Sin1 phosphorylation impairs mtorc2 complex integrity and inhibits downstream Akt signaling to suppress tumorigenesis
|
|
|
- Anastasia Grant
- 5 years ago
- Views:
Transcription
1 Sin1 phosphorylation impairs mtorc2 complex integrity and inhibits downstream Akt signaling to suppress tumorigenesis The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Liu, P., W. Gan, H. Inuzuka, A. S. Lazorchak, D. Gao, O. Arojo, D. Liu, et al Sin1 phosphorylation impairs mtorc2 complex integrity and inhibits downstream Akt signaling to suppress tumorigenesis. Nature cell biology 15 (11): /ncb2860. doi: /ncb Published Version doi: /ncb2860 Citable link Terms of Use This article was downloaded from Harvard University s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at nrs.harvard.edu/urn-3:hul.instrepos:dash.current.terms-ofuse#laa
2 NIH Public Access Author Manuscript Published in final edited form as: Nat Cell Biol November ; 15(11):. doi: /ncb2860. Sin1 phosphorylation impairs mtorc2 complex integrity and inhibits downstream Akt signaling to suppress tumorigenesis Pengda Liu 1, Wenjian Gan 1, Hiroyuki Inuzuka 1, Adam S Lazorchak 2, Daming Gao 1, Omotooke Arojo 2, Dou Liu 2, Lixin Wan 1, Bo Zhai 3, Yonghao Yu 3,4, Min Yuan 5, Byeong Mo Kim 6, Shavali Shaik 1, Suchithra Menon 7, Steven P. Gygi 3, Tae Ho Lee 6, John M Asara 5, Brendan D. Manning 7, John Blenis 3, Bing Su 2, and Wenyi Wei 1,8 1 Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA Department of Immunobiology and The Vascular Biology and Therapeutics Program, Yale Medical School, New Haven, CT Department of Cell Biology, Harvard Medical School, Boston, MA Department of Biochemistry, UT Southwestern Medical Center, Dallas, TX Department of Medicine, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA Division of Gerontology, Department of Medicine, Beth Israel Deaconess Medical Center, Boston, MA Department of Genetics & Complex Diseases, Harvard School of Public Health, Boston, MA Abstract The mechanistic target of rapamycin (mtor) functions as a critical regulator of cellular growth and metabolism by forming multi-component, yet functionally distinct complexes mtorc1 and mtorc2. Although mtorc2 has been implicated in mtorc1 activation, little is known about how mtorc2 is regulated. Here we report that phosphorylation of Sin1 at T86 and T398 suppresses mtorc2 kinase activity by dissociating Sin1 from mtorc2. Importantly, Sin1 phosphorylation, triggered by S6K or Akt, in a cellular context-dependent manner, inhibits not only insulin/igf-1-mediated, but also PDGF or EGF-induced Akt phosphorylation by mtorc2, demonstrating a negative regulation of mtorc2 independent of IRS-1 and Grb10. Lastly, a cancer patient-derived Sin1-R81T mutation impairs Sin1 phosphorylation, leading to hypermtorc2 activation via bypassing this negative regulation. Together, our work reveals a Sin1 phosphorylation-dependent mtorc2 regulation, providing a potential molecular mechanism by which mutations in the mtorc1/s6k/sin1 signaling axis might cause aberrant hyper-activation of mtorc2/akt that facilitates tumorigenesis. 8 To whom correspondence should be addressed: Wenyi Wei, Ph.D., Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School, 330 Brookline Ave, Boston, MA 02215, Phone: , wwei2@bidmc.harvard.edu. Author Contributions P.L., W.G and H.I. performed most of the experiments with assistance from D.G., L.W., A.L., S.S. and O.A. W.W. and B.S. designed the experiments and supervised the study. P.L. and W.W. wrote the manuscript. All authors commented on the manuscript. Competing Financial Interests The authors declare no competing financial interests. Note: Supplementary Information is available in the online version of the paper.
3 Liu et al. Page 2 Introduction Results mtor is a highly conserved important regulator of cell growth and proliferation in a plethora of biological settings in all eukaryotes 1 8. As such, deregulated mtor function leads to a variety of human diseases, ranging from cancer 5,9 to immune dysfunction 3 and diabetes 4,10. mtor functions as a critical and essential catalytic core in at least two known functionally distinct complexes, mtorc1 and mtorc2 3,5. A unique subset of associated proteins defines each complex, such as Raptor 11 in mtorc1, or Rictor 12 and Sin in mtorc2. Biologically, mtorc2 mainly promotes cell proliferation and survival 2,16 through phosphorylation of the AGC kinase family members Akt and SGK 6, in addition to its initially defined role in regulating cell skeletal organization 17. On the other hand, mtorc1 directly promotes mrna translation and protein synthesis by phosphorylating S6K1 and 4EBP1 18, inhibits autophagy through phosphorylating ULK1 19 and indirectly enhances ribosome biogenesis via promoting nucleophosmin (NPM) oncogene translation 20. As most mtorc1 functions are high energy consuming, regulation of mtorc1 activity is tightly coupled to the energy status of the cell and regulated by nutrients, energy, stress and growth factors, thereby ensuring that cells stop growing under unfavorable conditions 1,8,21,22. Compared to well-defined mechanisms of mtorc1 activation such as mtorc2/aktmediated phosphorylation of TSC2 23,24 or PRAS40 25,26, the upstream signaling that governs mtorc2 activation is just beginning to be appreciated. To this end, mtorc2 s association with ribosome was recently found to be necessary for its activation 4. Furthermore, mtorc1/s6k-mediated phosphorylation of IRS-1 27,28 and Grb10 28,29 also constitutes negative feedback mechanisms to block mtorc2 activation by insulin/igf-1. However, both IRS-1 and Grb10 function by suppressing insulin/igf-1 signaling upstream of PI3K to affect both mtorc1 and mtorc2. Thus, it remains elusive whether mtorc1 could directly regulate mtorc2/akt without broadly suppressing the PI3K pathway and how mtorc1 suppresses mtorc2/akt in stimulation conditions other than insulin/igf-1. Here we show that in response to a wide spectrum of stimuli including insulin, IGF-1, PDGF and EGF, phosphorylation of Sin1 dissociates Sin1 from mtorc2 to terminate mtorc2 kinase activity, revealing a negative regulation of mtorc2 function independent of the previously identified negative feedback regulators IRS-1 and Grb10 in suppressing mtorc2. S6K phosphorylates Sin1 on both T86 and T398 sites In keeping with previous reports 27 30, an inverse correlation between mtorc1/s6k and mtorc2 activities was confirmed (Supplementary Fig. S1a d). Intriguingly, the critical role of Grb10 29,30 and IRS-1 27,28 in regulation of mtorc2/akt was found to be restricted to insulin/igf-1, but not PDGF or EGF stimulation (Supplementary Fig. S1e g). Consistently, inhibition of mtorc1/s6k signaling by a specific S6K1 inhibitor, S6K1-I 31 or mtorc1 inhibitor, rapamycin 11,32,33 could still augment Akt activation in TSC2 / MEFs depleted of endogenous IRS-1 and/or Grb10 (Supplementary Fig. S1h m), advocating that mtorc1/s6k could exert its negative regulation of mtorc2/akt following PDGF or EGF stimulation through uncharacterized negative feedback loop(s) other than IRS-1 and Grb10. As Rictor and Sin1 are the only two unique essential mtorc2 components 13,34, we reasoned that the mtorc1/s6k-dependent regulation of mtorc2 might occur through Rictor or Sin1. However, S6K-dependent phosphorylation of Rictor does not affect mtorc2 kinase activity 32,35,36, which prompted us to investigate whether Sin1 is the primary target to mediate the regulation of mtorc2 by mtorc1/s6k.
4 Liu et al. Page 3 In line with previous reports 13,15, insulin, IGF-1, PDGF and EGF all could effectively induce Akt S473 phosphorylation in WT but not Sin1 / MEFs (Supplementary Fig. S1n) or Sin1 depleted TSC2 / cells (Supplementary Fig. S1o). This finding prompted us to further examine whether other than IRS-1 and Grb10, modifications of Sin1 may play a critical role in mtorc1-mediated feedback regulation of mtorc2. In support of this hypothesis, phosphorylation of Sin1 was significantly reduced upon inhibition of mtorc1 or S6K, and moderately decreased by the Akt inhibitor, AktVIII (Fig. 1a). Consistently, we detected in vivo Sin1 phosphorylation triggered by S6K1 and to a lesser extent, Akt1, but not other characterized AGC kinases (Fig. 1b and Supplementary Fig. S2a). Notably, S6K1 inhibition led to reduced Sin1 phosphorylation while overexpressing a WT-S6K1 (Fig. 1c), but not a kinase-dead-s6k1 mutant 37 (Supplementary Fig. S2b), augmented Sin1 phosphorylation. Consistently, depletion of TSC2, which resulted in elevated S6K activity, also led to increased Sin1 phosphorylation (Supplementary Fig. S2c). In support of Sin1 being a S6K substrate, we identified two canonical AGC family kinase-recognition motifs (RxRxxpS/ pt) 38 located at T86 and T398, respectively (Fig. 1d). Notably, mutation of both sites to alanines abolished S6K1-mediated Sin1 phosphorylation in cells (Fig. 1e) or in vitro (Fig. 1f). Interestingly, mutation of either T86 or T398 to an alanine did not completely abolish the S6K-dependent phosphorylation of Sin1 (Fig. 1e), indicating that both T86 and T398 might be potential S6K1 sites in vivo. Importantly, phosphorylation of T86 and T398 were detected by mass spectrometry (Supplementary Fig. S2d e). To gain further mechanistic insights into how Sin1 phosphorylation may affect mtorc2 activation under physiological conditions, we developed phospho-specific antibodies against pt86-sin1 or pt398-sin1, respectively (Supplementary Fig. S2f g). Using these antibodies we observed an increase in Sin1-pT86 and Sin1-pT398 upon induction by insulin (Fig. 1g and Supplementary Fig. S2h), IGF-1, PDGF or EGF (Fig. 1h), whereas these phosphorylation events could be attenuated by inactivating S6K1 either through depleting Raptor (Fig. 1h and Supplementary Fig. S2i), or by S6K1-I (Supplementary Fig. S2j). Notably, S6K1 knockdown only partially reduced Sin1 phosphorylation (Supplementary Fig. S2i), suggesting that other S6K isoforms might phosphorylate Sin1. Nevertheless, recombinant S6K1 phosphorylated Sin1 in vitro (Supplementary Fig. S2k) and conversely, rapamycin could attenuate Sin1-pT86 mediated by WT-S6K1 but not a rapamycin-resistant form of S6K1 37 (Supplementary Fig. 2l), further supporting S6K1 as a physiological kinase for Sin1. Both Akt and S6K may phosphorylate Sin1 in a context-dependent manner Although depletion of endogenous Akt1 also led to a moderate decrease in Sin1 phosphorylation, its effects were less compared to inhibiting S6K1 or mtorc1 in our experimental conditions (Supplementary Fig. S2m n). Moreover, Akt might regulate Sin1 phosphorylation indirectly through activating mtorc1/s6k 11,13. In support of this model, Sin1-T86 phosphorylation correlated positively with S6K1 activity (evidenced by ps6) but inversely with Akt phosphorylation (Supplementary Fig. S2o p), indicating that Sin1 phosphorylation by S6K1 might act as a physiological negative regulator of Akt phosphorylation in fibroblasts. However, this conclusion might be cellular contextdependent as a recent study has suggested Akt as the Sin1-T86 phosphorylating kinase in adipocytes 39. To gain further mechanistic insights into upstream kinases responsible for phosphorylating Sin1, we examined the possible role of S6K versus Akt in mediating Sin1 phosphorylation on T86 and T398 in 3T3-L1 cells 40. Consistent with Humphrey et al 39, Sin1-pT86 signals were largely blocked by Akt inhibition, but only moderately reduced by inhibiting mtorc1 or S6K (Supplementary Fig. S2q), confirming Akt as the major Sin1-T86 phosphorylating kinase in 3T3-L1 cells. However, inhibitors targeting either Akt or S6K1 led to a dramatic
5 Liu et al. Page 4 reduction in Sin1-pT398, suggesting that both Akt and S6K are involved in phosphorylating Sin1-T398 in 3T3-L1 adipocytes. On the other hand, in epithelium-derived HeLa cells, in stark contrast to 3T3-L1, inhibition of mtorc1/s6k, rather than Akt, led to a more severe reduction in both Sin1-pT86 and Sin1-pT398 signals (Supplementary Fig S2r), indicating that in HeLa cells, S6K, but not Akt, is the major kinase responsible for Sin1-T86 and -T398 phosphorylation. Collectively, these results implicate that physiological upstream kinase(s) responsible for Sin1-T86 or Sin1-T398 phosphorylation might be tissue-specific or cellular context-dependent. A similar tissue specific phosphorylation atlas of mouse proteins has been established 41. Hence, adipocytes versus epithelial cells might require tightly regulated and specialized, phosphorylation-dependent intracellular signaling, thereby utilizing distinct upstream signaling routes to regulate effector pathways such as mtorc2. As carcinomas primarily derive from epithelial cells 42,43, we chose to focus on understanding how regulation of mtorc2 by S6K-mediated Sin1 phosphorylation contributes to tumorigenesis in the epithelial cell settings. Phosphorylation of Sin1 at both T86 and T398 dissociates Sin1 from the mtorc2 complex As Sin1 is an essential component that governs mtorc2 integrity 13 15, we next examined whether Sin1 phosphorylation on T86 and T398 affects mtorc2 function. Strikingly, compared to WT-Sin1, the Sin1 phospho-mimetic mutant (T86E/T398E, Sin1-EE), failed to interact with the essential mtorc2 components Rictor, mtor or GβL (Fig. 2a and Supplementary Fig. S3a c), indicating that the assembly of a functional mtorc2 complex was prevented by Sin1 phosphorylation. In supporting of this model, S6K1-mediated phosphorylation of Sin1-WT, but not the Sin1 phospho-deficient mutant (T86A/T398A, Sin1-AA), reduced Sin1 interaction with Rictor (Fig. 2b), mtor (Fig. 2c) and GβL (Fig. 2d). Notably, disruption of the Sin1/Rictor interaction required Sin1 phosphorylation on both T86 and T398 (Supplementary Fig. S3d,e). To further investigate the underlying molecular mechanism(s), we performed gel-filtration assays to examine mtorc2 complex assembly in cells, and observed that WT-Sin1, but not Sin1-EE, co-eluted with mtorc2 components including Rictor, mtor, and GβL (Fig. 2e). Furthermore, depletion of TSC2 led to elevated mtorc1/s6k activity and shifted a significant portion of Sin1 to mtorc2- free fractions (Supplementary Fig. S3f,g). Strikingly, the mtorc2-free Sin1 species were enriched with pt86-sin1 (Supplementary Fig. S3f). Consistently, less Rictor was detected in association with Sin1 in TSC2 / cells (Fig. 2f), whereas inhibiting S6K1 activity in TSC2 / cells by S6K1-I partially restored Rictor interaction with Sin1 (Supplementary Fig. S3h,i), supporting a crucial role of S6K in regulating the Sin1-Rictor interaction. To further understand the role of each Sin1 phosphorylation event in mediating mtorc2 complex organization, we truncated Sin1 into four regions (Fig. 2g) and observed that Sin1 mainly interacts with Rictor through its N-terminal region, with mtor kinase domain via its PH domain, and with GβL through its N, RBD and PH domains (Fig. 2h). Interestingly, phosphorylation of Sin1 at T86 in the N-terminal region abolished Sin1-N interaction with Rictor (Fig. 2i), while phosphorylation on T398 in the PH domain reduced Sin1 interaction with the mtor kinase domain (Fig. 2j), indicating the distinct role of each phosphorylation site in possibly mediating the organization of the mtorc2 complex, further supporting that phosphorylation of both T86 and T398 is required to functionally inactivate mtorc2. More importantly, under physiological stimulation conditions such as EGF (Fig. 3a and Supplementary Fig. S4a) or insulin (Fig. 3b and Supplementary Fig. S4b) induction, we observed an inverse correlation between the induced Sin1 phosphorylation and a dissociation of Sin1 from Rictor or mtor, which could be abolished by mutating either T86 or T398 to alanine (Fig. 3c), or by rapamycin treatment (Supplementary Fig. S4c f), demonstrating a physiological role of Sin1 phosphorylation in negatively regulating mtorc2 function. Notably, compared with Sin1-WT, impairing Sin1 phosphorylation by
6 Liu et al. Page 5 mutating either T86 or T398 (Fig. 3d), inhibiting S6K activity (Fig. 3e and Supplementary Fig. S4g), or depleting endogenous Raptor (Supplementary Fig. S4h,i), led to sustained Akt phosphorylation under EGF or insulin stimulation. Conversely, activating S6K by either depletion of endogenous TSC2 (Supplementary Fig. S4j) or genetic ablation of TSC2 (Supplementary Fig. S4k), resulted in reduced Akt S473 phosphorylation. In keeping with previous reports 27,44, Akt phosphorylation in response to various stimuli was attenuated in TSC2 / cells with elevated S6K activity (Supplementary Fig. S4l), confirming a negative role for S6K-mediated phosphorylation of Sin1 in Akt activation. Phosphorylation of Sin1 on both T86 and T398 suppresses mtorc2-mediated activation of Akt Notably, Sin1 phosphorylation did not affect Sin1 stability (Supplementary Fig. S4m,n). However, phosphorylation of Sin1 specifically reduced its interaction with the mtorc2 substrate, Akt1 (Fig. 4a c) but not with another characterized mtorc2 substrate SGK1 45,46 (Fig. 4d e), nor the other AGC kinase S6K1 (Fig. 4f g), which might explain why Sin1 phosphorylation leads to impaired Akt activation. To further verify the critical roles of Sin1 phosphorylation in regulating mtorc2 kinase activity, we performed in vitro kinase assays to show that unlike Sin1-WT or Sin1-AA, immunoprecipitated Sin1-EE failed to phosphorylate GST-Akt1 in vitro, presumably due to its deficiency in forming a functional mtorc2 complex (Fig. 5a b). By coupling gel filtration with in vitro kinase assays, we demonstrated that only the gel-filtration fractions with molecular weights corresponding to the full mtorc2 complex retained the mtorc2 kinase activity towards GST-Akt in vitro (Fig. 5c d). Strikingly, consistent with previous studies 47 49, insulin stimulation led to an oscillated pattern of Akt activation with reduced Akt phosphorylation after prolonged treatment in various cell lines (Supplementary Fig. S5a c), a phenotype observed in many signaling pathways 47,50. However, the underlying mechanism of the oscillation of Akt phosphorylation remains not fully understood. Notably, prolonged (45 minutes of) insulin treatment led to a severe reduction of mtorc2-mediated in vitro phosphorylation of Akt in part by inducing pt86-sin1 (Fig. 5c d) to disrupt the mtorc2 complex integrity. More importantly, rapamycin treatment effectively restored mtorc2 complex integrity and kinase activity under prolonged EGF stimulation (Fig. 5e), emphasizing that S6K negatively regulates mtorc2 activity in part through phosphorylating Sin1, serving as a molecular switch to shut down EGF or insulin-induced activation of mtorc2/akt in a timely fashion (Supplementary Fig. S5d). Given the critical role of the mtorc2/akt signaling in cell survival and tumorigenesis, we next examined the biological significance of S6K-mediated Sin1 phosphorylation on mtorc2 function. Consistent with in vitro kinase assay results (Fig. 5a), Sin1-WT, but not Sin1-EE, rescued in vivo Akt-S473 phosphorylation in Sin1 / MEFs (Fig. 6a,b and Supplementary Fig. S6a) 13,15. Furthermore, in echoing the in vitro kinase results (Fig. 6c), immuno-purified endogenous Akt1 from Sin1-EE-expressing Sin1 / MEFs was deficient in phosphorylating the well-characterized Akt substrate, crosstide 51,52 in vitro (Fig. 6d). Consistently, compared with Sin1-WT expressing Sin1 / cells, Sin1 / cells expressing Sin1-EE exhibited reduced levels of phospho-foxos (Fig. 6a) 53, thereby more sensitive to cell death 54,55 induced by etoposide (Fig. 6e,f and Supplementary Fig. S6b e) or cisplatin (Fig. 6g). Similar apoptotic sensitization was observed after introducing Sin1-EE, in comparison to Sin1-WT, into Sin1-depleted OVCAR5 cells (Supplementary Fig. S6f i, supporting a physiological role of Sin1 phosphorylation in regulating cellular survival in part through the Akt/FOXO signaling pathway 56,57.
7 Liu et al. Page 6 The ovarian cancer patient-derived Sin1-R81T mutation displayed elevated oncogenic activity in part by bypassing Sin1 phosphorylation-mediated suppression of mtorc2/akt signaling Interestingly, two natural Sin1 mutations (R81T and S84L) were recently identified in ovarian cancer patients 58 and skin cancer patients, respectively (Fig. 7a) from two publically available cancer genome databases: the Cosmic database ( cancer.sanger.ac.uk/cancergenome/projects/cosmic/) 59 and the CBio database ( Notably, these mutations appear to disrupt the canonical S6K phosphorylation motif (Fig. 7a), and indeed we observed a significant reduction in pt86 in both Sin1-R81T (Fig. 7b) and S84L (Supplementary Fig. S7a b) mutants, suggesting that they functionally mimicked Sin1-T86A. More importantly, a synthesized pt86-containing peptide could be efficiently recognized by the Sin1-pT86 antibody regardless of Sin1-R81T mutation status (Fig. 7c), excluding a secondary antibody effect. As the R81T mutant impairs the stringent AGC substrate motif, we focused on studying the pathophysiological effects of this mutant in cells. We found that Sin1-pT86 signal was nearly abolished in the Sin1-R81T mutant in response to a wide spectrum of stimuli, including insulin, IGF-1, PDGF and EGF (Fig. 7d). Unlike WT-Sin1 (Fig. 3b c), R81T-Sin1 demonstrated a relatively sustained interaction with mtorc2 components (Fig. 7e and Supplementary Fig. S7c,d), resulting in prolonged mtorc2 kinase activity towards phosphorylating Akt in vitro (Fig. 5d). In keeping with these results, in response to various external stimuli including EGF (Fig. 7f and Supplementary Fig. S7e), insulin (Fig. 7g) and PDGF (Supplementary Fig. S7f), there is a more sustained Akt activation in Sin1-R81T compared with Sin1-WT expressing OVCAR5 cells. These results support a model that the R81T mutation may enhance tumorigenesis in part through augmenting Akt activation (Supplementary Fig. S7g). Biologically, expression of Sin1-R81T in either Sin1 / MEFs or shsin1-ovcar5 cells led to elevated phosphorylation of Akt and the Akt substrate FOXOs (Supplementary Fig. S8a,b), which subsequently conferred resistance to etoposide (Fig. 8a and Supplementary Fig. S8c g) or cisplatin (Fig. 8b,c). Moreover, re-introduction of R81T, but not WT-Sin1, significantly promoted the anchorage-independent growth of Sin1-depleted OVCAR5 cells, arguing that R81T is a gain-of-function oncogenic mutation that favors cellular transformation (Fig. 8d). This finding was echoed by the significantly enhanced in vivo tumor formation ability of R81T expressing OVCAR5 cells in a mouse xenograft model, (Fig. 8e g and Supplementary Fig. 8h). Although further investigation is required, these results provide an important clinical relevance for the oncogenic role of Sin1-R81T mutant, in part by bypassing the mtorc1-s6k-mediated negative feedback regulation on mtorc2/akt activation. Interestingly, we observed that Sin1-R81T-expressing OVCAR5 cells are more resistant to the Akt inhibitor, AktVIII (Supplementary Fig. S8i k), indicating that cancer patients bearing the Sin1-R81T mutation might poorly respond to Akt or mtor inhibitor treatments. Given that abnormal mtorc2/akt activities are associated with numerous human diseases 3,6,7, impaired Sin1 phosphorylation (such as caused by the R81T mutation) might contribute to deregulated mtorc2/akt activity in many types of tissues and diseases. In this regard, we found that rapamycin treatment attenuated Sin1 T86 phosphorylation in splenic B cells freshly isolated from mice, leading to elevated Akt phosphorylation in vivo (Supplementary Fig. S8l o). Similar results were also observed from rapamycin-treated mouse livers (Fig. 8h). Consistently, an inverse correlation between pt86-sin1 and ps473- Akt was observed in a panel of T-ALL cell lines as well as in ovarian caner patient samples (Fig. 8i and Supplementary Fig. S8p r), emphasizing the possible pathological importance of the identified negative feedback mechanism in human cancer settings. However, further
8 Liu et al. Page 7 studies with larger sample sizes are warranted to determine a significant inverse correlation between pt86-sin1 and ps473-akt. Discussion Methods Recent studies have begun to reveal a complicated cross-communication between the two mtor-containing complexes mtorc1 and mtorc2, while the exact molecular mechanism(s) remain largely elusive. Here we define an independent negative feedback regulation through which S6K or Akt directly phosphorylates Sin1 to repress mtorc2 activation in epithelial cells or adipocytes, respectively, thereby providing further molecular insights into a direct and efficient strategy to timely suppress the mtorc2/akt signaling in a possible tissue and cellular context dependent manner. Nonetheless, phosphorylation of both T86 and T398 of Sin1 is required for the complete inactivation of mtorc2 kinase activity in part by dissociating Sin1 from other mtorc2 components. As illustrated in Supplementary Fig. S8s, mtorc1 could be regulated indirectly by mtorc2 in response to extra cellular growth factors, or controlled by multiple in vivo signal fluxes, such as amino acids, nutrients and stresses. As frequently observed in many signaling pathways like those initiated by Ras or PI3K 61, the mtorc2-mediated growth factor signal is only activated transiently followed by a quench of the signaling through multiple negative feedback regulations to ensure that their activations are presented only in a pulse manner 62. Most significantly, an ovarian cancer patient-derived R81T mutation of Sin1 could bypass the identified Sin1 phosphorylation-mediated negative regulation of mtorc2 due to the lack of Sin1 phosphorylation motif, providing a molecular mechanism for the elevated mtorc2/akt activation that could potentially promote tumorigenesis in the ovarian cancer settings. In summary, our work identified a negative regulation mechanism mediated largely by Sin1 phosphorylation on both T86 and T398 to specifically suppress mtorc2 integrity and kinase activity towards Akt-S473 phosphorylation. Moreover, we demonstrated that deregulation of this negative regulation in pathological conditions may contribute to accelerated tumor formation. Methods and any associated references are available in the online version of the paper. Supplementary Material Acknowledgments References Refer to Web version on PubMed Central for supplementary material. We thank Alex Toker, Jianping Guo, Kai Xu, Alan W Lau and Adriana Tron for critical reading of the manuscript, Steve Elledge, Shunsuke Ishii, William Hahn, Dos Sarvassov and Jamie Dempsey for providing valuable reagents, Bruce Spiegelman for providing 3T3-L1 adipocyte cells, Lewis Cantley and Alex Toker for helpful suggestions, and members of the Wei, Blennis and Su laboratories for useful discussions. W.W. is an ACS research scholar and a LLS research scholar. Y. Yu is a CPRIT Scholar (CPRIT R1103) in Cancer Research and a Virginia Murchison Linthicum Scholar in Medical Research. P.L. is NRSA T32 trainee and supported by 5T32HL This work was supported in part by the NIH grants (W.W., GM089763, GM and CA177910; and B.S., AI and PR093728). 1. Zoncu R, Efeyan A, Sabatini DM. mtor: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol. 2011; 12: [PubMed: ]
9 Liu et al. Page 8 2. Wullschleger S, Loewith R, Hall MN. TOR signaling in growth and metabolism. Cell. 2006; 124: [PubMed: ] 3. Laplante M, Sabatini DM. mtor signaling in growth control and disease. Cell. 2012; 149: [PubMed: ] 4. Guertin DA, Sabatini DM. Defining the role of mtor in cancer. Cancer Cell. 2007; 12:9 22. [PubMed: ] 5. Sabatini DM. mtor and cancer: insights into a complex relationship. Nat Rev Cancer. 2006; 6: [PubMed: ] 6. Dazert E, Hall MN. mtor signaling in disease. Curr Opin Cell Biol. 2011; 23: [PubMed: ] 7. Alayev A, Holz MK. mtor signaling for biological control and cancer. J Cell Physiol. 2013; 228: [PubMed: ] 8. Jewell JL, Russell RC, Guan KL. Amino acid signalling upstream of mtor. Nat Rev Mol Cell Biol. 2013; 14: [PubMed: ] 9. Guertin DA, Sabatini DM. An expanding role for mtor in cancer. Trends Mol Med. 2005; 11: [PubMed: ] 10. Inoki K, Corradetti MN, Guan KL. Dysregulation of the TSC-mTOR pathway in human disease. Nat Genet. 2005; 37: [PubMed: ] 11. Kim DH, et al. mtor interacts with raptor to form a nutrient-sensitive complex that signals to the cell growth machinery. Cell. 2002; 110: [PubMed: ] 12. Sarbassov DD, et al. Rictor, a novel binding partner of mtor, defines a rapamycin-insensitive and raptor-independent pathway that regulates the cytoskeleton. Curr Biol. 2004; 14: [PubMed: ] 13. Jacinto E, et al. SIN1/MIP1 maintains rictor-mtor complex integrity and regulates Akt phosphorylation and substrate specificity. Cell. 2006; 127: [PubMed: ] 14. Frias MA, et al. msin1 is necessary for Akt/PKB phosphorylation, and its isoforms define three distinct mtorc2s. Curr Biol. 2006; 16: [PubMed: ] 15. Yang Q, Inoki K, Ikenoue T, Guan KL. Identification of Sin1 as an essential TORC2 component required for complex formation and kinase activity. Genes Dev. 2006; 20: [PubMed: ] 16. Hung CM, Garcia-Haro L, Sparks CA, Guertin DA. mtor-dependent cell survival mechanisms. Cold Spring Harb Perspect Biol. 2012; Jacinto E, et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat Cell Biol. 2004; 6: [PubMed: ] 18. Ma XM, Blenis J. Molecular mechanisms of mtor-mediated translational control. Nat Rev Mol Cell Biol. 2009; 10: [PubMed: ] 19. Chan EY. mtorc1 phosphorylates the ULK1-mAtg13-FIP200 autophagy regulatory complex. Sci Signal. 2009; 2:pe51. [PubMed: ] 20. Pelletier CL, et al. TSC1 sets the rate of ribosome export and protein synthesis through nucleophosmin translation. Cancer Res. 2007; 67: [PubMed: ] 21. Dibble CC, Manning BD. Signal integration by mtorc1 coordinates nutrient input with biosynthetic output. Nat Cell Biol. 2013; 15: [PubMed: ] 22. Efeyan A, Zoncu R, Sabatini DM. Amino acids and mtorc1: from lysosomes to disease. Trends Mol Med. 2012; 18: [PubMed: ] 23. Inoki K, Li Y, Zhu T, Wu J, Guan KL. TSC2 is phosphorylated and inhibited by Akt and suppresses mtor signalling. Nat Cell Biol. 2002; 4: [PubMed: ] 24. Manning BD, Tee AR, Logsdon MN, Blenis J, Cantley LC. Identification of the tuberous sclerosis complex-2 tumor suppressor gene product tuberin as a target of the phosphoinositide 3-kinase/akt pathway. Mol Cell. 2002; 10: [PubMed: ] 25. Sancak Y, et al. PRAS40 is an insulin-regulated inhibitor of the mtorc1 protein kinase. Mol Cell. 2007; 25: [PubMed: ] 26. Vander Haar E, Lee SI, Bandhakavi S, Griffin TJ, Kim DH. Insulin signalling to mtor mediated by the Akt/PKB substrate PRAS40. Nat Cell Biol. 2007; 9: [PubMed: ]
10 Liu et al. Page Harrington LS, et al. The TSC1-2 tumor suppressor controls insulin-pi3k signaling via regulation of IRS proteins. J Cell Biol. 2004; 166: [PubMed: ] 28. Shah OJ, Wang Z, Hunter T. Inappropriate activation of the TSC/Rheb/mTOR/S6K cassette induces IRS1/2 depletion, insulin resistance, and cell survival deficiencies. Curr Biol. 2004; 14: [PubMed: ] 29. Hsu PP, et al. The mtor-regulated phosphoproteome reveals a mechanism of mtorc1-mediated inhibition of growth factor signaling. Science. 2011; 332: [PubMed: ] 30. Yu Y, et al. Phosphoproteomic analysis identifies Grb10 as an mtorc1 substrate that negatively regulates insulin signaling. Science. 2011; 332: [PubMed: ] 31. Pearce LR, et al. Characterization of PF , a novel and highly specific inhibitor of p70 ribosomal S6 kinase (S6K1). The Biochemical journal. 2010; 431: [PubMed: ] 32. Gao D, et al. Rictor forms a complex with Cullin-1 to promote SGK1 ubiquitination and destruction. Mol Cell. 2010; 39: [PubMed: ] 33. Kim DH, et al. GbetaL, a positive regulator of the rapamycin-sensitive pathway required for the nutrient-sensitive interaction between raptor and mtor. Mol Cell. 2003; 11: [PubMed: ] 34. Guertin DA, et al. Ablation in mice of the mtorc components raptor, rictor, or mlst8 reveals that mtorc2 is required for signaling to Akt-FOXO and PKCalpha, but not S6K1. Dev Cell. 2006; 11: [PubMed: ] 35. Dibble CC, Asara JM, Manning BD. Characterization of Rictor phosphorylation sites reveals direct regulation of mtor complex 2 by S6K1. Mol Cell Biol. 2009; 29: [PubMed: ] 36. Treins C, Warne PH, Magnuson MA, Pende M, Downward J. Rictor is a novel target of p70 S6 kinase-1. Oncogene. 2010; 29: [PubMed: ] 37. Romanelli A, Dreisbach VC, Blenis J. Characterization of phosphatidylinositol 3-kinase-dependent phosphorylation of the hydrophobic motif site Thr(389) in p70 S6 kinase 1. J Biol Chem. 2002; 277: [PubMed: ] 38. Obata T, et al. Peptide and protein library screening defines optimal substrate motifs for AKT/ PKB. J Biol Chem. 2000; 275: [PubMed: ] 39. Humphrey SJ, et al. Dynamic Adipocyte Phosphoproteome Reveals that Akt Directly Regulates mtorc2. Cell Metab. 2013; 17: [PubMed: ] 40. Gupta RK, et al. Transcriptional control of preadipocyte determination by Zfp423. Nature. 2010; 464: [PubMed: ] 41. Huttlin EL, et al. A tissue-specific atlas of mouse protein phosphorylation and expression. Cell. 2010; 143: [PubMed: ] 42. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144: [PubMed: ] 43. Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000; 100: [PubMed: ] 44. Zhang H, et al. PDGFRs are critical for PI3K/Akt activation and negatively regulated by mtor. J Clin Invest. 2007; 117: [PubMed: ] 45. Garcia-Martinez JM, Alessi DR. mtor complex 2 (mtorc2) controls hydrophobic motif phosphorylation and activation of serum- and glucocorticoid-induced protein kinase 1 (SGK1). The Biochemical journal. 2008; 416: [PubMed: ] 46. Yan L, Mieulet V, Lamb RF. mtorc2 is the hydrophobic motif kinase for SGK1. The Biochemical journal. 2008; 416:e [PubMed: ] 47. Purvis JE, Lahav G. Encoding and Decoding Cellular Information through Signaling Dynamics. Cell. 2013; 152: [PubMed: ] 48. Ikeda F, Lahav G. Signal transduction and signaling networks. Mol Biol Cell. 2013; 24:676. [PubMed: ] 49. Purvis JE, Lahav G. Decoding the insulin signal. Mol Cell. 2012; 46: [PubMed: ] 50. Purvis JE, et al. p53 dynamics control cell fate. Science. 2012; 336: [PubMed: ]
11 Liu et al. Page Meier R, Alessi DR, Cron P, Andjelkovic M, Hemmings BA. Mitogenic activation, phosphorylation, and nuclear translocation of protein kinase Bbeta. J Biol Chem. 1997; 272: [PubMed: ] 52. Baer K, et al. Activation of a GST-tagged AKT2/PKBbeta. Biochim Biophys Acta. 2005; 1725: [PubMed: ] 53. Zhang X, Tang N, Hadden TJ, Rishi AK. Akt, FoxO and regulation of apoptosis. Biochim Biophys Acta. 2011; 1813: [PubMed: ] 54. Paik JH, et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell. 2007; 128: [PubMed: ] 55. Gan B, et al. FoxOs enforce a progression checkpoint to constrain mtorc1-activated renal tumorigenesis. Cancer Cell. 2010; 18: [PubMed: ] 56. Vogt PK, Jiang H, Aoki M. Triple layer control: phosphorylation, acetylation and ubiquitination of FOXO proteins. Cell Cycle. 2005; 4: [PubMed: ] 57. Brunet A, et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999; 96: [PubMed: ] 58. Network CGAR. Integrated genomic analyses of ovarian carcinoma. Nature. 2011; 474: [PubMed: ] 59. Forbes SA, et al. COSMIC (the Catalogue of Somatic Mutations in Cancer): a resource to investigate acquired mutations in human cancer. Nucleic acids research. 2010; 38:D [PubMed: ] 60. Cerami E, et al. The cbio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer discovery. 2012; 2: [PubMed: ] 61. Shankaran H, et al. Rapid and sustained nuclear-cytoplasmic ERK oscillations induced by epidermal growth factor. Mol Syst Biol. 2009; 5:332. [PubMed: ] 62. Chen JY, Lin JR, Cimprich KA, Meyer T. A Two-Dimensional ERK-AKT Signaling Code for an NGF-Triggered Cell-Fate Decision. Mol Cell. 2012; 45: [PubMed: ]
12 Liu et al. Page 11 Figure 1. S6K phosphorylates Sin1 on both T86 and T398 sites a. Immunoblot (IB) analysis of whole cell lysates (WCL) and Flag-immunoprecipitates (IP) derived from Flag-Sin1-transfected HeLa cells that were serum-starved for 24 hours and then collected after serum stimulation for 30 minutes. Where indicated, the kinase inhibitors (AktVIII: 10 μm, PP242: 1 μm, Rapamycin: 20 nm, S6K1-I: 10 μm) were added together with insulin (100 nm). DMSO was used as a negative control. b. IB analysis of WCL and IP derived from 293T cells transfected with Flag-Sin1 and the indicated HA-tagged constitutive active AGC family kinases. c. IB analysis of WCL and IP derived from 293T cells transfected with Flag-Sin1 and HA- S6K1 (or empty vector as a negative control). Where indicated, the S6K inhibitor was added. d. Schematic illustration of the two evolutionarily conserved putative S6K phosphorylation sites, T86 and T398 within Sin1. e. IB analysis of WCL and IP derived from 293T cells transfected with constitutive active form of S6K (HA-S6K-R3A) and the indicated Flag-Sin1 constructs. f. In vitro kinase assays depicting major S6K phosphorylation sites in Sin1. Please note that the GST-Rictor fusion protein used here is not the full-length protein but rather the truncated version that contains the S6K phosphorylation site T1135 (GST-Rictor-C-tail [aa ]). g. IB analysis of WCL and Flag-IP derived from HeLa cells transfected with the indicated Flag-Sin1 constructs. Where indicated, cells were serum starved for 12 hours and stimulated by 100 nm insulin for 30 minutes before harvesting. h. IB analysis of WCL and Flag-IP derived from HeLa cells depleted of Raptor transfected with Flag-Sin1 (shgfp as a negative control). Where indicated, cells were serum starved for 12 hours and stimulated by the indicated stimuli before harvesting.
13 Liu et al. Page 12 Figure 2. S6K-dependent phosphorylation of Sin1 dissociates Sin1 from the mtorc2 complex a. Immunoblot (IB) analysis of whole cell lysates (WCL) and Flag immunoprecipitates (IP) derived from 293T cells transfected with the indicated Flag-Sin1 constructs (EV: empty vector control; WT: Sin1-WT; AA: Sin1-T86A/T398A; EE: Sin1-T86E/T398E). b d. GST pull down assays to demonstrate that S6K phosphorylation of GST-Sin1-WT-FL (full-length) but not GST-Sin1-T86A/T398A led to impaired interaction with Rictor (b), mtor-kinase domain (KD) (c) or GβL (d). As indicated, GST-Sin1 proteins were phosphorylated by active recombinant S6K in vitro for 1 hour before using as a bait to pull down HA-Rictor (b), mtor-kinase domain (KD) (c) or GβL (d) expressed in 293T cells. e. Gel filtration experiments to illustrate that comparing with WT-Sin1, Sin1-EE lost interaction with the functional mtorc2 complex components in vivo. IB analysis of the indicated fractionations derived from the gel filtration experiment with HeLa cells cotransfected with HA-Sin1-WT and Flag-Sin1-EE constructs. Prior to running cell lysates, the molecular weight resolution of the column was first estimated by running native molecular weight markers (Thyroglobulin ~669KD, Ferritin ~440KD, Aldolase ~158KD, Conalbumin ~75KD and Ovalbumin ~44KD) to determine their retention times on coomassie-stained SDS-PAGE protein gels. f. Deletion of endogenous TSC2, which led to increased S6K kinase activity, resulted in a reduction of Rictor association with Sin1. IB analysis of WCL and anti-sin1-ip derived from TSC2 +/+ or TSC2 / MEFs. g. Schematic representation of the indicated domains of Sin1 as well as the locations of the two Sin1 phosphorylation sites: T86 is in the N-terminal domain while T398 is located in the PH domain. h. GST pull down assays to depict the Sin1 domains that interact with Rictor, mtor-kd or GβL, respectively (* indicates the sizes of GST-Sin1 proteins). I j. GST pull down assays to demonstrate that Sin1 T86E or T398E mutation led to reduced interaction with Rictor (i) or mtor-kd (j), respectively.
14 Liu et al. Page 13 Figure 3. Sin1 phosphorylation induced by various stimuli impairs mtorc2 integrity a. Immunoblot (IB) analysis of whole cell lysates (WCL) and endogenous Sin1 immunoprecipitates (IP) derived from Sin1-WT MEFs that were serum-starved for 24 hours and then collected after EGF (100 ng/ml) stimulation for the indicated time periods. b. IB analysis of WCL and Flag-IP derived from Flag-Sin1-transfected HeLa cells that were serum-starved for 24 hours and then collected after insulin stimulation for the indicated time periods. c. Either Sin1-T86A or T398A mutation impaired the dynamic interaction between Sin1 and other essential mtorc2 components. IB analysis of WCL and Flag-IP derived from HeLa cells transfected with the indicated Flag-Sin1 constructs that were serum starved for 12 hours and then treated with the EGF (100 ng/ml) for the indicated time periods before harvesting for IB analysis. d. Either Sin1-T86A or T398A mutation led to sustained Akt activation upon EGF stimulation. IB analysis of WCL derived from HeLa cells transfected with the indicated Flag-Sin1 constructs that were serum starved for 12 hours and then treated with the EGF (100 ng/ml) for the indicated time periods before harvesting for IB analysis. e. Rapamycin or S6K1-I treatment led to a relatively sustained Akt-pS473 upon insulin stimulation. IB analysis of WCL derived from HeLa cells serum starved for 12 hours and stimulated with 100 ng/ml insulin before harvesting at the indicated time points. Where indicated, 20 nm rapamycin or 10 μm S6K1-I was added.
15 Liu et al. Page 14 Figure 4. Sin1 phosphomimetic mutation is deficient in interacting with the mtorc2 substrate Akt1, but not SGK1 a b. Immunoblot (IB) analysis of whole cell lysates (WCL) and HA (a) or Flag (b) immunoprecipitates (IP) derived from 293T cells that were transfected with the indicated Flag-Sin1 constructs with HA-Akt1. c. Sin1-T86E or Sin1-T398E disrupts Sin1-N-terminus or Sin1-PH domain interaction with Akt1. Indicated GST-Sin1 proteins were used as a bait to pull down HA-Akt1 expressed in 293T cells. d e. IB analysis of WCL and HA-IP derived from 293T cells that were transfected with the indicated Flag-Sin1 constructs with HA-SGK-Δ60. f g. IB analysis of WCL and HA-IP or Flag-IP derived from 293T cells that were transfected with the indicated Flag-Sin1 constructs with HA-S6K1.
16 Liu et al. Page 15 Figure 5. Sin1 phosphorylation suppresses mtorc2 kinase activity towards phosphorylating Akt in vitro a b. 293T cells were transfected with the indicated Flag-tagged Sin1 constructs. 36 hours post-transfection, whole cell lysates (WCL) were collected and the mtorc2 complex was purified by Flag-immunoprecipitation (IP). The Flag-IPs were incubated in vitro with purified GST-Akt1 in the presence of ATP and the kinase reaction buffer. Thirty minutes later, the reaction was stopped by the addition of the loading buffer. Akt1 phosphorylation status was examined by immunoblot (IB) analysis. c d. Prolonged insulin treatment (45 min) induces Sin1 phosphorylation, leading to dissociation of mtorc2 complex and abolished Akt activation. Flag-Sin1-WT or R81T mutant was transfected into HeLa cells and 48 hours later the transfected cells were harvested upon insulin (100 nm) stimulation for 45 min after 12 hours of serum starvation in CHAPS buffer. The whole cell lysates were filtered and run through FPLC superdex 200 column. 500 μl elute was collected for each fraction and 1/20 volume of each fraction was incubated with 2 μg GST-Akt-tail (aa ) at 30 C for 30 min. Afterwards, the resulting samples were resolved on SDS-PAGE and subjected to IB analysis. e f. Rapamycin treatment restored Sin1 phosphorylation resulted from EGF treatment, leading to reassembly of mtorc2 complex and Akt activation. Flag-Sin1-WT was transfected into TSC2-depleted HeLa cells and 24 hours later, the transfected cells were treated with 20 nm rapamycin for another 12 hours prior to EGF stimulation (100 nm) before harvested and analyzed as in (c d).
17 Liu et al. Page 16 Figure 6. Sin1 phosphorylation attenuates mtorc2 kinase activity towards phosphorylating Akt in vivo a. Sin1 / MEFs were transfected with the indicated Flag-Sin1 constructs. 30 hours posttransfection, the resulting cells were serum-starved for 24 hours and then collected after stimulation with insulin for 30 minutes for immunoblot (IB) analysis. b. Reconstitution of Sin1 / MEFs with WT-, but not EE-Sin1, could restore Akt-Ser473 phosphorylation under various stimulation conditions. Sin1 / MEFs were transfected with the indicated Flag-Sin1 constructs and serum-starved for 24 hours before harvesting after treatment with indicated stimuli for IB analysis. c. The indicated Flag-Sin1 constructs were transfected into Sin1 / MEFs and Flag immunoprecipitation (IP) was recovered as the kinase source to phosphorylate GST-Akt1- tail (aa ) in vitro. d. The indicated Flag-Sin1 constructs were transfected into Sin1 / MEFs and endogenous Akt IP was performed as the kinase source to phosphorylate crosstide in vitro. Data was shown as mean ± SD for n= 3 independent experiments. e g. Sin1 / MEFs were transfected with the indicated Flag-Sin1 constructs (with empty vector as a negative control). 24 hours post-transfection, the resulting cells were cultured in 10% FBS-containing medium with the indicated concentrations of etoposide (e) or cisplatin (f) for 48 hours before performing the cell viability assays (e,f) or IB analysis (g). Data was shown as mean ± SD from n=3 independent experiments. * indicates p < 0.05 (Student s t- test).
18 Liu et al. Page 17 Figure 7. The pathological Sin1-R81T mutation led to attenuated Sin1-T86 phosphorylation and sustained Akt phosphorylation upon physiological stimulations a. Schematic illustrations of the ovarian cancer patient-derived Sin1-R81T mutation and the skin cancer patient-derived S84L mutation. b. Immunoblot (IB) analysis of whole cell lysate (WCL) and Flag-immunoprecipitates (IP) derived from 293T cells transfected with the indicated Flag-Sin1 constructs and HA-S6K1. c. Sin1-R81T does not interfere with the Sin1-pT86 antibody to recognize Sin1-pT86. Indicated Sin1 synthetic peptides were dotted on nitrocellulose membrane for IB analysis. d. HeLa cells were transfected with the indicated Flag-Sin1 constructs and serum-starved for 24 hours and then collected after stimulation with the indicated stimuli for 30 minutes for IB analysis and Flag-IP. e. Sin1 / MEFs were transfected with the indicated Flag-Sin1 constructs and serum-starved overnight followed by IB analysis upon 100 ng/ml EGF stimulation for the indicated time points. f g. Sin1 depleted OVCAR5 cells stably expressing Sin1-WT or -R81T were serum starved overnight followed by IB analysis after treatment with 100 ng/ml EGF (f) or 100 nm insulin (g) for the indicated time points.
19 Liu et al. Page 18 Figure 8. The pathological Sin1-R81T mutation displayed elevated oncogenic activity in part by bypassing Sin1 phosphorylation-mediated negative regulation of Akt-pS473 a c. Sin1 / MEFs were transfected with the indicated Flag-Sin1 constructs and were treated with the indicated concentrations of etoposide (a) or cisplatin (b,c) for 48 hours before performing the cell viability assays (a,b) or immunoblot (IB) analysis (c). Data was shown as mean ± SD from n=3 independent experiments. * indicates p < 0.05 (t-test). d. Soft agar assays for Sin1-depleted OVCAR5 cells stably expressing EV, WT or R81T. Data was presented as mean ± SD from n=3 independent experiments. e f. Growth curves (e) and mass of the dissected tumors (f) from xenograft experiments with the indicated cells injected subcutaneously into n=10 mice for each cell line. The visible tumors were measured at the indicated days. Error bars, ±SEM and * indicates p < 0.05 (ttest). g. Representative images of the dissected tumors presented in Figure 8e,f. h. Eight-week-old mice were fasted overnight and then refed for 6 hours following a 30 min pretreatment with vehicle or rapamycin (10 mg/kg). N=4 mice per condition. Livers were dissected and liver lysates were subjected to IB analysis. i. Four representative images of IHC with indicated Sin1 and Akt phosphorylation status out of 58 ovarian patient samples under 400x magnification. Scale bar represents 100 μm.
HA-Sin1-N (aa 1-137), HA-Sin1-CRIM (aa ) and HA-Sin1-RBD (aa ) were constructed by
 EXTENDED METHODS Plasmids HA-Sin1-N (aa 1-137), HA-Sin1-CRIM (aa 138-266) and HA-Sin1-RBD (aa 267-376) were constructed by cloning the corresponding cdnas into psuper-ha-cdna3 vector via BamHI/BglII and
EXTENDED METHODS Plasmids HA-Sin1-N (aa 1-137), HA-Sin1-CRIM (aa 138-266) and HA-Sin1-RBD (aa 267-376) were constructed by cloning the corresponding cdnas into psuper-ha-cdna3 vector via BamHI/BglII and
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy se
 A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
Phospho-AKT Sampler Kit
 Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
m 6 A mrna methylation regulates AKT activity to promote the proliferation and tumorigenicity of endometrial cancer
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
The PI3K/AKT axis. Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia. Introduction
 The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION
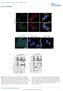 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
William C. Comb, Jessica E. Hutti, Patricia Cogswell, Lewis C. Cantley, and Albert S. Baldwin
 Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
TITLE: A Genetic Approach to Define the Importance of Rheb in Tuberous Sclerosis
 AD Award Number: W81XWH-05-1-0164 TITLE: A Genetic Approach to Define the Importance of Rheb in Tuberous Sclerosis PRINCIPAL INVESTIGATOR: Fuyuhiko Tamanoi, Ph.D. CONTRACTING ORGANIZATION: The University
AD Award Number: W81XWH-05-1-0164 TITLE: A Genetic Approach to Define the Importance of Rheb in Tuberous Sclerosis PRINCIPAL INVESTIGATOR: Fuyuhiko Tamanoi, Ph.D. CONTRACTING ORGANIZATION: The University
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
Growth and Differentiation Phosphorylation Sampler Kit
 Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
HHS Public Access Author manuscript Nat Cell Biol. Author manuscript; available in PMC 2011 September 01.
 Inactivation of Rheb by PRAK-mediated phosphorylation is essential for energy depletion-induced suppression of mtorc1 Min Zheng 1, Yan-Hai Wang 1, Xiao-Nan Wu 1, Su-Qin Wu 1, Bao-Ju Lu 2, Meng-Qiu Dong
Inactivation of Rheb by PRAK-mediated phosphorylation is essential for energy depletion-induced suppression of mtorc1 Min Zheng 1, Yan-Hai Wang 1, Xiao-Nan Wu 1, Su-Qin Wu 1, Bao-Ju Lu 2, Meng-Qiu Dong
Supplemental Data. Prolonged Rapamycin Treatment Inhibits mtorc2 Assembly and Akt/PKB. Supplemental Experimental Procedures
 Supplemental Data Prolonged Rapamycin Treatment Inhibits mtorc2 Assembly and Akt/PKB Dos D. Sarbassov, Siraj M. Ali, Shomit Sengupta, Joon-Ho Sheen, Peggy P. Hsu, Alex F. Bagley, Andrew L. Markhard, and
Supplemental Data Prolonged Rapamycin Treatment Inhibits mtorc2 Assembly and Akt/PKB Dos D. Sarbassov, Siraj M. Ali, Shomit Sengupta, Joon-Ho Sheen, Peggy P. Hsu, Alex F. Bagley, Andrew L. Markhard, and
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
doi:10.1038/nature11700 Figure 1: RIP3 as a potential Sirt2 interacting protein. Transfected Flag-tagged Sirt2 was immunoprecipitated from cells and eluted from the Sepharose beads using Flag peptide.
SUPPLEMENTARY INFORMATION
 Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/10/471/eaah5085/dc1 Supplementary Materials for Phosphorylation of the exocyst protein Exo84 by TBK1 promotes insulin-stimulated GLUT4 trafficking Maeran Uhm,
www.sciencesignaling.org/cgi/content/full/10/471/eaah5085/dc1 Supplementary Materials for Phosphorylation of the exocyst protein Exo84 by TBK1 promotes insulin-stimulated GLUT4 trafficking Maeran Uhm,
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
NIH Public Access Author Manuscript Science. Author manuscript; available in PMC 2013 September 15.
 NIH Public Access Author Manuscript Published in final edited form as: Science. 2013 March 15; 339(6125): 1323 1328. doi:10.1126/science.1228792. Stimulation of de novo pyrimidine synthesis by growth signaling
NIH Public Access Author Manuscript Published in final edited form as: Science. 2013 March 15; 339(6125): 1323 1328. doi:10.1126/science.1228792. Stimulation of de novo pyrimidine synthesis by growth signaling
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
www.sciencesignaling.org/cgi/content/full/6/283/ra57/dc1 Supplementary Materials for JNK3 Couples the Neuronal Stress Response to Inhibition of Secretory Trafficking Guang Yang,* Xun Zhou, Jingyan Zhu,
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Supplementary Material
 Supplementary Material The Androgen Receptor is a negative regulator of eif4e Phosphorylation at S209: Implications for the use of mtor inhibitors in advanced prostate cancer Supplementary Figures Supplemental
Supplementary Material The Androgen Receptor is a negative regulator of eif4e Phosphorylation at S209: Implications for the use of mtor inhibitors in advanced prostate cancer Supplementary Figures Supplemental
RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh-
 1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
Supplemental Information. NRF2 Is a Major Target of ARF. in p53-independent Tumor Suppression
 Molecular Cell, Volume 68 Supplemental Information NRF2 Is a Major Target of ARF in p53-independent Tumor Suppression Delin Chen, Omid Tavana, Bo Chu, Luke Erber, Yue Chen, Richard Baer, and Wei Gu Figure
Molecular Cell, Volume 68 Supplemental Information NRF2 Is a Major Target of ARF in p53-independent Tumor Suppression Delin Chen, Omid Tavana, Bo Chu, Luke Erber, Yue Chen, Richard Baer, and Wei Gu Figure
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Predictive PP1Ca binding region in BIG3 : 1,228 1,232aa (-KAVSF-) HEK293T cells *** *** *** KPL-3C cells - E E2 treatment time (h)
 Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
a b G75 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server.
 a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
S1a S1b S1c. S1d. S1f S1g S1h SUPPLEMENTARY FIGURE 1. - si sc Il17rd Il17ra bp. rig/s IL-17RD (ng) -100 IL-17RD
 SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
A Self-Propelled Biological Process Plk1-Dependent Product- Activated, Feed-Forward Mechanism
 A Self-Propelled Biological Process Plk1-Dependent Product- Activated, Feed-Forward Mechanism The Harvard community has made this article openly available. Please share how this access benefits you. Your
A Self-Propelled Biological Process Plk1-Dependent Product- Activated, Feed-Forward Mechanism The Harvard community has made this article openly available. Please share how this access benefits you. Your
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
In the format provided by the authors and unedited. 2 3 4 DOI: 10.1038/NMAT4893 EGFR and HER2 activate rigidity sensing only on rigid matrices Mayur Saxena 1,*, Shuaimin Liu 2,*, Bo Yang 3, Cynthia Hajal
The IKK-related kinase TBK1 activates mtorc1 directly in response to growth factors and innate immune agonists
 Article The IKK-related kinase TBK1 activates mtorc1 directly in response to growth factors and innate immune agonists Cagri Bodur 1, Dubek Kazyken 1,, Kezhen Huang 1,, Bilgen Ekim Ustunel 1, Kate A Siroky
Article The IKK-related kinase TBK1 activates mtorc1 directly in response to growth factors and innate immune agonists Cagri Bodur 1, Dubek Kazyken 1,, Kezhen Huang 1,, Bilgen Ekim Ustunel 1, Kate A Siroky
Supplementary Figure 1
 Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/430/ra57/dc1 Supplementary Materials for The 4E-BP eif4e axis promotes rapamycinsensitive growth and proliferation in lymphocytes Lomon So, Jongdae Lee, Miguel
www.sciencesignaling.org/cgi/content/full/9/430/ra57/dc1 Supplementary Materials for The 4E-BP eif4e axis promotes rapamycinsensitive growth and proliferation in lymphocytes Lomon So, Jongdae Lee, Miguel
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3311 A B TSC2 -/- MEFs C Rapa Hours WCL 0 6 12 24 36 pakt.s473 AKT ps6k S6K CM IGF-1 Recipient WCL - + - + - + pigf-1r IGF-1R pakt ps6 AKT D 1 st SILAC 2 nd SILAC E GAPDH FGF21 ALKPGVIQILGVK
DOI: 10.1038/ncb3311 A B TSC2 -/- MEFs C Rapa Hours WCL 0 6 12 24 36 pakt.s473 AKT ps6k S6K CM IGF-1 Recipient WCL - + - + - + pigf-1r IGF-1R pakt ps6 AKT D 1 st SILAC 2 nd SILAC E GAPDH FGF21 ALKPGVIQILGVK
Tuberous sclerosis complex (TSC) is an autosomal dominant
 Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase Philippe P. Roux*, Bryan A. Ballif*, Rana Anjum*, Steven P. Gygi*,
Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase Philippe P. Roux*, Bryan A. Ballif*, Rana Anjum*, Steven P. Gygi*,
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as
 Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
(A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939 (5 µm)
 Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Jewell et al., http://www.jcb.org/cgi/content/full/jcb.201007176/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. IR Munc18c association is independent of IRS-1. (A and
Supplemental material Jewell et al., http://www.jcb.org/cgi/content/full/jcb.201007176/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. IR Munc18c association is independent of IRS-1. (A and
Signal Transduction Pathway Smorgasbord
 Molecular Cell Biology Lecture. Oct 28, 2014 Signal Transduction Pathway Smorgasbord Ron Bose, MD PhD Biochemistry and Molecular Cell Biology Programs Washington University School of Medicine Outline 1.
Molecular Cell Biology Lecture. Oct 28, 2014 Signal Transduction Pathway Smorgasbord Ron Bose, MD PhD Biochemistry and Molecular Cell Biology Programs Washington University School of Medicine Outline 1.
Supplementary Materials
 Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
DEPTOR Is an mtor Inhibitor Frequently Overexpressed in Multiple Myeloma Cells and Required for Their Survival
 DEPTOR Is an mtor Inhibitor Frequently Overexpressed in Multiple Myeloma Cells and Required for Their Survival Timothy R. Peterson, 1,2 Mathieu Laplante, 1,2 Carson C. Thoreen, 1,2 Yasemin Sancak, 1,2
DEPTOR Is an mtor Inhibitor Frequently Overexpressed in Multiple Myeloma Cells and Required for Their Survival Timothy R. Peterson, 1,2 Mathieu Laplante, 1,2 Carson C. Thoreen, 1,2 Yasemin Sancak, 1,2
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/7/308/ra4/dc1 Supplementary Materials for Antipsychotics Activate mtorc1-dependent Translation to Enhance Neuronal Morphological Complexity Heather Bowling, Guoan
www.sciencesignaling.org/cgi/content/full/7/308/ra4/dc1 Supplementary Materials for Antipsychotics Activate mtorc1-dependent Translation to Enhance Neuronal Morphological Complexity Heather Bowling, Guoan
mtor plays critical roles in pancreatic cancer stem cells through specific and stemness-related functions
 Supplementary Information mtor plays critical roles in pancreatic cancer stem cells through specific and stemness-related functions Shyuichiro Matsubara 1, Qiang Ding 1, Yumi Miyazaki 1, Taisaku Kuwahata
Supplementary Information mtor plays critical roles in pancreatic cancer stem cells through specific and stemness-related functions Shyuichiro Matsubara 1, Qiang Ding 1, Yumi Miyazaki 1, Taisaku Kuwahata
Biol403 MAP kinase signalling
 Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
SUPPLEMENTARY INFORMATION
 doi:.38/nature234 Note 1. LINC00961 encodes a lysosomal Type I transmembrane polypeptide In order to determine if the polypeptide encoded by LINC00961 was an integral membrane protein or simply associated
doi:.38/nature234 Note 1. LINC00961 encodes a lysosomal Type I transmembrane polypeptide In order to determine if the polypeptide encoded by LINC00961 was an integral membrane protein or simply associated
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Nature Immunology doi: /ni.3268
 Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
mtorc2 phosphorylates protein kinase Cf to regulate its stability and activity
 Scientific Report mtorc2 phosphorylates protein kinase Cf to regulate its stability and activity Xin Li 1 & Tianyan Gao 1,2,* Abstract Protein kinase Cf (PKCf) is phosphorylated at the activation loop
Scientific Report mtorc2 phosphorylates protein kinase Cf to regulate its stability and activity Xin Li 1 & Tianyan Gao 1,2,* Abstract Protein kinase Cf (PKCf) is phosphorylated at the activation loop
Signaling. Dr. Sujata Persad Katz Group Centre for Pharmacy & Health research
 Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Supplementary Figure 1
 Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Tbk1-TKO! DN cells (%)! 15! 10!
 a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
a! T Cells! TKO! B Cells! TKO! b! CD4! 8.9 85.2 3.4 2.88 CD8! Tbk1-TKO! 1.1 84.8 2.51 2.54 c! DN cells (%)! 4 3 2 1 DP cells (%)! 9 8 7 6 CD4 + SP cells (%)! 5 4 3 2 1 5 TKO! TKO! TKO! TKO! 15 1 5 CD8
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature05732 SUPPLEMENTARY INFORMATION Supplemental Data Supplement Figure Legends Figure S1. RIG-I 2CARD undergo robust ubiquitination a, (top) At 48 h posttransfection with a GST, GST-RIG-I-2CARD
doi: 10.1038/nature05732 SUPPLEMENTARY INFORMATION Supplemental Data Supplement Figure Legends Figure S1. RIG-I 2CARD undergo robust ubiquitination a, (top) At 48 h posttransfection with a GST, GST-RIG-I-2CARD
Expanding mtor signaling
 666 REVIEW Cell Research (2007) 17:666-681. 2007 IBCB, SIBS, CAS All rights reserved 1001-0602/07 $ 30.00 www.nature.com/cr Qian Yang 1,2, Kun-Liang Guan 1,2,3 1 Life Sciences Institute; 2 Department of
666 REVIEW Cell Research (2007) 17:666-681. 2007 IBCB, SIBS, CAS All rights reserved 1001-0602/07 $ 30.00 www.nature.com/cr Qian Yang 1,2, Kun-Liang Guan 1,2,3 1 Life Sciences Institute; 2 Department of
TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer
 Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Mighty RapaLink-1 vanquishes undruggable mutant mtor in glioblastoma
 Editorial Mighty RapaLink-1 vanquishes undruggable mutant mtor in glioblastoma Meena Jhanwar-Uniyal Department of Neurosurgery, New York Medical College, Valhalla, New York, USA Correspondence to: Meena
Editorial Mighty RapaLink-1 vanquishes undruggable mutant mtor in glioblastoma Meena Jhanwar-Uniyal Department of Neurosurgery, New York Medical College, Valhalla, New York, USA Correspondence to: Meena
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643
 Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Supplemental Information
 Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway
 AWARD NUMBER: W81XWH-12-1-0560 TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway PRINCIPAL INVESTIGATOR: Andrew S. Kraft, MD CONTRACTING ORGANIZATION:
AWARD NUMBER: W81XWH-12-1-0560 TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway PRINCIPAL INVESTIGATOR: Andrew S. Kraft, MD CONTRACTING ORGANIZATION:
SUPPLEMENTAL FIGURE LEGENDS
 SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
Role of Dual PI3/Akt and mtor Inhibition in Waldenstrom's Macroglobulinemia
 Role of Dual PI3/Akt and mtor Inhibition in Waldenstrom's Macroglobulinemia The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
Role of Dual PI3/Akt and mtor Inhibition in Waldenstrom's Macroglobulinemia The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters.
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Expanded View Figures
 MO reports PR3 dephosphorylates TZ Xian-o Lv et al xpanded View igures igure V1. PR3 dephosphorylates and inactivates YP/TZ., Overexpression of tight junction proteins Pals1 () or LIN7 () has no effect
MO reports PR3 dephosphorylates TZ Xian-o Lv et al xpanded View igures igure V1. PR3 dephosphorylates and inactivates YP/TZ., Overexpression of tight junction proteins Pals1 () or LIN7 () has no effect
injected subcutaneously into flanks of 6-8 week old athymic male nude mice (LNCaP SQ) and body
 SUPPLEMENTAL FIGURE LEGENDS Figure S1: Generation of ENZR Xenografts and Cell Lines: (A) 1x10 6 LNCaP cells in matrigel were injected subcutaneously into flanks of 6-8 week old athymic male nude mice (LNCaP
SUPPLEMENTAL FIGURE LEGENDS Figure S1: Generation of ENZR Xenografts and Cell Lines: (A) 1x10 6 LNCaP cells in matrigel were injected subcutaneously into flanks of 6-8 week old athymic male nude mice (LNCaP
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA. 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT. Acc1 AGCAGATCCGCAGCTTG ACCTCTGCTCGCTGAGTGC
 Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
Receptor mediated Signal Transduction
 Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
supplementary information
 DOI: 1.138/ncb1 Control Atg7 / NAC 1 1 1 1 (mm) Control Atg7 / NAC 1 1 1 1 (mm) Lamin B Gstm1 Figure S1 Neither the translocation of into the nucleus nor the induction of antioxidant proteins in autophagydeficient
DOI: 1.138/ncb1 Control Atg7 / NAC 1 1 1 1 (mm) Control Atg7 / NAC 1 1 1 1 (mm) Lamin B Gstm1 Figure S1 Neither the translocation of into the nucleus nor the induction of antioxidant proteins in autophagydeficient
Singapore. 2 Both authors contributed equally to this work. 3 Present address: Immunology Program, Memorial Sloan-Kettering Cancer
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 16, pp. 14190 14198, April 22, 2011 2011 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. mtor Complex 2 Targets
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 16, pp. 14190 14198, April 22, 2011 2011 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. mtor Complex 2 Targets
Genome of Hepatitis B Virus. VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department
 Genome of Hepatitis B Virus VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department Proto Oncogen and Oncogen Oncogen Proteins that possess the ability to cause
Genome of Hepatitis B Virus VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department Proto Oncogen and Oncogen Oncogen Proteins that possess the ability to cause
Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway
 Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway Hui H. Zhang 1, Jingxiang Huang 1, Katrin Düvel 1, Bernard Boback 1, Shulin Wu 2, Rachel M. Squillace 3, Chin-Lee Wu 2, Brendan D. Manning
Insulin Stimulates Adipogenesis through the Akt-TSC2- mtorc1 Pathway Hui H. Zhang 1, Jingxiang Huang 1, Katrin Düvel 1, Bernard Boback 1, Shulin Wu 2, Rachel M. Squillace 3, Chin-Lee Wu 2, Brendan D. Manning
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Supplementary Figures
 J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
J. Cell Sci. 128: doi:10.1242/jcs.173807: Supplementary Material Supplementary Figures Fig. S1 Fig. S1. Description and/or validation of reagents used. All panels show Drosophila tissues oriented with
The Tumor Suppressor DAP Kinase Is a Target of RSK-Mediated Survival Signaling
 Current Biology, Vol. 15, 1762 1767, October 11, 2005, 2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.08.050 The Tumor Suppressor DAP Kinase Is a Target of RSK-Mediated Survival Signaling
Current Biology, Vol. 15, 1762 1767, October 11, 2005, 2005 Elsevier Ltd All rights reserved. DOI 10.1016/j.cub.2005.08.050 The Tumor Suppressor DAP Kinase Is a Target of RSK-Mediated Survival Signaling
A. Generation and characterization of Ras-expressing autophagycompetent
 Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Supplemental Material Supplemental Figure Legends Fig. S1 A. Generation and characterization of Ras-expressing autophagycompetent and -deficient cell lines. HA-tagged H-ras V12 was stably expressed in
Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death
 www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death Supplementary
www.impactjournals.com/oncotarget/ Oncotarget, Supplementary Materials 2016 Tumor suppressor Spred2 interaction with LC3 promotes autophagosome maturation and induces autophagy-dependent cell death Supplementary
