes Bi DAPI Angpt2 Angpt2 Supplementary Figure 12: Pericytes inhibit endothelial Angpt2 expression in vitro Purified murine brain endothelial cells were grown in culture alone (A) or with a feeding layer of purified brain pericytes (B) and stained for DAPI (blue, ) and anti-angpt2 (green). Scale bar represents 100µm. 20
21 Legend Cells Endothelial cells Neural progenitors Pericytes Astrocyte endfeet Molecules Tight Junction Proteins Nutrient transporters Efflux transporters Leukocyte adhesion molecules Transcytotic vesicles Supplementary Figure 13: Model for cellular interactions that regulate blood-brain barrier formation During embryonic angiogenesis neural progenitors induce endothelial cells to express blood-brain barrier specific proteins, including tight junction molecules and specific nutrient transporters. Pericytes then strengthen the endothelial barrier during embryogenesis by regulating tight junction structure, limiting the rate of transcytosis, and inhibiting the expression of leukocyte adhesion molecules. Astrocytes further aid in regulating the function of the barrier during adulthood, injury and disease. 21
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
www.sciencetranslationalmedicine.org/cgi/content/full/4/117/117ra8/dc1 Supplementary Materials for Notch4 Normalization Reduces Blood Vessel Size in Arteriovenous Malformations Patrick A. Murphy, Tyson
Tissue repair. (3&4 of 4)
 Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Tissue repair (3&4 of 4) What will we discuss today: Regeneration in tissue repair Scar formation Cutaneous wound healing Pathologic aspects of repair Regeneration in tissue repair Labile tissues rapid
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Tissue renewal and Repair. Nisamanee Charoenchon, PhD Department of Pathobiology, Faculty of Science
 Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
Tissue renewal and Repair Nisamanee Charoenchon, PhD Email: nisamanee.cha@mahidol.ac.th Department of Pathobiology, Faculty of Science Topic Objectives 1. Describe processes of tissue repair, regeneration
Supplemental Table S1. Primers used in qrt-pcr analyses. Supplemental Figure S1, related to Figure 4. Extracellular matrix proteins
 Supplemental Material PDGFRb regulates craniofacial development through homodimers and functional heterodimers with PDGFRa Katherine A. Fantauzzo and Philippe Soriano Supplemental materials provided: Supplemental
Supplemental Material PDGFRb regulates craniofacial development through homodimers and functional heterodimers with PDGFRa Katherine A. Fantauzzo and Philippe Soriano Supplemental materials provided: Supplemental
Supplementary Information
 Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
Supplementary Information Title Degeneration and impaired regeneration of gray matter oligodendrocytes in amyotrophic lateral sclerosis Authors Shin H. Kang, Ying Li, Masahiro Fukaya, Ileana Lorenzini,
The blood-brain barrier: barrier or customs border?
 The blood-brain barrier: barrier or customs border? Jerome Badaut, Phd Brain Molecular Imaging group Badaut et al. 2000 Neuro (green) vascular (red) unit (NVU) (Obenaus, Coats, Krucker) (brain s vascular
The blood-brain barrier: barrier or customs border? Jerome Badaut, Phd Brain Molecular Imaging group Badaut et al. 2000 Neuro (green) vascular (red) unit (NVU) (Obenaus, Coats, Krucker) (brain s vascular
Brain Development III
 Brain Development III Neural Development In the developing nervous system there must be: 1. The formation of different regions of the brain. 2. The ability of a neuron to differentiate. 3. The ability
Brain Development III Neural Development In the developing nervous system there must be: 1. The formation of different regions of the brain. 2. The ability of a neuron to differentiate. 3. The ability
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/487/eaag2476/dc1 Supplementary Materials for Gene expression profiles of brain endothelial cells during embryonic development at bulk and single-cell levels
www.sciencesignaling.org/cgi/content/full/10/487/eaag2476/dc1 Supplementary Materials for Gene expression profiles of brain endothelial cells during embryonic development at bulk and single-cell levels
What Cell Make Up the Brain and Spinal Cord
 What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
Cellular components of CNS
 Cellular components of CNS Cellular components of CNS Neurons Glial cells: Astrocytes (including radial glia), oligodendrocytes, microglia, ependymal cells Epithelial cells of choroid plexus Endothelial
Cellular components of CNS Cellular components of CNS Neurons Glial cells: Astrocytes (including radial glia), oligodendrocytes, microglia, ependymal cells Epithelial cells of choroid plexus Endothelial
SUPPLEMENTARY FIG. S2. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of dnscs.
 Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Nature Neuroscience: doi: /nn Supplementary Figure 1. PICALM expression in brain capillary endothelium in human brain and in mouse brain.
 Supplementary Figure 1 PICALM expression in brain capillary endothelium in human brain and in mouse brain. a, Double immunostaining for PICALM (red, left) and lectin positive endothelial profiles (blue,
Supplementary Figure 1 PICALM expression in brain capillary endothelium in human brain and in mouse brain. a, Double immunostaining for PICALM (red, left) and lectin positive endothelial profiles (blue,
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Table 1. Genes analysed for expression by angiogenesis gene-array.
 Supplementary Table 1. Genes analysed for expression by angiogenesis gene-array. Gene symbol Gene name TaqMan Assay ID UniGene ID 18S rrna 18S ribosomal RNA Hs99999901_s1 Actb actin, beta Mm00607939_s1
Supplementary Table 1. Genes analysed for expression by angiogenesis gene-array. Gene symbol Gene name TaqMan Assay ID UniGene ID 18S rrna 18S ribosomal RNA Hs99999901_s1 Actb actin, beta Mm00607939_s1
label the basement membrane). Different fixation methods of EB-perfused P8 mice to optimize the combination
 Supplementary Figure 1 Optimization of the tissue fixation protocol to combine EB perfusion and IB4 endothelial tip cell staining in the postnatal mouse brain. a-l Labeling of EB-perfused P8 mice with
Supplementary Figure 1 Optimization of the tissue fixation protocol to combine EB perfusion and IB4 endothelial tip cell staining in the postnatal mouse brain. a-l Labeling of EB-perfused P8 mice with
The Angiopoietin Axis in Cancer
 Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Diagram of BBB and brain chips.
 Supplementary Figure 1 Diagram of BBB and brain chips. (a) Schematic of the BBB Chip demonstrates the 3 parts of the chip, Top PDMS channel, membrane and Bottom PDMS channel; (b) Image of 2 BBB Chips,
Supplementary Figure 1 Diagram of BBB and brain chips. (a) Schematic of the BBB Chip demonstrates the 3 parts of the chip, Top PDMS channel, membrane and Bottom PDMS channel; (b) Image of 2 BBB Chips,
B16F1 B16F10 Supplemental Figure S1
 B16F1 B16F1 Supplemental Figure S1 Representative microangiography images of B16F1 and B16F1 tumors grown in the cranial windows. FITC-dextran (2 million MW) was injected systemically to visualize blood
B16F1 B16F1 Supplemental Figure S1 Representative microangiography images of B16F1 and B16F1 tumors grown in the cranial windows. FITC-dextran (2 million MW) was injected systemically to visualize blood
CELL BIOLOGY - CLUTCH CH CELL JUNCTIONS AND TISSUES.
 !! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
!! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/385/ra70/dc1 Supplementary Materials for The interaction of heparan sulfate proteoglycans with endothelial transglutaminase-2 limits VEGF 165 -induced angiogenesis
www.sciencesignaling.org/cgi/content/full/8/385/ra70/dc1 Supplementary Materials for The interaction of heparan sulfate proteoglycans with endothelial transglutaminase-2 limits VEGF 165 -induced angiogenesis
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/8/e1700521/dc1 Supplementary Materials for Functional vascularized lung grafts for lung bioengineering N. Valerio Dorrello, Brandon A. Guenthart, John D. O Neill,
advances.sciencemag.org/cgi/content/full/3/8/e1700521/dc1 Supplementary Materials for Functional vascularized lung grafts for lung bioengineering N. Valerio Dorrello, Brandon A. Guenthart, John D. O Neill,
Thomas HAIDER Journal Club
 Thomas HAIDER Journal Club 20.10.2014 Background Immunology of the CNS - History Ehrlich, 1885 & 1904 dye did not stain brain -> BBB Shirai, Y. (1921) On the transplantation of the rat sarcoma in adult
Thomas HAIDER Journal Club 20.10.2014 Background Immunology of the CNS - History Ehrlich, 1885 & 1904 dye did not stain brain -> BBB Shirai, Y. (1921) On the transplantation of the rat sarcoma in adult
Boucher et al NCOMMS B
 1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
1 Supplementary Figure 1 (linked to Figure 1). mvegfr1 constitutively internalizes in endothelial cells. (a) Immunoblot of mflt1 from undifferentiated mouse embryonic stem (ES) cells with indicated genotypes;
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
DOI: 10.1038/ncb3021 Supplementary figure 1 Characterisation of TIMPless fibroblasts. a) Relative gene expression of TIMPs1-4 by real time quantitative PCR (RT-qPCR) in WT or ΔTimp fibroblasts (mean ±
Angiogenesis in Human Development. Vascular Development
 Angiogenesis in Human Development Jan Kitajewski ICRC 217B, ph 851-4688, email: jkk9 BACKGROUND READING: Vascular Development Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan. Science 277:
Angiogenesis in Human Development Jan Kitajewski ICRC 217B, ph 851-4688, email: jkk9 BACKGROUND READING: Vascular Development Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan. Science 277:
Development of Quantitative Tools for the Characterization and Analysis of the Blood Brain Barrier in Normal and MPS IIIB Mice
 Dominican Scholar Master's Theses and Capstone Projects Theses and Capstone Projects 2015 Development of Quantitative Tools for the Characterization and Analysis of the Blood Brain Barrier in Normal and
Dominican Scholar Master's Theses and Capstone Projects Theses and Capstone Projects 2015 Development of Quantitative Tools for the Characterization and Analysis of the Blood Brain Barrier in Normal and
Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants
 SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
SUPPLEMENTAL FIGURE LEGENDS: Supplemental Figure 1: Leydig cells are reduced at multiple stages in both male sterile mutants (Sgpl1 -/- and Plekha1 -/- ). Using an antibody against CYP11a1 to label Leydig
Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System
 Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products
 stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
Signaling Vascular Morphogenesis and Maintenance
 Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Signaling Vascular Morphogenesis and Maintenance Douglas Hanahan Science 277: 48-50, in Perspectives (1997) Blood vessels are constructed by two processes: vasculogenesis, whereby a primitive vascular
Astrocytic laminin regulates pericyte differentiation and maintains blood brain barrier integrity
 RTICL Received 1 Oct 13 ccepted 1 Feb 14 ublished 3 Mar 14 strocytic laminin regulates pericyte differentiation and maintains blood brain barrier integrity Yao Yao 1, Zu-Lin Chen 1, rin H. Norris 1 & Sidney
RTICL Received 1 Oct 13 ccepted 1 Feb 14 ublished 3 Mar 14 strocytic laminin regulates pericyte differentiation and maintains blood brain barrier integrity Yao Yao 1, Zu-Lin Chen 1, rin H. Norris 1 & Sidney
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
Inner ear development Nervous system development
 Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
Upcoming Sessions April 22: Nervous System Development Lecture April 24: Reviews of Axonal Pathfinding in Sensory Systems April 29: Inner Ear Development Lecture May 1: May 6: May 8: Auditory System Pathfinding
ground and enzymatically digested or mechanically homogenized. The EVs or total vesicles were
 Supplementary Figure 1. Characterization of EVs from intestine. (a, b) Intestinal tissues were ground and enzymatically digested or mechanically homogenized. The EVs or total vesicles were isolated by
Supplementary Figure 1. Characterization of EVs from intestine. (a, b) Intestinal tissues were ground and enzymatically digested or mechanically homogenized. The EVs or total vesicles were isolated by
Contact: Course outline: Contact for other times.
 Contact: kdelaney@uvic.ca Course outline: http://web.uvic.ca/~kdelaney/b367 Scheduled office hours: 1:00-3:00, M&Th Cunn. 259A Contact kdelaney@uvic.ca for other times. Quiz (0.5 hrs) midterm (1.4 hrs)
Contact: kdelaney@uvic.ca Course outline: http://web.uvic.ca/~kdelaney/b367 Scheduled office hours: 1:00-3:00, M&Th Cunn. 259A Contact kdelaney@uvic.ca for other times. Quiz (0.5 hrs) midterm (1.4 hrs)
Supplementary Figure 1.
 Supplementary Figure 1. Increased expression of cell cycle pathway genes in insulin + Glut2 low cells of STZ-induced diabetic islets. A) random blood glucose measuers of STZ and vehicle treated MIP-GFP
Supplementary Figure 1. Increased expression of cell cycle pathway genes in insulin + Glut2 low cells of STZ-induced diabetic islets. A) random blood glucose measuers of STZ and vehicle treated MIP-GFP
CD146 coordinates brain endothelial cell pericyte communication for blood brain barrier development
 CD146 coordinates brain endothelial cell pericyte communication for blood brain barrier development Jianan Chen a,b,1, Yongting Luo c,1, Hui Hui d, Tanxi Cai a, Hongxin Huang e, Fuquan Yang a, Jing Feng
CD146 coordinates brain endothelial cell pericyte communication for blood brain barrier development Jianan Chen a,b,1, Yongting Luo c,1, Hui Hui d, Tanxi Cai a, Hongxin Huang e, Fuquan Yang a, Jing Feng
Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo
 Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Behavioural effects of ketamine in non-stressed and stressed mice. Naive C57BL/6 adult male mice (n=10/group) were given a single dose of saline vehicle or ketamine (3.0 mg/kg,
Supplementary Figure 1. Behavioural effects of ketamine in non-stressed and stressed mice. Naive C57BL/6 adult male mice (n=10/group) were given a single dose of saline vehicle or ketamine (3.0 mg/kg,
VWF other roles than hemostasis. Summary 1: VWF & hemostasis synthesis 11/4/16. Structure/function relationship & functions kDa.
 VWF other roles than hemostasis Len$ng PJ, Casari C et al JTH 2012 Summary 1: VWF & hemostasis synthesis Structure/function relationship & functions (HMWM) 20.000kDa multimerization propeptide FVIII GPIb
VWF other roles than hemostasis Len$ng PJ, Casari C et al JTH 2012 Summary 1: VWF & hemostasis synthesis Structure/function relationship & functions (HMWM) 20.000kDa multimerization propeptide FVIII GPIb
Stepwise Recruitment of Transcellular and Paracellular Pathways Underlies Blood-Brain Barrier Breakdown in Stroke
 Article Stepwise Recruitment of Transcellular and Paracellular Pathways Underlies Blood-Brain Barrier Breakdown in Stroke Daniel Knowland, 1,8 Ahmet Arac, 2,3,8 Kohei J. Sekiguchi, 4,5 Martin Hsu, 1 Sarah
Article Stepwise Recruitment of Transcellular and Paracellular Pathways Underlies Blood-Brain Barrier Breakdown in Stroke Daniel Knowland, 1,8 Ahmet Arac, 2,3,8 Kohei J. Sekiguchi, 4,5 Martin Hsu, 1 Sarah
Nature Methods: doi: /nmeth.4257
 Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Supplementary Figure 1 Screen for polypeptides that affect cellular actin filaments. (a) Table summarizing results from all polypeptides tested. Source shows organism, gene, and amino acid numbers used.
Macmillan Publishers Limited. All rights reserved
 TTR doi:1.138/nature13324 Mfsd2a is critical for the formation and function of the blood brain barrier Ayal Ben-Zvi 1, Baptiste acoste 1, sther Kur 1, Benjamin J. Andreone 1, Yoav Mayshar 2, Han Yan 1
TTR doi:1.138/nature13324 Mfsd2a is critical for the formation and function of the blood brain barrier Ayal Ben-Zvi 1, Baptiste acoste 1, sther Kur 1, Benjamin J. Andreone 1, Yoav Mayshar 2, Han Yan 1
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Cell Migration II: CNS Cell Migration. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Oct 18) 9:00-10:00am Surdyk s Café in Northrop Auditorium Stop
Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Oct 18) 9:00-10:00am Surdyk s Café in Northrop Auditorium Stop
Connective Tissue Response in IBD
 Connective Tissue Response in IBD Dr I C Lawrance MB BS, PhD FRACP School of Medicine and Pharmacology, University of Western Australia, Fremantle Hospital Intestinal response to Chronic Inflammation Control
Connective Tissue Response in IBD Dr I C Lawrance MB BS, PhD FRACP School of Medicine and Pharmacology, University of Western Australia, Fremantle Hospital Intestinal response to Chronic Inflammation Control
Cells of the nervous system
 Neurobiology Cells of the nervous system Anthony Heape 2011 1 Cells of the nervous system Neuroglia : part 2 The non excitable cells of the nervous system that provide support to neuronal survival and
Neurobiology Cells of the nervous system Anthony Heape 2011 1 Cells of the nervous system Neuroglia : part 2 The non excitable cells of the nervous system that provide support to neuronal survival and
Neurovascular Unit: a Focus on Pericytes
 DOI 10.1007/s12035-012-8244-2 Neurovascular Unit: a Focus on Pericytes Inês Sá-Pereira & Dora Brites & Maria Alexandra Brito Received: 22 November 2011 / Accepted: 1 February 2012 # Springer Science+Business
DOI 10.1007/s12035-012-8244-2 Neurovascular Unit: a Focus on Pericytes Inês Sá-Pereira & Dora Brites & Maria Alexandra Brito Received: 22 November 2011 / Accepted: 1 February 2012 # Springer Science+Business
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation
 SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
Healing & Repair. Tissue Regeneration
 Healing & Repair Dr. Srikumar Chakravarthi Repair & Healing: Are they same? Repair :Regeneration of injured cells by cells of same type, as with regeneration of skin/oral mucosa (requires basement membrane)
Healing & Repair Dr. Srikumar Chakravarthi Repair & Healing: Are they same? Repair :Regeneration of injured cells by cells of same type, as with regeneration of skin/oral mucosa (requires basement membrane)
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
BLOOD-BRAIN BARRIER MODELS IN CHIP DEVICES
 BLOOD-BRAIN BARRIER MODELS IN CHIP DEVICES Mária A. Deli Institute of Biophysics, Biological Research Centre Hungarian Academy of Sciences, Szeged, Hungary Centre of Excellence of the European Union WHAT
BLOOD-BRAIN BARRIER MODELS IN CHIP DEVICES Mária A. Deli Institute of Biophysics, Biological Research Centre Hungarian Academy of Sciences, Szeged, Hungary Centre of Excellence of the European Union WHAT
Intercellular indirect communication
 Intercellular indirect communication transmission of chemical signals: sending cell signal transmitting tissue hormone medium receiving cell hormone intercellular fluid blood neurocrine neurotransmitter
Intercellular indirect communication transmission of chemical signals: sending cell signal transmitting tissue hormone medium receiving cell hormone intercellular fluid blood neurocrine neurotransmitter
Irene Yu, class of 2019
 Irene Yu, class of 2019 Nerve Cells and Glia Overview What is the brain made of - types of cells What do neurons do; How are they specialized to do it? CONCEPTS communication via signalling changeability
Irene Yu, class of 2019 Nerve Cells and Glia Overview What is the brain made of - types of cells What do neurons do; How are they specialized to do it? CONCEPTS communication via signalling changeability
Microglia-derived extracellular vesicles regulate the proliferation and differentiation of oligodendrocyte precursor cells
 University of Turin CNR Institute of Neuroscience Microglia-derived extracellular vesicles regulate the proliferation and differentiation of oligodendrocyte precursor cells Roberta Parolisi Turin, December
University of Turin CNR Institute of Neuroscience Microglia-derived extracellular vesicles regulate the proliferation and differentiation of oligodendrocyte precursor cells Roberta Parolisi Turin, December
mm Distance (mm)
 b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
Searching for the Mind
 Searching for the Mind with Jon Lieff, M.D. The Very Intelligent Choroid Plexus Epithelial Cell September 20, 2015 Small lining cells along the border of the brain s ventricles are the gateway and supervisor
Searching for the Mind with Jon Lieff, M.D. The Very Intelligent Choroid Plexus Epithelial Cell September 20, 2015 Small lining cells along the border of the brain s ventricles are the gateway and supervisor
ErbB4 migrazione I parte. 3- ErbB4- NRG1
 ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
ErbB4 migrazione I parte 3- ErbB4- NRG1 1 In rodent brains postnatal neuronal migration is evident in three main areas: the cerebellum (CB), the hippocampus (Hipp) and the rostral migratory stream (RMS).
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
DOI: 10.1038/ncb2610 Figure S1 FSMCs derived from MSLN CLN transgenic mice express smooth muscle-specific proteins. Beta-galactosidase is ubiquitously expressed within cultured FSMCs derived from MSLN
Supplementary Figure 1. Nature Neuroscience: doi: /nn.4547
 Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Peroxisomes. Endomembrane System. Vacuoles 9/25/15
 Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature11463 %Sox17(+) 9 8 7 6 5 4 3 2 1 %Sox17(+) #Sox17(+) d2 d4 d6 d8 d1 d12 d14 d18 25 2 15 1 5 Number of Sox17(+) cells X 1 Supplementary Figure 1: Expression of
SUPPLEMENTARY INFORMATION doi:1.138/nature11463 %Sox17(+) 9 8 7 6 5 4 3 2 1 %Sox17(+) #Sox17(+) d2 d4 d6 d8 d1 d12 d14 d18 25 2 15 1 5 Number of Sox17(+) cells X 1 Supplementary Figure 1: Expression of
Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal
 Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus Fadel TISSIR, Yibo QU, Mireille MONTCOUQUIOL, Libing ZHOU, Kouji KOMATSU, Dongbo SHI, Toshihiko FUJIMORI,
Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus Fadel TISSIR, Yibo QU, Mireille MONTCOUQUIOL, Libing ZHOU, Kouji KOMATSU, Dongbo SHI, Toshihiko FUJIMORI,
Cell Migration II: CNS Cell Migration. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
Vascular Basement Membrane Components in Angiogenesis An Act of Balance
 Directions in Science TheScientificWorldJOURNAL (2008) 8, 1246 1249 ISSN 1537-744X; DOI 10.1100/tsw.2008.155 Vascular Basement Membrane Components in Angiogenesis An Act of Balance Lars Jakobsson 1, *
Directions in Science TheScientificWorldJOURNAL (2008) 8, 1246 1249 ISSN 1537-744X; DOI 10.1100/tsw.2008.155 Vascular Basement Membrane Components in Angiogenesis An Act of Balance Lars Jakobsson 1, *
Zhu et al, page 1. Supplementary Figures
 Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Supplementary Table 1. List of primers used in this study
 Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Microfilaments. myosin. In muscle cells. Microfilaments. Microfilaments. Video: Cytoplasmic Streaming. amoeboid movement. Pseudopodia.
 Microfilaments Fig, 6-27a myosin Microfilaments protein func3ons in cellular mo3lity in addi3on to ac3n In muscle cells Thousands of ac3n filaments are arranged parallel to one another Thicker myosin filaments
Microfilaments Fig, 6-27a myosin Microfilaments protein func3ons in cellular mo3lity in addi3on to ac3n In muscle cells Thousands of ac3n filaments are arranged parallel to one another Thicker myosin filaments
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the
 Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Figure 1 Expression of Crb3 in mouse sciatic nerve: biochemical analysis (a) Schematic of Crb3 isoforms, ERLI and CLPI, indicating the location of the transmembrane (TM), FRM binding (FB)
Supplementary Information
 Nature Immunology doi:1.138/ni.2477 Supplementary Information Capillary and arteriolar pericytes attract innate leukocytes exiting through venules and instruct them with pattern recognition and motility
Nature Immunology doi:1.138/ni.2477 Supplementary Information Capillary and arteriolar pericytes attract innate leukocytes exiting through venules and instruct them with pattern recognition and motility
Real-time imaging reveals the single steps of brain metastasis fo mation r
 Real-time imaging reveals the single steps of brain metastasis fo mation r Yvonne Kienast, Louisa von Baumgarten, Martin Fuhrmann, Wolfgang E.F. Klinkert, Roland Goldbrunner, Jochen Herms and Frank Winkler
Real-time imaging reveals the single steps of brain metastasis fo mation r Yvonne Kienast, Louisa von Baumgarten, Martin Fuhrmann, Wolfgang E.F. Klinkert, Roland Goldbrunner, Jochen Herms and Frank Winkler
Major Structures of the Nervous System. Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors
 Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted
 Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted Ctns -/- mice. Cells immunolabeled for the proliferation marker (Ki-67) were counted in sections (n=3 WT, n=4
Supplemental Figure 1. Quantification of proliferation in thyroid of WT, Ctns -/- and grafted Ctns -/- mice. Cells immunolabeled for the proliferation marker (Ki-67) were counted in sections (n=3 WT, n=4
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure S1: Fibroblast-induced elongation of cancer cells requires direct contact with living fibroblasts. A. Representative images of HT29-GFP cultured in the presence
SUPPLEMENTARY FIGURES Supplementary Figure S1: Fibroblast-induced elongation of cancer cells requires direct contact with living fibroblasts. A. Representative images of HT29-GFP cultured in the presence
Histology = the study of tissues. Tissue = a complex of cells that have a common function
 { EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
{ EPITHELIAL TISSUE Histology = the study of tissues Tissue = a complex of cells that have a common function The Four Primary Tissue Types: Epithelium (epithelial tissue) covers body surfaces, lines body
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
MEMBRANE STRUCTURE AND FUNCTION
 MEMBRANE STRUCTURE AND FUNCTION selective permeability permits some substances to cross it more easily than others Figure 7.1 Scientists studying the plasma Reasoned that it must be a phospholipid bilayer
MEMBRANE STRUCTURE AND FUNCTION selective permeability permits some substances to cross it more easily than others Figure 7.1 Scientists studying the plasma Reasoned that it must be a phospholipid bilayer
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
1.The metastatic cascade. 2.Pathologic features of metastasis. 3.Therapeutic ramifications
 Metastasis 1.The metastatic cascade 2.Pathologic features of metastasis 3.Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of bone Paget s disease of the
Metastasis 1.The metastatic cascade 2.Pathologic features of metastasis 3.Therapeutic ramifications Sir James Paget (1814-1899) British Surgeon/ Pathologist Paget s disease of bone Paget s disease of the
Pericyte Influence on Drug Delivery Across the Blood-Brain Barrier
 Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 250 Pericyte Influence on Drug Delivery Across the Blood-Brain Barrier Implications for Therapy of Neurodegenerative
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 250 Pericyte Influence on Drug Delivery Across the Blood-Brain Barrier Implications for Therapy of Neurodegenerative
Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and
 Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and stomach cancer were stained with SA-β-Gal and nuclear fast
Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and stomach cancer were stained with SA-β-Gal and nuclear fast
The basic mechanism of increased microvascular permeability
 Chinese Journal of Pathophysiology 2003,19 (4) : 549-553 549 [ ] 1000-4718 (2003) 04-0549 - 05 3, (, 510515) The basic mechanism of increased microvascular permeability ZHAO Ke - sen, HUANG Qiao - bing
Chinese Journal of Pathophysiology 2003,19 (4) : 549-553 549 [ ] 1000-4718 (2003) 04-0549 - 05 3, (, 510515) The basic mechanism of increased microvascular permeability ZHAO Ke - sen, HUANG Qiao - bing
Inhibition of DYRK1A stimulates human beta-cell proliferation
 Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Inhibition of DYRK1A stimulates human beta-cell proliferation Ercument Dirice 1,, Deepika Walpita 2,, Amedeo Vetere 2, Bennett C. Meier 2,5, Sevim Kahraman 1, Jiang Hu 1, Vlado Dančík 2, Sean M. Burns
Supplemental Information. Menin Deficiency Leads to Depressive-like. Behaviors in Mice by Modulating. Astrocyte-Mediated Neuroinflammation
 Neuron, Volume 100 Supplemental Information Menin Deficiency Leads to Depressive-like Behaviors in Mice by Modulating Astrocyte-Mediated Neuroinflammation Lige Leng, Kai Zhuang, Zeyue Liu, Changquan Huang,
Neuron, Volume 100 Supplemental Information Menin Deficiency Leads to Depressive-like Behaviors in Mice by Modulating Astrocyte-Mediated Neuroinflammation Lige Leng, Kai Zhuang, Zeyue Liu, Changquan Huang,
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70%
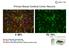 Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70% Pictures: 9 days after electroporation Red: MAP2 Blue: GFAP Green: GFP The cells were from Embryonic Day 14 Mouse Cerebral Cortex Primary Mouse Hippocampal
Primary Mouse Cerebral Cortex Neurons V: 80% TE: 70% Pictures: 9 days after electroporation Red: MAP2 Blue: GFAP Green: GFP The cells were from Embryonic Day 14 Mouse Cerebral Cortex Primary Mouse Hippocampal
Cord blood monocytes as a source of cell therapy products for treatment of brain injuries ISCT/CBA 2015 Cord Blood Workshop Wednesday, May 27, 2015
 Cord blood monocytes as a source of cell therapy products for treatment of brain injuries ISCT/CBA 2015 Cord Blood Workshop Wednesday, May 27, 2015 Andrew E. Balber, PhD Senior Scientific Advisor CT 2,
Cord blood monocytes as a source of cell therapy products for treatment of brain injuries ISCT/CBA 2015 Cord Blood Workshop Wednesday, May 27, 2015 Andrew E. Balber, PhD Senior Scientific Advisor CT 2,
Growth Factors. BIT 230 Walsh Chapter 7
 Growth Factors BIT 230 Walsh Chapter 7 3 Definitions Autocrine: a mode of hormone action in which a hormone affects the function of the cell type that produced it. Paracrine: Relating to the release of
Growth Factors BIT 230 Walsh Chapter 7 3 Definitions Autocrine: a mode of hormone action in which a hormone affects the function of the cell type that produced it. Paracrine: Relating to the release of
