Angiopoietin 2 Induces Pericyte Apoptosis via a3b1 Integrin Signaling in Diabetic Retinopathy
|
|
|
- Lesley Parker
- 6 years ago
- Views:
Transcription
1 Diabetes Volume 63, September Sung Wook Park, 1,2 Jang-Hyuk Yun, 3,4 Jin Hyoung Kim, 1 Kyu-Won Kim, 5 Chung-Hyun Cho, 3,4 and Jeong Hun Kim 1,2,6 Angiopoietin 2 Induces Pericyte Apoptosis via a3b1 Integrin Signaling in Diabetic Retinopathy Diabetes 2014;63: DOI: /db Pericyte loss is an early characteristic change in diabetic retinopathy (DR). Despite accumulating evidence that hyperglycemia-induced angiopoietin 2 (Ang2) has a central role in pericyte loss, the precise molecular mechanism has not been elucidated. This study investigated the role of Ang2 in pericyte loss in DR. We demonstrated that pericyte loss occurred with Ang2 increase in the diabetic mouse retina and that the source of Ang2 could be the endothelial cell. Ang2 induced pericyte apoptosis via the p53 pathway under high glucose, whereas Ang2 alone did not induce apoptosis. Integrin, not Tie-2 receptor, was involved for Ang2-induced pericyte apoptosis under high glucose as an Ang2 receptor. High glucose changed the integrin expression pattern, which increased integrin a3 and b1 in the pericyte. Furthermore, Ang2-induced pericyte apoptosis in vitro was effectively attenuated via p53 suppression by blocking integrin a3 and b1. Although intravitreal injection of Ang2 induced pericyte loss in C57BL/6J mice retina in vivo, intravitreal injection of anti-integrin a3 and b1 antibodies attenuated Ang2-induced pericyte loss. Taken together, Ang2 induced pericyte apoptosis under high glucose via a3b1 integrin. Glycemic control or blocking Ang2/integrin signaling could be a potential therapeutic target to prevent pericyte loss in early DR. Diabetic retinopathy (DR) is the leading cause of visual loss in working-aged people and the most common microvascular complication in diabetic patients despite the recent improvement in the management of DR via glycemic control and photocoagulation (1). Macular edema (leakage) and neovascularization (angiogenesis) both cause severe vision loss in DR (2), and pericyte loss is one of the earliest and most characteristic changes of DR (3). The pericyte plays two major important clinical roles in DR. First, the pericyte enwraps endothelial cells to keep the integrity of inner blood retinal barrier (BRB) with the role of microvascular autoregulation (4). Thus, pericyte loss could weaken the inner BRB, even when endothelial cells are intact, and lead to capillary instability and vascular leakage in macular edema. Second, microaneurysm and neovascularization occur in proliferating endothelial cells at the site of pericyte loss (5). Because pericyte loss is an early diabetic change, preventing pericyte loss for the primary prevention of DR would be beneficial. Although pericyte loss is important in early DR, the mechanism by which hyperglycemia leads to pericyte loss remains largely unknown. However, Ang2 plays a critical role in pericyte loss in DR. Hyperglycemia causes pericyte apoptosis and, ultimately, pericyte loss (6 8). Ang2 increases in the vitreous of patients with proliferative DR (9). In addition, Ang2 is upregulated by hyperglycemia in the diabetic retina and in endothelial cells (10 12). Ang2 induces pericyte loss in normal mice retina and in mice overexpressing Ang2 (10,13). Thus, we postulated that hyperglycemia increases Ang2, which in turn induces pericyte apoptosis in DR. COMPLICATIONS 1 Fight against Angiogenesis-Related Blindness Laboratory, Biomedical Research Institute, Seoul National University Hospital, Seoul, Korea 2 Department of Biomedical Sciences, College of Medicine, Seoul National University, Seoul, Korea 3 Department of Pharmacology and Ischemic/Hypoxic Disease Institute, College of Medicine, Seoul National University, Seoul, Korea 4 Cancer Research Institute, College of Medicine, Seoul National University, Seoul, Korea 5 Department of Pharmacy, Seoul National University, Seoul, Korea 6 Department of Ophthalmology, Seoul National University Hospital, Seoul, Korea Corresponding authors: Jeong Hun Kim, steph25@snu.ac.kr; and Chung-Hyun Cho, iamhyun@snu.ac.kr. Received 24 December 2013 and accepted 31 March This article contains Supplementary Data online at S.W.P. and J.-H.Y. contributed equally to this work by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered.
2 3058 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September 2014 The role of Ang2 in pericyte loss has been studied (10,11,13 15). Apoptosis and migration are both suggested mechanisms of pericyte loss by Ang2; however, the precise mechanism by which Ang2 induces pericyte loss has not yet been fully elucidated. Ang2 has been known to bind to the endothelial-specific Tie-2 tyrosine receptor with similar affinity to Ang1 (16). Ang2 acts in an autocrine manner in angiogenesis. This endothelial cell derived antagonistic ligand of vessel maturation and remodeling controls the Ang1 Tie-2 signaling axis (17). Although Ang2 has been postulated to naturally bind to the Tie-2 receptor in the pericyte as the Ang-Tie system (11,13), whether Tie-2 indeed serves as a receptor for Ang2-induced pericyte loss is not clear. Integrin was recently found to mediate platelet-derived growth factor-bb induced pericyte loss in tumor vessels (18). Also, Ang2 binds to integrin and regulates angiogenesis through integrin signaling (19). Thus, we hypothesized that Ang2 induces pericyte apoptosis via integrin signaling. In this study, we demonstrated that Ang2 induced pericyte apoptosis via the p53 pathway under high glucose. Interestingly, integrin, not Tie-2 receptor, was important for Ang2-induced pericyte apoptosis under high glucose. High glucose increased integrin a3b1 in pericytes. Furthermore, our results showed Ang2-induced pericyte apoptosis was effectively attenuated by blocking integrin a3b1 in vitro and in vivo. Taken together, Ang2 induced pericyte apoptosis via a3b1 integrin signaling in DR. RESEARCH DESIGN AND METHODS Cell Cultures Human umbilical vein endothelial cells (HUVECs; Lonza), human retina microvascular endothelial cells (HRMECs; ACBRI), human brain astrocytes (ACBRI), and human pericytes (PromoCell) were maintained in EBM-2, M199 medium, DMEM with 20% FBS, and pericyte media containing growth factors (PromoCell), respectively. All cells were cultured at 37 C in an incubator with a humidified atmosphere of 95% O 2 and 5% CO 2. Reagents and Antibodies Recombinant mouse and human Ang2, human integrin a3b1, and phycoerythrin (PE)-conjugated anti Tie-2 and mouse IgG antibodies were purchased from R&D Systems. Other reagents and antibodies were anti Bcl-2 family antibodies (Epitomics); anti phospho-p53, anti poly ADP ribose polymerase (PARP), anti-cleaved caspase-3, anti-integrin b1 antibodies (Cell Signaling Technology); anti-p53, anti-tie1, peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology); anti-tie2 antibody and H-Gly-Arg-Gly-Asp-Ser-OH (GRGDS) peptide (Millipore); MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Sigma-Aldrich); fluorescein isothiocyanate conjugated annexin V/propidium iodide assay kit (BD Biosciences); anti neuron-glial antigen 2 (NG2), antiintegrin a3b1, anti-ang2 antibodies (Abcam); and TUNEL fluorescein kit (Roche). Anti-integrin a1 (clone FB12, MAB1973), anti-integrin a3 (clone P1B5, MAB1952), anti-integrin b1 (clone 6S6, MAB2253), and a-integrin blocking and immunohistochemistry kit (a1-6, v) were purchased from Millipore and used for functional blocking. Small interfering (si)rnas for p53 were purchased from Bioneer (Daejeon, Korea). Animals All animal experiments in this study were in strict agreement with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and the guidelines of the Seoul National University Animal Care and Use Committee. Eight-week-old, pathogen-free male C57BL/6J mice were purchased from Central Laboratory Animal Inc. After an 8-h fast, diabetes was induced by one intraperitoneal injection of freshly prepared streptozotocin (STZ; Sigma-Aldrich) at a concentration of 180 mg/kg body weight in 10 mmol/l citrate buffer (ph 4.5). Agematched controls received citrate buffer only. Mice with blood glucose levels.300 mg/dl 4 days after STZ injection were deemed diabetic. Diabetic and nondiabetic mice were killed 6 months after diabetes induction. Eyes were collected under deep anesthesia and immediately frozen at 280 C for ELISA or were fixed in 4% paraformaldehyde for retinal digestion. Glucose levels and body weight were monitored consecutively, and glycated hemoglobin was determined before mice were killed. Retinal Digest Preparations Vascular preparations of whole-mount retinas were performed using a trypsin digestion technique. Briefly, retinas were fixed for at least 24 h in 4% paraformaldehyde, incubated in water for 1 h, and then digested in 2.5% trypsin (Gibco) at 37 C for 1 h. After careful removal of the inner limiting membrane, the retinal vessels were isolated by careful irrigation with filtered water. The retinal digest samples were dried and stained with periodic acid Schiff base for 15 min and hematoxylin. Morphological Quantification of Pericytes and Acellular Capillaries To determine numbers of retinal pericytes and acellular capillary, retinal digest preparations (n = 7 8) were analyzed. Pericytes were identified according to the morphology and relative location to capillaries. Total pericytes were counted in 10 randomly selected areas (original magnification 3400) in the middle one-third of the retinal capillary area. The number of pericytes and acellular capillaries were standardized to the capillary area (numbers of cells or acellular capillaries/mm 2 capillary area). The capillary area was calculated using the NIS-Elements AR 3.2 program (Nikon, Tokyo, Japan). Samples were evaluated in a masked fashion. Intravitreal Injection of Ang2 and Anti-Integrin Antibodies Normal 6-week-old male mice were used. An intravitreal injection was performed under deep anesthesia with 1 ml
3 diabetes.diabetesjournals.org Park and Associates 3059 containing 100 ng Ang2 and 500 ng anti-integrin a3 (clone P1B5) or anti-integrin b1 (clone 6S6). Sterile PBS was injected for control. After 10 days, retinas underwent digestion preparation. Transferase-Mediated TUNEL Assay and Immunofluorescence Retinal digestion preparations were incubated with antirabbit NG2 antibody (1:100) and a TUNEL fluorescein kit. Nuclei were counterstained with DAPI. TUNEL and NG2 double-positive cells were evaluated with a fluorescence microscope (Nikon). Cell Viability Assay In all experiments, cells were seeded into 96-well plates. After 24 h, cells were treated with Ang1 (300 ng/ml), Ang2 (300 ng/ml) under normal glucose (5 mmol/l glucose), high glucose (25 mmol/l glucose), and high mannitol (5 mmol/l glucose and 20 mmol/l mannitol) as an osmotic control for 48 h. Cell viability was determined by MTT assay according to the manufacturer s instructions. Three independent experiments were performed for each experimental condition. FACS Analysis To evaluate apoptosis, cells were treated with Ang2 (300 ng/ml) under normal glucose, high mannitol, and high glucose at 37 C for 48 h. To determine the effect of integrin blocking, cells were treated with anti integrin blocking antibodies (5 mg/ml) 1 h before the addition of Ang2. The cells were harvested and washed twice in PBS. Cells were stained with fluorescein isothiocyanate annexin-v and propidium iodide (PI) for 15 min and analyzed by flow cytometry. Annexin V positive/pi-negative cells were determined to be apoptotic. To evaluate Tie-2 expression, pericytes and HUVECs ( ) were suspended in the complete media (25 ml) for each experiment. PE-conjugated anti-tie2 antibody was added to the each sample at 4 C for 1 h. PE-conjugated mouse IgG antibody was used as a control. Quantitative RT-PCR All RNA was collected and isolated from cells using the RNeasy Plus Mini kit (Qiagen). cdnas were prepared from RNAs (1 mg) using 2.5 mmol/l oligo-dt primers, 1 mmol/l deoxyribonucleotide triphosphates, and murine leukemia virus RT. Quantitative (q)pcr assays were performed in qpcr Master Mix for SYBR Green PCR Master Mix (Applied Biosystems) using 7900HT real-time PCR (Applied Biosystems). Reaction conditions were 50 cycles of 95 C for 5 s and 60 C for 20 s for qpcr. Quantitative real-time PCR was performed using the following primers: Ang2 (forward: 59-ACTGTGTCCTCTTCCACCAC-39and reverse: 59-GGATGTTTAGGGTCTTGCTTT-39); Tie-2 (forward: 59-GCTTGCTCCTTTCTGGAACTGT-39 and reverse: 59-CGCCACCCAGAGGCAAT-39) (20); Tie-1 (forward: 59- AGAACCTAGCCTCCAAGATT-39 and reverse: 59-ACTGTA GTTCAGGGACTCAA-39); ITGA1 (forward: 59-GGTTCCT ACTTTGGCAGTATT-39 and reverse: 59- AACCTTGTCTGA TTGAGAGCA-39); ITGA3 (forward: 59-AAGGGACCTTCAG GTGCA-39 and reverse: 59-TGTAGCCGGTGATTTACCAT-39); ITGB1 (forward: 59-GAAGGGTTGCCCTCCAGA-39 and reverse: 59-GCTTGAGCTTCTCTGCTGTT-39); and b-actin (forward: 59-GCCGCCAGCTCACCAT-39 and reverse: 59- TCGATGGGGTACTTCAGGGT-39). A mean quantity was calculated from triplicate qpcr for each sample and normalized to the control gene. Different primers were used for Tie-2 RT-PCR (forward: 59-TGTTCCTGTGCCACAGGCTG-39 and reverse: 59-CACT GTCCCATCCGGCTTCA-39). PCR products were separated on 1% agarose gels and visualized using SYBR Safe DNA Gel Stain (Invitrogen) under ultraviolet transillumination. Immunoprecipitation and Immunoblotting For immunoprecipitation, Ang2 (500 ng), integrin a3b1 (500 ng), and anti-integrin a3b1 antibodies (2 mg) were incubated with G-Sepharose beads at 4 C overnight (19). Immune complexes were collected by centrifugation and washed with buffer three times. For immunoblotting, cells were harvested and lysed in radioimmunoprecipitation assay buffer with a protease inhibitor cocktail. Protein lysates were resolved by SDS-PAGE and transferred onto nitrocellulose membrane. The membranes were incubated with primary antibodies (1:1,000) at 4 C overnight and secondary antibodies (1:5,000) at room temperature for 1 h. The membranes were incubated with enhanced chemiluminescent substrate (Pierce) and exposed to film. Statistical Analysis Statistical analyses were performed using the standard two-tailed Student t test assuming unequal variances, and P, 0.01 was considered statistically significant. Quantitative data are given as mean 6 SD. Data in figures are depicted as mean 6 SE. RESULTS Retinal Capillary Pericyte Numbers Are Decreased in Diabetic Mice Retinas The numbers of pericytes and acellular capillaries in retinal digestion were compared between mice with 6-month STZinduced diabetes and age-matched nondiabetic control mice (Fig. 1A). On one hand, the number of retinal capillary pericytes was significantly decreased in the STZ-induced diabetic mice retinas ( ) compared with that of the control group (1, , P, 0.001; Fig. 1B). On the other hand, the number of retinal acellular capillaries was significantly increased in the STZ-induced diabetic mice retinas ( ) compared with that of the control group ( , P, 0.001; Fig. 1C). Table 1 reports the metabolic and physical parameters of the experimental groups. Ang2 Is Increased in Diabetic Retinas, and the Source of Ang2 Could Be Endothelial Cells The effect of hyperglycemia on Ang2 expression in STZinduced diabetic retinas was evaluated by qrt-pcr. In diabetic retinas at 6 months, Angpt2 mrna increased
4 3060 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September 2014 Figure 1 The number of retinal capillary pericytes is decreased in the diabetic mouse retina. Pericytes were identified in retinal digest preparations by morphologic criteria (shape, staining intensity, and relative position in the capillary) and quantitated in 6-month STZinduced diabetic mice (DM) and age-matched controls (Con). A: Representative examples of periodic acid Schiff and hematoxylin-stained retinal digest preparations of nondiabetic and diabetic mice after 6 months of diabetes are shown. The arrows indicate pericytes, and the arrowheads indicate acellular capillaries (original magnification 3400; scale bar = 20 mm). The number of retinal pericytes (B) and acellular capillaries (C) are shown normalized to the area of capillaries (mm 2 ) in which they were counted (n = 8 mice in each group). The bar graph represents mean 6 SE. *P < 0.01 by Student t test fold compared with nondiabetic normal retinas (P = 0.004; Fig. 2A). Also, Angpt1 mrna increased 2.54-fold (P = 0.004) and Vegfa mrna increased 1.6-fold (P = 0.027; Supplementary Fig. 1A and B). Next, to determine the source of Ang2 in the diabetic retinas, we examined the effects of high glucose on in vitro ANGPT2 mrna transcription by qrt-pcr in three major components of the BRB: HRMECs, pericytes, and astrocytes. On one hand, high glucose increased ANGPT2 mrna level in HRMECs more than 1.5-fold ( , P = 0.003) compared with normal glucose (Fig. 2B). On the other hand, high mannitol, an osmotic control, did not increase ANGPT2 mrna ( , P = 0.094) in HRMECs (Fig. 2B). Also, high glucose did not increase ANGPT2 mrna in pericytes ( , P = 0.418) and astrocytes ( , P = 0.418; Fig. 2B and C). These data demonstrate that Ang2 increases in the diabetic retina and that the source of Ang2 increase is retinal microvascular endothelial cells. Ang2 Plays Synergistic Role in Pericyte Apoptosis Under High-Glucose Conditions We determined the effect of Ang2 on the cell viability and apoptosis of pericytes under high glucose. Ang2 alone did not affect cell viability in pericytes ( %, P = 0.221). High glucose reduced cell viability in pericytes ( %, P = 0.020; Fig. 3A). Interestingly, Ang2 Table 1 Metabolic and physical parameters of the STZ-induced diabetic and age-matched nondiabetic control mice at 6 months Nondiabetic Diabetic P value Body weight (g) ,0.001 Blood glucose (mmol/l) ,0.001 HbA 1c (%) ,0.001 HbA 1c (mmol/mol) Data are presented as mean 6 SD.
5 diabetes.diabetesjournals.org Park and Associates 3061 Figure 2 Ang2 is increased in diabetic retinas, and the source of Ang2 could be endothelial cells. A: Angpt2 mrna levels were determined in 6-month STZ-induced diabetic mice (DM) retinas compared with age-matched control (Con) mice by qrt-pcr and normalized to Rn18s mrna. Angpt2 expression increased in 6-month STZ-induced DM retinas (n = 6 mice in each group). ANGPT2 mrna transcription is induced by high glucose (HG; 25 mmol/l) in HRMECs (B) but not in pericytes (C) or astrocytes (D). HRMEC, pericytes, and astrocytes were incubated for 48 h under 25 mmol/l HG, 20 mmol/l mannitol plus 5 mmol/l glucose (HM), or 5 mmol/l glucose (NG) as an osmotic control. ANGPT2 mrna transcription was assessed by qrt-pcr, with actin as an internal control. ANGPT2 mrna levels were normalized to ACTIN mrna and reported as fold-induction compared with cells exposed to 5 mmol/l glucose. Bar graph represents mean 6 SE. *P < 0.01, #P > 0.05 by Student t test. aggravated cell death under high glucose in pericytes ( %, P = 0.002; Fig. 3A). Next, pericyte apoptosis was assessed by annexin-v/pi flow cytometric analysis. Of importance, the number of apoptotic pericytes was increased under high glucose ( %, P, 0.001) compared that of the control group ( %). Furthermore, Ang2 significantly aggravated high glucose induced pericyte apoptosis ( %, P, 0.001), whereas Ang2 alone did not induce apoptosis ( %, P = 0.298) under normal glucose (Fig. 3B and Supplementary Fig. 2A). Next, we determined the effect of Ang2 on the cell viability and apoptosis of HRMECs under high-glucose conditions. Ang1 and Ang2 increased cell viability in HRMECs under high glucose ( % [P = 0.014] and % [P = 0.040], respectively; Fig. 3C). As expected, Ang2 significantly decreased the apoptotic cell population in HRMECs (Fig. 3D, and Supplementary Fig. 2B). These data suggested that Ang2 plays a synergistic role in pericyte apoptosis under a high-glucose condition and has a protective effect on the endothelial cell. Ang2 Induces Pericyte Apoptosis via the p53 Pathway Under High-Glucose Conditions Western blot studies confirmed that Ang2 induced the apoptosis pathway with an increase of Bax, cleaved PARP, and cleaved caspase-3 under high glucose but not under normal glucose (Fig. 4A). Next, we aimed to identify the mechanism that mediates Ang2-induced pericyte apoptosis under high glucose. We found that Ang2 induced p53 phosphorylation (Fig. 4B) and, subsequently, p53 accumulation (Fig. 4C) under high glucose. Then, to determine the role of the p53 pathway for the observed Ang2- mediated pericyte apoptosis, we treated pericytes with control sirna or two different p53 sirnas. The p53 sirnas effectively downregulated p53 expression (Fig. 4D) and also attenuated Ang2-induced pericyte apoptosis under high glucose (Fig. 4E). Interestingly, Ang2 phosphorylated extracellular signal related kinase (ERK), but not Akt (Fig. 4F). This ERK phosphorylation by Ang2 was inhibited by PD98059 (ERK inhibitor), and the ERK inhibitor attenuated Ang2-induced p53 phosphorylation (Fig. 4G). These data suggest that Ang2 induces pericyte apoptosis via the p53 pathway under highglucose conditions. Integrin, Not Tie-2, Is Important for Ang2-Induced Pericyte Apoptosis Under High Glucose As an Ang2 Receptor To determine whether the Tie-2 receptor is related with Ang2-induced pericyte apoptosis, Western blot analysis (Fig. 5A) andrt-pcr(fig.5b) for Tie-2 and Tie-1 were performed on lysates obtained from HUVECs and pericytes.
6 3062 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September 2014 Figure 3 Ang2 plays synergistic role in pericyte apoptosis under high glucose (HG; 25 mmol/l glucose). The effects of Ang2 on the cell viability and apoptosis of pericytes and HRMECs under HG conditions were determined. Pericytes and HRMECs were incubated for 48 h with and without Ang1 (300 ng/ml) or Ang2 (300 ng/ml) under HG and compared with control (Con). A and C: Cell viability was assessed by MTT assay. Ang2 induced cell death under HG in pericytes (A) but not in HRMECs (C). Pericytes (B) and HRMECs (D) were stained with annexin-v fluorescein isothiocyanate and PI and analyzed by flow cytometry. Cell apoptosis was expressed as the percentage of apoptotic cells in total cell populations. B: HG induced pericyte apoptosis, and Ang2 aggravated that apoptosis. D: Ang2 showed a protective effect on HRMEC apoptosis. The bar graph represents the mean 6 SE of three independent experiments. HM, high mannitol (5 mmol/l glucose and 20 mmol/l mannitol); NG, normal glucose (5 mmol/l glucose). *P < 0.01 by Student t test. Interestingly, pericytes did not express Tie-2 or Tie-1, whereas HUVECs expressed Tie-2 and Tie-1. These data were confirmed by qrt-pcr. TIE2 mrna and TIE1 mrna levels were significantly lower in pericytes than in HUVECs (Fig. 5C). Compared with HUVECs with Tie-2 expression, pericytes did not express Tie-2 in FACS analysis (Fig. 5D). To test whether integrins may serve as receptors for Ang2 in pericyte apoptosis under high glucose, we incubated pericytes under high glucose for 15 min with Ang2 and Gly-Arg-Gly-Asp-Ser (GRGDS) peptides, which can inhibit integrins that bind the Arg-Gly-Asp (RGD) sequence (21). GRGDS (0.5 mg/ml) attenuated Ang2- induced p53 phosphorylation (Fig. 5E). This result showed that integrin signaling is involved in Ang2-induced p53 phosphorylation. Of importance, these data suggested that integrin, not Tie-2, is important for Ang2- induced pericyte apoptosis under high glucose as an Ang2 receptor. High Glucose Increases Integrin a1, a3, and b1 in Pericytes As shown in Fig. 3C and Fig. 5E, Ang2 induced pericyte apoptosis via the integrin receptor under high glucose but not under normal glucose. We hypothesized that high glucose preconditioned pericytes susceptible to Ang2 by changing the integrin pattern. We screened the integrin a subunits (a1 6 and av) to determine which integrin subunit is responsible for Ang2-induced pericyte apoptosis. Integrin a1 and a3 blocking antibodies attenuated Ang2- induced p53 phosphorylation (Fig. 6A). Next, to determine whether high glucose changes the integrin expression pattern, we performed qrt-pcr and Western blot studies for a1, a3, and b1. The choice was made because integrin a1 anda3 can form the heterodimer with only integrin b1 (22). High glucose increased mrna levels at 24 and 48 h for ITGa1 (1.86- fold and 2.20-fold, P, 0.01; Fig. 6B), ITGa3 (1.50-fold and 1.83-fold, P, 0.01; Fig. 6C), and ITGb1 (1.50-fold and 1.43-fold, P, 0.01; Fig. 6D), respectively. In addition, high glucose increased integrin a1, a3, and b1 expression (Fig. 6E). However, the integrin a1 was rarely expressed compared with integrin a3. In this regard, we performed a coimmunoprecipitation assay to show direct binding of Ang2 to integrin a3b1. Indeed, Ang2 directly bound to integrin a3b1 (Fig. 6F). Integrin a3b1 Suppression Inhibits Ang2-Induced Pericyte Apoptosis From the result of integrin expression in pericytes under high glucose, integrin a1b1 or a3b1 were supposed to be
7 diabetes.diabetesjournals.org Park and Associates 3063 Figure 4 Ang2 induces pericyte apoptosis via the p53 pathway under high glucose (HG; 25 mmol/l glucose). A: Western blot analysis for Bax, Bcl-2, Bcl-xL, cleaved caspase-3, and cleaved PARP were performed on lysates obtained from pericytes treated with Ang2 (300 ng/ml) under normal glucose (NG; 5 mmol/l glucose), high mannitol (HM; 5 mmol/l glucose and 20 mmol/l mannitol), and HG for 48 h compared with control (Con). B: Western blot analysis for phospho (p)-p53 (Ser15) was performed on lysates obtained from pericytes treated with Ang2 for 15, 30, and 60 min under HG (25 mmol/l). C: Western blot analysis for p53 was performed on lysates obtained from pericytes treated with Ang2 for 24 and 48 h under HG (25 mmol/l). D: After the pericyte transfection with control sirna or p53 sirna, Western blot analysis for p53 was performed on cell lysates with Ang2 for 48 h under HG (25 mmol/l), with b-tubulin used as a loading control. Data represent three independent experiments. E: Apoptotic cell counts were assessed by FACS analysis 48 h after Ang2 treatment in sirna-transfected pericytes. The bar graph represents the mean 6 SE of three independent experiments. *P < 0.01 by Student t test. F: Ang2 was treated for 5, 15, 30, and 60 min under HG. Determination of p-erk, ERK, p-akt, and Akt was by Western blot. G: Pericytes were preincubated with Wortmannin (1 mmol/l) or PD98059 (20 mmol/l) for 1 h and treated with Ang2 for 15 min under HG. Determination of p-p53, p53, p-erk, ERK, p-akt, and Akt was by Western blot, with b-tubulin used as a loading control. the possible receptor of Ang2. To determine whether Ang2 induced pericyte apoptosis through integrin a1b1 or a3b1, pericytes were incubated under 25 mmol/l high glucose with Ang2 and anti-integrin a1, a3, and b1 antibodies for 48 h. Interestingly, Ang2-induced pericyte apoptosis under high glucose was attenuated by anti integrin a3 and b1 antibodies but not by the anti integrin a1 antibody (Fig. 7A, and Supplementary Fig. 3A). These results were confirmed for p53 expression on Western blot analysis (Fig. 7B). Ang2 significantly increased p53 expression under high glucose ( , P, 0.01) but was significantly attenuated by anti integrin a1, a3, and b1 antibodies ( , , and , respectively; P, 0.01).
8 3064 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September 2014 Figure 5 Integrin, not Tie-2, is important for Ang2-induced pericyte apoptosis under high glucose as an Ang2 receptor. Western blot (A) and RT-PCR (B) for Tie-2 and Tie-1 expression were performed on lysates obtained from HUVECs and pericytes. b-tubulin was used as an internal control. C: TIE2 and TIE1 mrna transcriptions were assessed by qrt-pcr. Actin was used as an internal control. TIE2 and TIE1 mrna levels were normalized to ACTIN mrna and reported as fold induction compared with HUVECs. *P < 0.01 by Student t test. D: HUVECs and pericytes were analyzed by flow cytometry for Tie-2 expression. E: Western blot analysis for phospho (p)-p53 (ser15) and p53 were performed on lysates obtained from pericytes treated with Ang2 (300 ng/ml) or GRGDS (0.5 mg/ml) under 25 mmol/l high glucose (HG) for 15 min. b-tubulin was used as a loading control. Data represent three independent experiments. Con, control. On the basis of in vitro experiments, we next intravitreously injected normal mice with 100 ng Ang2 with and without 500 ng anti-a3 or 500 ng anti-b1 antibodies. Ten days after the intravitreal injection, the isolated retinas were digested with trypsin for pericyte evaluation (Fig. 7C). The in vivo intravitreal injection of Ang2 induced pericyte loss in the retina compared with that in PBS injected control mice (1, and 1, cells/mm 2, P, 0.001). Ang2-induced pericyte loss was significantly attenuated by anti-a3 or anti-b1 antibodies (1, and 1, cells/mm 2, P, 0.001; Fig. 7D). In addition, Ang2-induced TUNEL and NG2 double-positive pericytes were decreased in mice injected with anti-a3 or anti-b1 antibodies (Fig. 7E). These in vivo data suggest that Ang2 induced pericyte loss via an apoptotic mechanism in the retina. Overall, these data demonstrate that integrin a3 and b1 are important for Ang2-induced pericyte apoptosis via the p53 pathway. DISCUSSION In this study, we demonstrated that pericyte loss occurred with Ang2 increase in diabetic mice retinas (10,11,13). Previously, Tie-2 was known to be related with Anginduced pericyte survival and recruitment (11); however, there was no direct evidence that Ang2 induced pericyte apoptosis via Tie-2. Unlike the previous study (11), pericytes did not express Tie-2 mrna or protein in our study. Although Ang2 alone did not induce apoptosis in vitro (11), Ang2 induced pericyte apoptosis under high glucose via the p53 pathway (Fig. 4). Thus, we postulated that Ang2 would induce Tie-2 independent apoptosis in pericytes. Accumulating evidence supports the role of integrin in Ang2 activity in endothelial cells (19); however, little is known about the Ang2/integrin system in the pericyte. We thus focused on the integrin receptors on pericytes. Integrins are cell adhesion molecules that are expressed on the surface of endothelial cells and pericytes. The contribution of endothelial and mural cell integrins to angiogenesis has been studied. Pericytes express integrins, including the collagen receptors a1b1 anda2b1; the laminin receptors a3b1, a6b1, a6b4, and a7b1; the fibronectin receptors a4b1 and a5b1; and the osteoponin receptors a8b1anda9b1 (22). Cytokines and extracellular matrix change integrin subtypes in pericytes (23). On the basis of the differential response to Ang2 in the pericytes depending on the glucose concentration (Fig. 3B and 4A),
9 diabetes.diabetesjournals.org Park and Associates 3065 Figure 6 High glucose (HG; 25 mmol/l) increases integrin a1, a3, and b1 in pericytes. A: Western blot analysis for phospho (p)-p53 (Ser15) and p53 were performed on lysates obtained from pericytes treated with Ang2 (300 ng/ml) or various integrin-blocking antibodies (5 mg/ml, a1, a2, a3, a4, a5, a6, and av) under HG for 15 min. b-tubulin was used as a loading control. Data represent three independent experiments. B D: Pericyte were incubated under HG for 24 and 48 h. ITGa1 (B), ITGa3 (C), and ITGb1 (D) mrna transcriptions were assessed by qrt-pcr. Actin was used as an internal control. ITGa1, ITGa3, and ITGb1 mrna levels were normalized to actin mrna and reported as fold induction compared with control. *P < 0.01 by Student t test. E: Western blot analyses for integrin a1, a3, and b1 were performed on lysates obtained from pericytes incubated under HG and normal glucose (NG; 5 mmol/l) for 48 h. b-tubulin was used as a loading control. Data represent three independent experiments. F: After incubation of Ang2 (500 ng), integrin a3b1 (500 ng), and integrin a3b1 antibody, immune complexes were coimmunoprecipitated to show direct binding of Ang2 to integrin a3b1 and then were analyzed for Ang2 and integrin b1. The same amounts of recombinant Ang2 and integrin a3b1 were used as an input. we hypothesized that high glucose could change the integrin subtype more susceptible to Ang2. During qrt-pcr array for screening the integrin subtype change response to high glucose, only ITGa1, ITGa2, ITGa3, and ITGb1 mrna increased more than 1.5-fold among ITGa1, ITGa2, ITGa3, ITGa4, ITGa5, ITGa6, ITGa7, ITGav, and ITGb1 mrna (data not shown). In addition, basal ITGa3 mrna was more than six times higher than ITGa1 or ITGa2 mrna in pericytes even under normal glucose conditions. Thus, we concluded that integrin a3 and b1 are both predominant and high-glucose inducible integrins in pericytes. The proportion of integrin a1 in pericytes is too minor to be responsible for Ang2-induced pericyte apoptosis (Fig. 6E and 7B). In addition, many integrins, including
10 3066 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September 2014 Figure 7 Ang2-induced pericyte apoptosis is inhibited by suppression of integrin a3b1. A and B: Pericytes were incubated under 25 mmol/l high glucose (HG) with Ang2 (300 ng/ml) and anti-integrin a1, a3, and b1 antibodies (Ab; 5 mg/ml) for 48 h. A: Pericyte apoptosis was analyzed by FACS and expressed as the percentage of apoptotic cells in total cell populations. B: Western blot analysis for p53 was performed on lysates obtained from pericyte. b-tubulin was used as a loading control. Data represent three independent experiments. (ImageJ quantitation; n = 3, mean 6 SD). *P < 0.01 compared with Ang2 treatment by two-tailed Student t test. C: Ang2 (100 ng), with and without anti-a3 or anti-b1 antibodies (500 ng), was intravitreously injected to normal mice (n = 8 mice in each group). Eyes were enucleated 10 days after the intravitreal injection, and the isolated retinas were digested with trypsin for pericyte evaluation. Pericytes were identified in periodic acid Schiff and hematoxylin-stained retinal digest preparations by morphologic criteria. Representative examples of retinal digest preparations are shown. Arrows indicate representative pericytes. D: The number of pericytes was normalized to the area of capillaries (mm 2 ) in which they were counted. *P < 0.01 by Student t test. E: Retinal digest preparations were immunostained with NG2 (red), TUNEL (green), and DAPI (blue). White arrows indicate TUNEL-positive pericytes. C and E: Original magnification Scale bar = 20 mm. Bar graph represents mean 6 SE.
11 diabetes.diabetesjournals.org Park and Associates 3067 avb3, a5b1, aiibb3, avb6, and a3b1, recognize the tripeptide RGD in their ligands (24). However, integrin a1b1 recognizes a configuration of residues formed by arginine and aspartic acid residues, not RGD. Attenuation of p53 by GRGDS supported that Ang2-induced pericyte apoptosis was effectively attenuated by blocking integrin a3b1 (Fig. 5E and 7B). Integrin receptors induced by hyperglycemia in DR could make the pericyte more susceptible to the Ang2, and in turn, lead to the pericyte apoptosis by the Ang2/ integrin signaling pathway. The laminin receptor a3b1 integrin is expressed in vascular endothelial cells and acts to suppress pathological angiogenesis in the retina and other organs (25,26). However, a role for integrin a3b1 in the pericyte is yet to be determined (22). Inhibition of b1 integrin induced a rounded morphology of the pericytes, suggesting pericyte adhesive properties were affected or that these cells were undergoing apoptosis (27). In this study, we suggested that integrin a3b1 was important for Ang2- induced apoptosis. This is notably different from the classical concept that Tie-2 mediates the effect of Ang2 as an Ang2 receptor. Absence of Tie-2 in the human pericytes used in this study was confirmed by various experiments, including RT-PCR with two different primers, Western blot, and FACS analysis (Fig. 5A D). In addition, Ang1 and Ang2 can both directly bind to some integrins and thereby signal in the absence of Tie-2 (28 31). Thus, we suggested that integrin mediated Ang2-induced apoptosis at least in Tie-2 negative pericytes. Previous studies have shown that vascular endothelial cells are the primary source of Ang2 (32 35). High glucose increases Ang2 mrna in HRMECs (36). The endothelial cell is regarded as potential source of Ang2 in the retina by transient rapid release from the Weibel-Palade body and chronic upregulation of Ang2 (32,37,38). Chronic hyperglycemia upregulates Ang2 in the diabetic retina (36,39), and Ang2 upregulation is causally involved in the pathogenesis of pericyte loss in DR (10). Hyperglycemia induces pericyte loss not only by increasing Ang2 in endothelial cells but also by changing the integrin subtype prone to Ang2 in pericytes. Our data support that glycemic control is important to prevent Ang2-induced pericyte loss in early DR. Glycemic control is the effective treatment to reduce the progression of DR (40 42). We showed that Ang2 induced pericyte apoptosis under high glucose via the p53 pathway. Furthermore, Ang2-induced pericyte apoptosis was effectively attenuated in vitro and in vivo by blocking integrin a3b1. Integrin a3 and b1 blockers reduced p53 signaling by Ang2. Ser-15 of p53 is phosphorylated by a mitogen-activated protein kinase (ERK1/2)-dependent pathway, and this step is required for apoptosis to occur (43). Others have suggested that hyperglycemia causes pericyte loss by apoptosis (6 8). In this study, we postulated that hyperglycemia-induced Ang2 overexpression in endothelial cells, in turn, caused apoptotic cell death of pericytes. In an animal model of DR, high glucose was induced by STZ injection as early as 3 days. Then, Ang2 had increased at 3 months with pericyte loss, and finally, acellular capillary at 6 months (10,44). The mechanism by which capillaries become acellular is largely unknown. Apoptosis preceding the formation of acellular capillaries in retinas from diabetic rats and humans is one mechanism by which endothelial cells could be eliminated from the diabetic capillary (8). Retinal capillary coverage with pericytes is crucial for the survival of endothelial cells. Loss of pericytes is related to the increase of acellular capillaries from hyperglycemic injury (5). Our data indicate Ang2 did not directly induce endothelial cell death (Fig. 3C and D); thus, we postulated that an acellular capillary could cause endothelial cell death secondary to pericyte loss by Ang2. In conclusion, we demonstrated that pericyte loss occurred with Ang2 increase in the diabetic mice retinas. Ang2 induced pericyte apoptosis via the p53 pathway under high glucose. High glucose increased integrin a3b1 in pericytes. Interestingly, integrin was involved in Ang2- induced pericyte apoptosis. Furthermore, Ang2-induced pericyte apoptosis was effectively attenuated by blocking integrin a3b1 both in vitro and in vivo. Taken together, Ang2 induced p53-dependent pericyte apoptosis via a3b1 integrin signaling in DR. We suggest that glycemic control or blocking Ang2/integrin signaling could be a potential therapeutic target to prevent pericyte loss in early DR. Funding. This study was supported by the Seoul National University Research Grant ( to Je.H.K.), the Seoul National University Hospital Research Fund ( to Je.H.K.), the Pioneer Research Center Program of the National Research Foundation for the Ministry of Education, Science and Technology (NRF/MEST; to Je.H.K.), the Bio-Signal Analysis Technology Innovation Program of NRF/MEST ( to Je.H.K.), the Global Research Laboratory Program of NRF/MEST ( to K.-W.K.) and the NRF of Korea grant funded by the Korea government (2012R1A2A2A , to C.-H.C. and to Je.H.K.). Duality of Interest. No potential conflicts of interest relevant to this article were reported. Author Contributions. S.W.P. and J.-H.Y. performed the experiments and wrote the manuscript. Ji.H.K. and K.-W.K. analyzed the data and reviewed the manuscript. C.-H.C. and Je.H.K. designed the study and critically revised the manuscript. Je.H.K. is the guarantor of this work, and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. References 1. Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med 2012; 366: Moss SE, Klein R, Klein BE. The incidence of vision loss in a diabetic population. Ophthalmology 1988;95: Cogan DG, Toussaint D, Kuwabara T. Retinal vascular patterns. IV. Diabetic retinopathy. Arch Ophthalmol 1961;66: Shepro D, Morel NM. Pericyte physiology. FASEB J 1993;7: Hammes HP, Lin J, Renner O, et al. Pericytes and the pathogenesis of diabetic retinopathy. Diabetes 2002;51: Romeo G, Liu WH, Asnaghi V, Kern TS, Lorenzi M. Activation of nuclear factor-kappab induced by diabetes and high glucose regulates a proapoptotic program in retinal pericytes. Diabetes 2002;51:
12 3068 Ang2 Induces Pericyte Loss via a3b1 Integrin Diabetes Volume 63, September Miller AG, Smith DG, Bhat M, Nagaraj RH. Glyoxalase I is critical for human retinal capillary pericyte survival under hyperglycemic conditions. J Biol Chem 2006;281: Mizutani M, Kern TS, Lorenzi M. Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J Clin Invest 1996;97: Watanabe D, Suzuma K, Suzuma I, et al. Vitreous levels of angiopoietin 2 and vascular endothelial growth factor in patients with proliferative diabetic retinopathy. Am J Ophthalmol 2005;139: Hammes HP, Lin J, Wagner P, et al. Angiopoietin-2 causes pericyte dropout in the normal retina: evidence for involvement in diabetic retinopathy. Diabetes 2004;53: Cai J, Kehoe O, Smith GM, Hykin P, Boulton ME. The angiopoietin/tie-2 system regulates pericyte survival and recruitment in diabetic retinopathy. Invest Ophthalmol Vis Sci 2008;49: Yao D, Taguchi T, Matsumura T, et al. High glucose increases angiopoietin-2 transcription in microvascular endothelial cells through methylglyoxal modification of msin3a. J Biol Chem 2007;282: Pfister F, Feng Y, vom Hagen F, et al. Pericyte migration: a novel mechanism of pericyte loss in experimental diabetic retinopathy. Diabetes 2008;57: Pfister F, Wang Y, Schreiter K, et al. Retinal overexpression of angiopoietin- 2 mimics diabetic retinopathy and enhances vascular damages in hyperglycemia. Acta Diabetol 2010;47: Feng Y, vom Hagen F, Pfister F, et al. Impaired pericyte recruitment and abnormal retinal angiogenesis as a result of angiopoietin-2 overexpression. Thromb Haemost 2007;97: Maisonpierre PC, Suri C, Jones PF, et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 1997;277: Augustin HG, Koh GY, Thurston G, Alitalo K. Control of vascular morphogenesis and homeostasis through the angiopoietin-tie system. Nat Rev Mol Cell Biol 2009;10: Hosaka K, Yang Y, Seki T, et al. Tumour PDGF-BB expression levels determine dual effects of anti-pdgf drugs on vascular remodelling and metastasis. Nat Commun 2013;4: Felcht M, Luck R, Schering A, et al. Angiopoietin-2 differentially regulates angiogenesis through TIE2 and integrin signaling. J Clin Invest 2012;122: Rabascio C, Muratori E, Mancuso P, et al. Assessing tumor angiogenesis: increased circulating VE-cadherin RNA in patients with cancer indicates viability of circulating endothelial cells. Cancer Res 2004;64: Ruoslahti E. RGD and other recognition sequences for integrins. Annu Rev Cell Dev Biol 1996;12: Silva R, D Amico G, Hodivala-Dilke KM, Reynolds LE. Integrins: the keys to unlocking angiogenesis. Arterioscler Thromb Vasc Biol 2008;28: Tigges U, Boroujerdi A, Welser-Alves JV, Milner R. TNF-a promotes cerebral pericyte remodeling in vitro, via a switch from a1 to a2 integrins. J Neuroinflammation 2013;10: Plow EF, Haas TA, Zhang L, Loftus J, Smith JW. Ligand binding to integrins. J Biol Chem 2000;275: Watson AR, Pitchford SC, Reynolds LE, et al. Deficiency of bone marrow beta3-integrin enhances non-functional neovascularization. J Pathol 2010;220: Lim Y, Jo DH, Kim JH, et al. Human apolipoprotein(a) kringle V inhibits ischemiainduced retinal neovascularization via suppression of fibronectin-mediated angiogenesis. Diabetes 2012;61: Carnevale E, Fogel E, Aplin AC, et al. Regulation of postangiogenic neovessel survival by beta1 and beta3 integrins in collagen and fibrin matrices. J Vasc Res 2007;44: Carlson TR, Feng Y, Maisonpierre PC, Mrksich M, Morla AO. Direct cell adhesion to the angiopoietins mediated by integrins. J Biol Chem 2001;276: Hu B, Jarzynka MJ, Guo P, Imanishi Y, Schlaepfer DD, Cheng SY. Angiopoietin 2 induces glioma cell invasion by stimulating matrix metalloprotease 2 expression through the alphavbeta1 integrin and focal adhesion kinase signaling pathway. Cancer Res 2006;66: Bezuidenhout L, Zilla P, Davies N. Association of Ang-2 with integrin beta 2 controls Ang-2/PDGF-BB-dependent upregulation of human peripheral blood monocyte fibrinolysis. Inflammation 2009;32: Thomas M, Felcht M, Kruse K, et al. Angiopoietin-2 stimulation of endothelial cells induces alphavbeta3 integrin internalization and degradation. J Biol Chem 2010;285: Gale NW, Thurston G, Hackett SF, et al. Angiopoietin-2 is required for postnatal angiogenesis and lymphatic patterning, and only the latter role is rescued by Angiopoietin-1. Dev Cell 2002;3: Huang YQ, Li JJ, Hu L, Lee M, Karpatkin S. Thrombin induces increased expression and secretion of angiopoietin-2 from human umbilical vein endothelial cells. Blood 2002;99: Hackett SF, Wiegand S, Yancopoulos G, Campochiaro PA. Angiopoietin-2 plays an important role in retinal angiogenesis. J Cell Physiol 2002;192: Oh H, Takagi H, Suzuma K, Otani A, Matsumura M, Honda Y. Hypoxia and vascular endothelial growth factor selectively up-regulate angiopoietin-2 in bovine microvascular endothelial cells. J Biol Chem 1999;274: Rangasamy S, Srinivasan R, Maestas J, McGuire PG, Das A. A potential role for angiopoietin 2 in the regulation of the blood-retinal barrier in diabetic retinopathy. Invest Ophthalmol Vis Sci 2011;52: Fiedler U, Scharpfenecker M, Koidl S, et al. The Tie-2 ligand angiopoietin-2 is stored in and rapidly released upon stimulation from endothelial cell Weibel- Palade bodies. Blood 2004;103: Fiedler U, Reiss Y, Scharpfenecker M, et al. Angiopoietin-2 sensitizes endothelial cells to TNF-alpha and has a crucial role in the induction of inflammation. Nat Med 2006;12: Ohashi H, Takagi H, Koyama S, et al. Alterations in expression of angiopoietins and the Tie-2 receptor in the retina of streptozotocin induced diabetic rats. Mol Vis 2004;10: Chew EY, Ambrosius WT, Davis MD, et al.; ACCORD Study Group; ACCORD Eye Study Group. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 2010;363: The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329: UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998; 352: Shih A, Davis FB, Lin HY, Davis PJ. Resveratrol induces apoptosis in thyroid cancer cell lines via a MAPK- and p53-dependent mechanism. J Clin Endocrinol Metab 2002;87: Feit-Leichman RA, Kinouchi R, Takeda M, et al. Vascular damage in a mouse model of diabetic retinopathy: relation to neuronal and glial changes. Invest Ophthalmol Vis Sci 2005;46:
Diabetic Retinopathy., Jeong Hun Kim. S.W.P. and J.-H.Y. contributed equally to this work. Seoul National University Hospital, Seoul , Korea
 Page 1 of 53 Angiopoietin 2 Induces Pericyte Apoptosis via α3β1 Integrin Signaling in Diabetic Retinopathy Sung Wook Park 1, 2, Jang-Hyuk Yun 3, 4, Jin Hyoung Kim 1, Kyu-Won Kim 5, Chung-Hyun Cho 3, 4#
Page 1 of 53 Angiopoietin 2 Induces Pericyte Apoptosis via α3β1 Integrin Signaling in Diabetic Retinopathy Sung Wook Park 1, 2, Jang-Hyuk Yun 3, 4, Jin Hyoung Kim 1, Kyu-Won Kim 5, Chung-Hyun Cho 3, 4#
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Inflammatory basis of diabetic retinopathy. Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH
 Inflammatory basis of diabetic retinopathy Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH Diabetic retinopathy currently is diagnosed based on vascular abnormalities
Inflammatory basis of diabetic retinopathy Tim Kern, PhD Case Western Reserve University and Stokes VA Hospital, Cleveland, OH Diabetic retinopathy currently is diagnosed based on vascular abnormalities
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
The Angiopoietin Axis in Cancer
 Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice.
 Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Dipeptidyl Peptidase-4 Inhibitor Increases Vascular Leakage in Retina through VE-cadherin. Phosphorylation
 Dipeptidyl Peptidase-4 Inhibitor Increases Vascular Leakage in Retina through VE-cadherin Phosphorylation Choon-Soo Lee, PhD, 1,2,4 * Yun Gi Kim, MD, 3 * Hyun-Jai Cho, MD, 1,2,3 Jonghanne Park, MD, 1,2,3
Dipeptidyl Peptidase-4 Inhibitor Increases Vascular Leakage in Retina through VE-cadherin Phosphorylation Choon-Soo Lee, PhD, 1,2,4 * Yun Gi Kim, MD, 3 * Hyun-Jai Cho, MD, 1,2,3 Jonghanne Park, MD, 1,2,3
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC
 Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC Supplementary Table 2. Drug content and loading efficiency estimated with F-NMR and UV- Vis Supplementary Table 3. Complete
Supplementary Table 1. Characterization of HNSCC PDX models established at MSKCC Supplementary Table 2. Drug content and loading efficiency estimated with F-NMR and UV- Vis Supplementary Table 3. Complete
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
marker. DAPI labels nuclei. Flies were 20 days old. Scale bar is 5 µm. Ctrl is
 Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1. (a) Nos is detected in glial cells in both control and GFAP R79H transgenic flies (arrows), but not in deletion mutant Nos Δ15 animals. Repo is a glial cell marker. DAPI labels
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and
 Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and stomach cancer were stained with SA-β-Gal and nuclear fast
Supplementary Figure 1. SA-β-Gal positive senescent cells in various cancer tissues. Representative frozen sections of breast, thyroid, colon and stomach cancer were stained with SA-β-Gal and nuclear fast
Supplementary Materials and Methods
 Supplementary Materials and Methods Hepatocyte toxicity assay. Freshly isolated hepatocytes were incubated for overnight with varying concentrations (-25 µm) of sodium glycochenodeoxycholate (GCDC) or
Supplementary Materials and Methods Hepatocyte toxicity assay. Freshly isolated hepatocytes were incubated for overnight with varying concentrations (-25 µm) of sodium glycochenodeoxycholate (GCDC) or
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
p = formed with HCI-001 p = Relative # of blood vessels that formed with HCI-002 Control Bevacizumab + 17AAG Bevacizumab 17AAG
 A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival
 Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
Supplementary Information for Proteomic profiling of small-molecule inhibitors reveals dispensability of MTH1 for cancer cell survival Tatsuro Kawamura 1, Makoto Kawatani 1, Makoto Muroi, Yasumitsu Kondoh,
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Supplemental information
 Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
Carcinoemryonic antigen-related cell adhesion molecule 6 (CEACAM6) promotes EGF receptor signaling of oral squamous cell carcinoma metastasis via the complex N-glycosylation y Chiang et al. Supplemental
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Supplementary Figure 1
 CD31 FN Supplementary Figure 1 a Multivariate Cox regression analysis of predicting factors for disease-free and overall survival in 435 HNSCC patients b FN staining in whole sections of HNSCC c FN expression
CD31 FN Supplementary Figure 1 a Multivariate Cox regression analysis of predicting factors for disease-free and overall survival in 435 HNSCC patients b FN staining in whole sections of HNSCC c FN expression
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Supplementary Information
 Supplementary Information Supplementary Figure 1. EBV-gB 23-431 mainly exists as trimer in HEK 293FT cells. (a) Western blotting analysis for DSS crosslinked FLAG-gB 23-431. HEK 293FT cells transfected
Supplementary Information Supplementary Figure 1. EBV-gB 23-431 mainly exists as trimer in HEK 293FT cells. (a) Western blotting analysis for DSS crosslinked FLAG-gB 23-431. HEK 293FT cells transfected
Supplemental Data. TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim.
 Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Essential Medium, containing 10% fetal bovine serum, 100 U/ml penicillin and 100 µg/ml streptomycin. Huvec were cultured in
 Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
CD31 5'-AGA GAC GGT CTT GTC GCA GT-3' 5 ' -TAC TGG GCT TCG AGA GCA GT-3'
 Table S1. The primer sets used for real-time RT-PCR analysis. Gene Forward Reverse VEGF PDGFB TGF-β MCP-1 5'-GTT GCA GCA TGA ATC TGA GG-3' 5'-GGA GAC TCT TCG AGG AGC ACT T-3' 5'-GAA TCA GGC ATC GAG AGA
Table S1. The primer sets used for real-time RT-PCR analysis. Gene Forward Reverse VEGF PDGFB TGF-β MCP-1 5'-GTT GCA GCA TGA ATC TGA GG-3' 5'-GGA GAC TCT TCG AGG AGC ACT T-3' 5'-GAA TCA GGC ATC GAG AGA
Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System
 Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
Japan-Mexico Workshop on Pharmacology and Nanobiology Feb. 25, 2009; Universidad Nacional Autönoma de Mëxico, Mexico City Angiostasis and Angiogenesis Regulated by Angiopoietin1-Tie2 Receptor System Shigetomo
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were
 Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure 1. Validation of astrocytes. Primary astrocytes were separated from the glial cultures using a mild trypsinization protocol. Anti-glial fibrillary acidic protein (GFAP) immunofluorescent
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was
 Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at
 Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
Supplementary Figure 1. EC-specific Deletion of Snail1 Does Not Affect EC Apoptosis. (a,b) Cryo-sections of WT (a) and Snail1 LOF (b) embryos at E10.5 were double-stained for TUNEL (red) and PECAM-1 (green).
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Fang et al. NMuMG. PyVmT unstained Anti-CCR2-PE MDA-MB MCF MCF10A
 A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
A NMuMG PyVmT 16.5+.5 47.+7.2 Fang et al. unstained Anti-CCR2-PE 4T1 Control 37.6+6.3 56.1+.65 MCF1A 16.1+3. MCF-7 3.1+5.4 MDA-M-231 42.1+5.5 unstained Secondary antibody only Anti-CCR2 SUPPLEMENTAL FIGURE
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Annals of Oncology Advance Access published January 10, 2005
 Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by
 Nakano et al. Supplementary information 1. Supplementary Figure 2. Methods 3. References Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into
Nakano et al. Supplementary information 1. Supplementary Figure 2. Methods 3. References Bone marrow-derived mesenchymal stem cells improve diabetes-induced cognitive impairment by exosome transfer into
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 EGFR inhibition activates signaling pathways (a-b) EGFR inhibition activates signaling pathways (a) U251EGFR cells were treated with erlotinib (1µM) for the indicated times followed
Supplementary Figure 1 EGFR inhibition activates signaling pathways (a-b) EGFR inhibition activates signaling pathways (a) U251EGFR cells were treated with erlotinib (1µM) for the indicated times followed
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure S1: Fibroblast-induced elongation of cancer cells requires direct contact with living fibroblasts. A. Representative images of HT29-GFP cultured in the presence
SUPPLEMENTARY FIGURES Supplementary Figure S1: Fibroblast-induced elongation of cancer cells requires direct contact with living fibroblasts. A. Representative images of HT29-GFP cultured in the presence
The Natural History of Diabetic Retinopathy and How Primary Care Makes A Difference
 The Natural History of Diabetic Retinopathy and How Primary Care Makes A Difference We will discuss - How exactly does blood sugar control affect retinopathy? - What are other factors that we measure in
The Natural History of Diabetic Retinopathy and How Primary Care Makes A Difference We will discuss - How exactly does blood sugar control affect retinopathy? - What are other factors that we measure in
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information
 Supplementary Information Figure S1. Int6 gene silencing efficiency. (A) Western Blot analysis of Int6 expression at different times after sirna transfection. Int6 expression is strongly silenced in Int6
Supplementary Information Figure S1. Int6 gene silencing efficiency. (A) Western Blot analysis of Int6 expression at different times after sirna transfection. Int6 expression is strongly silenced in Int6
Supplementary Figure 1 ITGB1 and ITGA11 increase with evidence for heterodimers following HSC activation. (a) Time course of rat HSC activation
 Supplementary Figure 1 ITGB1 and ITGA11 increase with evidence for heterodimers following HSC activation. (a) Time course of rat HSC activation indicated by the detection of -SMA and COL1 (log scale).
Supplementary Figure 1 ITGB1 and ITGA11 increase with evidence for heterodimers following HSC activation. (a) Time course of rat HSC activation indicated by the detection of -SMA and COL1 (log scale).
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
RayBio KinaseSTAR TM Akt Activity Assay Kit
 Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial
 Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial Jeffrey S. Heier, MD On behalf of the RUBY Investigators Ophthalmic Consultants of Boston
Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial Jeffrey S. Heier, MD On behalf of the RUBY Investigators Ophthalmic Consultants of Boston
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Figure 1:
 Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
Supplementary Figure 1: (A) Whole aortic cross-sections stained with Hematoxylin and Eosin (H&E), 7 days after porcine-pancreatic-elastase (PPE)-induced AAA compared to untreated, healthy control aortas
A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified
 Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
Cell culture and animal model A549 and A549-fLuc cells were maintained in high glucose Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum at 37 C in humidified atmosphere containing
SUPPLEMENTARY DATA. Assessment of cell survival (MTT) Assessment of early apoptosis and cell death
 Overexpression of a functional calcium-sensing receptor dramatically increases osteolytic potential of MDA-MB-231 cells in a mouse model of bone metastasis through epiregulinmediated osteoprotegerin downregulation
Overexpression of a functional calcium-sensing receptor dramatically increases osteolytic potential of MDA-MB-231 cells in a mouse model of bone metastasis through epiregulinmediated osteoprotegerin downregulation
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
MicroRNA 98 suppresses cell growth and invasion of retinoblastoma via targeting the IGF1R/k Ras/Raf/MEK/ERK signaling pathway
 INTERNATIONAL JOURNAL OF ONCOLOGY 54: 807-820, 2019 MicroRNA 98 suppresses cell growth and invasion of retinoblastoma via targeting the IGF1R/k Ras/Raf/MEK/ERK signaling pathway LONG GUO 1, YU BAI 2, SHUZHE
INTERNATIONAL JOURNAL OF ONCOLOGY 54: 807-820, 2019 MicroRNA 98 suppresses cell growth and invasion of retinoblastoma via targeting the IGF1R/k Ras/Raf/MEK/ERK signaling pathway LONG GUO 1, YU BAI 2, SHUZHE
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 )
 770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the
 Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
Supplementary Figure 1. HOPX is hypermethylated in NPC. (a) Methylation levels of HOPX in Normal (n = 24) and NPC (n = 24) tissues from the genome-wide methylation microarray data. Mean ± s.d.; Student
http / / cjbmb. bjmu. edu. cn Chinese Journal of Biochemistry and Molecular Biology COX-2 NTera-2 NTera-2 RT-PCR FasL caspase-8 caspase-3 PARP.
 ISSN 1007-7626 CN 11-3870 / Q http / / cjbmb bjmu edu cn Chinese Journal of Biochemistry and Molecular Biology 2012 7 28 7 630 ~ 636 NTera-2 ** ** * 410081 COX-2 NTera-2 MTT NTera-2 NTera-2 Hoechest 33258
ISSN 1007-7626 CN 11-3870 / Q http / / cjbmb bjmu edu cn Chinese Journal of Biochemistry and Molecular Biology 2012 7 28 7 630 ~ 636 NTera-2 ** ** * 410081 COX-2 NTera-2 MTT NTera-2 NTera-2 Hoechest 33258
SUPPLEMENT. Materials and methods
 SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2)
 Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
Supplemental Methods Cells and reagents. Synaptopodin knockdown (1) and dynamin knockdown (2) podocytes were cultured as described previously. Staurosporine, angiotensin II and actinomycin D were all obtained
University of Groningen. Vasoregression in incipient diabetic retinopathy Pfister, Frederick
 University of Groningen Vasoregression in incipient diabetic retinopathy Pfister, Frederick IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from
University of Groningen Vasoregression in incipient diabetic retinopathy Pfister, Frederick IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from
Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways
![Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways](/thumbs/96/128474991.jpg) Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2016 Supplementary Data Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2016 Supplementary Data Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Supporting Information
 Supporting Information Identification of Novel ROS Inducers: Quinone Derivatives Tethered to Long Hydrocarbon Chains Yeonsun Hong,, Sandip Sengupta,, Wooyoung Hur, *, Taebo Sim,, * KU-KIST Graduate School
Supporting Information Identification of Novel ROS Inducers: Quinone Derivatives Tethered to Long Hydrocarbon Chains Yeonsun Hong,, Sandip Sengupta,, Wooyoung Hur, *, Taebo Sim,, * KU-KIST Graduate School
Stewart et al. CD36 ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer
 NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Financial Disclosures
 Financial Disclosures DB is a scientific advisor for Regeneron/ Bayer and Genentech/ Roche & a DB member of the Regeneron Combination Products Steering Committee RCH receives grant support from Regeneron/
Financial Disclosures DB is a scientific advisor for Regeneron/ Bayer and Genentech/ Roche & a DB member of the Regeneron Combination Products Steering Committee RCH receives grant support from Regeneron/
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
DOI:.38/ncb3399 a b c d FSP DAPI 5mm mm 5mm 5mm e Correspond to melanoma in-situ Figure a DCT FSP- f MITF mm mm MlanaA melanoma in-situ DCT 5mm FSP- mm mm mm mm mm g melanoma in-situ MITF MlanaA mm mm
Plasmids Western blot analysis and immunostaining Flow Cytometry Cell surface biotinylation RNA isolation and cdna synthesis
 Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
Plasmids psuper-retro-s100a10 shrna1 was constructed by cloning the dsdna oligo 5 -GAT CCC CGT GGG CTT CCA GAG CTT CTT TCA AGA GAA GAA GCT CTG GAA GCC CAC TTT TTA-3 and 5 -AGC TTA AAA AGT GGG CTT CCA GAG
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells
 CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
CD34 + VEGFR-3 + progenitor cells have a potential to differentiate towards lymphatic endothelial cells Tan YZ et al. J Cell Mol Med. (2014 Mar;18(3):422-33) Denise Traxler-Weidenauer April 2014 Introduction
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Supplementary Figure 1. Blood glucose and insulin levels in mice during 4-day infusion.
 Supplementary Figure 1. Blood glucose and insulin levels in mice during 4-day infusion. (A-B) WT and HT mice infused with saline or glucose had overlapping achieved blood glucose and insulin levels, necessitating
Supplementary Figure 1. Blood glucose and insulin levels in mice during 4-day infusion. (A-B) WT and HT mice infused with saline or glucose had overlapping achieved blood glucose and insulin levels, necessitating
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Page 39 of 44. 8h LTA & AT h PepG & AT h LTA
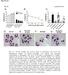 Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
SUPPLEMENTARY DATA. Supplementary Table 1. Characteristics of Subjects.
 Supplementary Table 1. Characteristics of Subjects. a includes one patient who had an aqueous sample taken from the same eye twice b includes one patients who had an aqueous sample taken from the same
Supplementary Table 1. Characteristics of Subjects. a includes one patient who had an aqueous sample taken from the same eye twice b includes one patients who had an aqueous sample taken from the same
Epidemiology and Pathophysiology of Diabetic Retinopathy
 Epidemiology and Pathophysiology of Diabetic Retinopathy Vincent Reppucci, MD Director, Retina Service Mt. Sinai St. Luke s-roosevelt Hospital Attending Physician, Retina Service New York Eye and Ear Infirmary
Epidemiology and Pathophysiology of Diabetic Retinopathy Vincent Reppucci, MD Director, Retina Service Mt. Sinai St. Luke s-roosevelt Hospital Attending Physician, Retina Service New York Eye and Ear Infirmary
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
Supplementary Information and Figure legends
 Supplementary Information and Figure legends Table S1. Primers for quantitative RT-PCR Target Sequence (5 -> 3 ) Target Sequence (5 -> 3 ) DAB2IP F:TGGACGATGTGCTCTATGCC R:GGATGGTGATGGTTTGGTAG Snail F:CCTCCCTGTCAGATGAGGAC
Supplementary Information and Figure legends Table S1. Primers for quantitative RT-PCR Target Sequence (5 -> 3 ) Target Sequence (5 -> 3 ) DAB2IP F:TGGACGATGTGCTCTATGCC R:GGATGGTGATGGTTTGGTAG Snail F:CCTCCCTGTCAGATGAGGAC
