Myeloid cell-specific ABCA1 deletion does not worsen insulin resistance in HF dietinduced or genetically obese mouse models
|
|
|
- Percival Copeland
- 5 years ago
- Views:
Transcription
1 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2013 Myeloid cell-specific ABCA1 deletion does not worsen insulin resistance in HF dietinduced or genetically obese mouse models Xuewei Zhu Department of Pathology/Lipid Sciences and Wake Forest School of Medicine, Winston-Salem, NC Soonkyu Chung University of Nebraska-Lincoln, schung4@unl.edu Xin Bi Department of Pathology/Lipid Sciences and Wake Forest School of Medicine, Winston-Salem, NC Chia-Chi Chuang Department of Pathology/Lipid Sciences and Wake Forest School of Medicine, Winston-Salem, NC Amanda L. Brown Department of Pathology/Lipid Sciences and Wake Forest School of Medicine, Winston-Salem, NC; See next page for additional authors Follow this and additional works at: Zhu, Xuewei; Chung, Soonkyu; Bi, Xin; Chuang, Chia-Chi; Brown, Amanda L.; Liu, Mingxia; Seo, Jeongmin; Cuffe, Helen; Gebre, Abraham K.; Boudyguina, Elena; and Parks, John S., "Myeloid cell-specific ABCA1 deletion does not worsen insulin resistance in HF dietinduced or genetically obese mouse models" (2013). Nutrition and Health Sciences -- Faculty Publications This Article is brought to you for free and open access by the Nutrition and Health Sciences, Department of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Nutrition and Health Sciences -- Faculty Publications by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln.
2 Authors Xuewei Zhu, Soonkyu Chung, Xin Bi, Chia-Chi Chuang, Amanda L. Brown, Mingxia Liu, Jeongmin Seo, Helen Cuffe, Abraham K. Gebre, Elena Boudyguina, and John S. Parks This article is available at of Nebraska - Lincoln:
3 J Lipid Res Oct; 54(10): doi: /jlr.M PMCID: PMC Received 2013 Apr 11; Revised 2013 Jul 23 Copyright 2013 by the American Society for Biochemistry and Molecular Biology, Inc. Myeloid cell-specific ABCA1 deletion does not worsen insulin resistance in HF dietinduced or genetically obese mouse models Xuewei Zhu, * Soonkyu Chung, * Xin Bi, * Chia-Chi Chuang, * Amanda L. Brown, * Mingxia Liu, * Jeongmin Seo, * Helen Cuffe, * Abraham K. Gebre, * Elena Boudyguina, * and John S. Parks 1,* * Department of Pathology/Lipid Sciences and Wake Forest School of Medicine, Winston-Salem, NC; Department of Biochemistry, Wake Forest School of Medicine, Winston-Salem, NC; and Department of Food Science and Human Nutrition, University of Florida, Gainesville, FL 1 To whom correspondence should be addressed. ude.htlaehekaw@skrapj Abstract Obesity-associated low-grade chronic inflammation plays an important role in the development of insulin resistance. The membrane lipid transporter ATP-binding cassette transporter A1 (ABCA1) promotes formation of nascent HDL particles. ABCA1 also dampens macrophage inflammation by reducing cellular membrane cholesterol and lipid raft content. We tested the hypothesis that myeloid-specific ABCA1 deletion may exacerbate insulin resistance by increasing the obesity-associated chronic low-grade inflammation. Myeloid cell-specific ABCA1 knockout (MSKO) and wild-type (WT) mice developed obesity, insulin resistance, mild hypercholesterolemia, and hepatic steatosis to a similar extent with a 45% high-fat (HF) diet feeding or after crossing into the ob/ob background. Resident peritoneal macrophages and stromal vascular cells from obese MSKO mice accumulated significantly more cholesterol. Relative to chow, HF diet markedly induced macrophage infiltration and inflammatory cytokine
4 expression to a similar extent in adipose tissue of WT and MSKO mice. Among proinflammatory cytokines examined, only IL-6 was highly upregulated in MSKO-ob/ob versus ob/ob mouse peritoneal macrophages, indicating a nonsignificant effect of myeloid ABCA1 deficiency on obesity-associated chronic inflammation. In conclusion, myeloid-specific ABCA1 deficiency does not exacerbate obesity-associated low-grade chronic inflammation and has minimal impact on the pathogenesis of insulin resistance in both HF diet-induced and genetically obese mouse models. Keywords: cholesterol, macrophage, inflammation, obesity, high-fat In the past decade, obesity-associated chronic low-grade inflammation has been recognized as a major cause of insulin resistance and type 2 diabetes. For example, overexpansion of adipose tissues increases the production of pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-α) (1), and chemokines, such as monocyte chemoattractant protein-1 (MCP-1) (2). TNF-αactivated c-jun N-terminal kinase (JNK) impairs insulin signaling, which contributes to insulin resistance (3). MCP-1 recruits immune cells, especially macrophages (40% of infiltrated immune cells), to adipose tissue, exacerbating inflammation and insulin resistance (4). Interestingly, Tolllike receptor 4 (TLR4), a pathogen-associated molecular pattern receptor, has been implicated as a link between inflammation and insulin resistance by functioning as a receptor for saturated fatty acids (5 7). Genetic deletion of key components of the TLR4 signaling pathway, such as TLR4 (8), JNK1 (9), or inhibitor of NF-κB-kinase β (IKKβ) (10) in hematopoietic or myeloid cells further established the mechanistic link between macrophage inflammation and insulin resistance. ATP-binding cassette transporter A1 (ABCA1), a member of a large family of membrane transporters, effluxes free cholesterol (FC) and phospholipids across the plasma membrane to combine with apolipoprotein A-I, forming nascent high density lipoprotein (HDL) particles (11, 12). Mutations that inactivate ABCA1 lead to Tangier disease, a disorder characterized by near absence of HDL, elevated plasma triglycerides (TGs), reduced low density lipoprotein concentrations, and increased storage of cholesteryl esters (CEs) in macrophages (13 15). Insulin resistance has not been documented in people with Tangier disease, perhaps because the disease is so rare (16). However, ABCA1 single nucleotide polymorphisms are associated with altered glucose metabolism and insulin resistance in humans (17 20). Of interest, leukocyte ABCA1 gene expression is associated with fasting glucose concentrations in normoglycemic men, and reduced leukocyte ABCA1 gene expression is associated with type 2 diabetes (21, 22). To date, only one study has directly linked ABCA1 expression to hyperglycemia, reporting that pancreatic β cellspecific ABCA1 deletion results in defective insulin release from islet cells (23). Whether deletion of ABCA1 expression in other cell types, such as macrophages, alters glucose metabolism is unknown. Using myeloid cell (macrophage and neutrophil)-specific ABCA1 knockout (MSKO) mice, we demonstrated that macrophages from MSKO mice have a significant increase in FC and are more responsive to pro-inflammatory stimuli [e.g., lipopolysaccharide (LPS)] in vivo and in vitro compared with wild-type (WT) mice (24). This response was mediated through the TLR and myeloid differentiation primary-response protein 88 (MyD88)-dependent pathway and was
5 independent of alterations in plasma lipid concentrations (24). Hypersensitivity to LPS is most likely due to increased lipid raft content and increased trafficking of TLR4 into plasma membrane lipid rafts in MSKO mouse macrophages (25). Furthermore, MSKO mice challenged with the Listeria monocytogenes cleared the bacterium better than WT mice (26). These studies clearly demonstrated a regulatory role of myeloid ABCA1 in macrophage inflammation and innate immunity. Based on the documented relationship between macrophage inflammation and insulin resistance, we hypothesized that myeloid-specific ABCA1 deficiency may exacerbate obesity-induced chronic inflammation and insulin resistance in mice fed a HF diet or crossed into the leptindeficient ob/ob background. However, MSKO and WT mice developed obesity, adipose inflammation, and insulin resistance to a similar extent, suggesting that myeloid ABCA1 expression does not significantly worsen obesity-induced chronic inflammation and insulin resistance. METHODS Animals WT (ABCA1 +/+ ) and MSKO (ABCA1 M/ M ) mice were generated as described previously (24, 25). Mice were backcrossed into the C57BL/6 background for six generations before use in these studies and housed in a specific pathogen-free facility with a 12 h light/dark cycle. Experiments were conducted in conformity with the Public Health Service Policy on Humane Care and Use of Laboratory Animals, and the experimental protocol was approved by the Wake Forest University Animal Care and Use Committee. At 8 weeks of age, male mice were switched from chow to a HF diet containing 45% of energy as lard and 0.015% cholesterol for weeks. The HF diet was made by our institutional diet kitchen (see detailed diet composition in supplementary Table I). Body weight (BW) was measured biweekly. To generate MSKO-ob/ob mice, MSKO mice were first bred with heterozygous ob/ob (ob/+) mice. The resulting double heterozygous MSKO-ob/+ mice were intercrossed to generate ob/+ or MSKO-ob/+ mice, which were then intercrossed to generate ob/ob or MSKO-ob/ob mice, respectively. Ob/ob and MSKO-ob/ob male mice were fed chow for weeks for the described studies. Cell culture Peritoneal macrophages were harvested from mice by flushing the peritoneal cavity with cold PBS. The peritoneal cells were plated in RPMI media containing 100 U/ml penicillin, 100 μg/ml streptomycin, and 1% Nutridoma SP media (Roche Applied Science). After a 2 h incubation, floating cells were removed by washing with PBS and adherent macrophages were used for experiments. Plasma lipid, glucose, and insulin analysis Blood samples were taken from tail veins following a 4 h fast during the light cycle (chow phase) before, and at 2 4 week intervals after initiation of HF diet feeding. Plasma cholesterol (Wako)
6 and TGs (Roche) were determined by enzymatic analysis according to the manufacturer's instructions. Plasma glucose levels were measured using a glucometer (Ascensia Contour, Bayer). Plasma insulin levels were measured by ELISA (Crystal Chem, Inc., Downers Grove, IL). For fed and overnight fasting plasma samples, blood was collected by tail vein at 9:00 AM in ad libitum fed mice (fed) or in mice fasted during the dark cycle for 15 h (5:00 PM 8:00 AM). Hepatic lipid analysis Liver lipids were extracted with chloroform:methanol (2:1) and the extract was used for enzymatic assays (cholesterol, Wako; TG, Roche). Data were normalized to liver protein mass, measured by the Lowry protein assay. Glucose homeostasis analysis Glucose tolerance tests (GTTs) and insulin tolerance tests (ITTs) were done after weeks of HF diet consumption or with ob/ob mice at weeks of age. Briefly, for GTTs, mice were fasted overnight before intraperitoneal (ip) injection of 1 g glucose/kg BW. Blood was collected before and after injection (0, 15, 30, 60, and 120 min) to measure glucose concentrations using a commercial glucose monitor. One week later, the same groups of mice were used for ip injection of 1.5 U of regular human insulin/kg BW (HF diet-fed mice) or 3 U/kg BW (ob/ob mice) after a 5 h fast. Blood glucose concentrations were measured at 0, 15, 30, 60, and 120 min after injection. Real-time PCR Total RNA in peritoneal macrophages and white adipose tissue was extracted using Trizol (Invitrogen) and RNeasy lipid tissue kits (Qiagen), respectively, according to the manufacturer's protocols. cdna preparation and real-time PCR were conducted as described previously (24). Immunohistochemistry staining Epididymal fat tissue sections were incubated with the primary antibodies to CD68 (abd Serotec) or cleaved caspase-3 (Cell Signaling), followed by the biotinylated secondary antibody. The staining was visualized using ABC reagent (ABC vector kit; Vector) and DAB substrate chromogen (Dako). The area of fat sections covered by CD68 + or cleaved caspase-3 + cells was measured using Image-Pro software to quantify the percentage of positive staining cells in adipose tissue. Stromal vascular cell isolation Stromal vascular cells were fractioned according to the procedure described by Brown et al. (27). Briefly, epididymal fat was minced and enzymatically digested for 45 min with 5 ml/g tissue of collagenase type I (1 mg/ml, Worthington Biochemical Corporation) media. The digestion mixture was filtered through a 100 μm cell strainer and stromal vascular cells were pelleted by centrifugation (3,000 rpm for 10 min). The stromal vascular fraction (SVF) was then used for flow cytometry.
7 Flow cytometry After blocking the Fcγ receptor with purified anti-mouse CD16/CD32 antibody (Fcγ receptor III/II; BD Biosciences), SVF cells were incubated at 4 C for 30 min with isotype controls or the following Abs: PE-anti-F4/80 (BM8; BD Pharmingen), PE-Cy7-anti-CD11c (HL3; BD Pharmingen), and Alexa Fluor 647-anti-CD206 (MR5D3; AbD Serotec). Cell fluorescence was measured using a FACSCanto II flow cytometer and data analyzed with FlowJo software. Cholesterol content of macrophages and stromal vascular cells Macrophages or stromal vascular cells isolated from adipose tissue were extracted with isopropanol (including 5α-cholestane as internal standard) at room temperature overnight and analyzed for cholesterol content by gas-liquid chromatography (27). Statistical analysis Data are presented as the mean ± SEM unless indicated otherwise. Differences were compared with two-tailed Student's t-test or one-way ANOVA using GraphPad Prism software. P < 0.05 was considered statistically significant. RESULTS Macrophages from MSKO versus WT obese mice had significantly more cholesterol accumulation, but comparable inflammatory cytokine expression MSKO macrophages have increased plasma membrane FC and lipid rafts, resulting in increased response to TLR4 stimulation (LPS) compared with WT cells (24). Saturated fatty acids also activate TLR4 and induce chronic inflammation in the setting of obesity (5 7). We hypothesized that increased dietary saturated fatty acids (i.e., HF diet), functioning as TLR4 agonists, might increase inflammation during diet-induced obesity, worsening development of insulin resistance in MSKO versus WT mice. To test this possibility, we first measured cholesterol content in the resident peritoneal macrophages from obese mice. Consistent with our previous findings, macrophages from HF diet-fed MSKO mice (Fig. 1A, C) or chow-fed MSKO-ob/ob mice (Fig. 1B, D) had significantly more FC and CE accumulation compared with their WT counterparts. However, similar inflammatory cytokine gene expression was observed for macrophages from 24 week HF diet-fed WT and MSKO mice (Fig. 1E). Among the examined cytokines, only IL-6 was upregulated in macrophages from chow-fed MSKO-ob/ob versus ob/ob mice (Fig. 1F). In addition, the expression of TLR4 and its lipid-binding coreceptor CD36 (28) did not differ between genotypes (Fig. 1G, H). These data suggest that despite significant increases in macrophage cholesterol accumulation resulting from ABCA1 deficiency, HF diet feeding was not sufficient to stimulate pro-inflammatory activation of macrophages in vivo. Myeloid-specific ABCA1 deficiency does not worsen insulin resistance in HF dietfed mice or chow-fed ob/ob mice
8 Obesity-associated low-grade chronic inflammation underlies the pathogenesis of type 2 diabetes and insulin resistance (29, 30). Macrophage ABCA1 dampens pro-inflammation via downregulating MyD88-dependent TLR signaling (24, 25). We hypothesized that myeloidspecific ABCA1 deficiency may exacerbate chronic inflammation and insulin resistance associated with diet-induced or genetically obese mice. To test this hypothesis, we first fed the WT and MSKO male mice a HF diet containing 45% of energy as lard and 0.015% cholesterol. When challenged with the HF diet, WT and MSKO mice gained similar BW over the 24 week diet feeding period (Fig. 2A). Liver and fat mass were comparable between genotypes after 24 weeks of diet consumption (supplementary Fig. IA, B). We then assessed several key parameters of glucose homeostasis in WT and MSO mice. Blood glucose concentrations after a 4 h fast were comparable between both groups of mice (Fig. 2B). MSKO mice also exhibited similar plasma glucose clearance (GTT) compared with WT mice (Fig. 2C). Consistent with this finding, the ability of insulin to lower blood glucose concentrations during ITTs was equivalent in both groups of mice (Fig. 2D). We next measured plasma insulin concentrations in overnight-fasted and fed mice; again, no differences were observed between genotypes (Fig. 2E, F). Comparable glucose homeostasis between the two genotypes also occurred with prolonged diet feeding (e.g., weeks) using different sets of mice. Adding cholesterol (0.2%) to the HF diet did not significantly alter the phenotype (data not shown). Similar outcomes were also observed for HF diet-fed female mice (data not shown). Together, our data suggest that myeloid cell-specific ABCA1 deletion does not affect the development of systemic insulin resistance in the presence of HF diet-induced obesity. Next, we tested our hypothesis using an early-onset genetic obesity mouse model by crossing MSKO mice into the ob/ob background. At 8 and 14 weeks of age, ob/ob and MSKO-ob/ob mice had comparable BWs (Fig. 3A, B). No differences in liver or fat mass were observed between genotypes (supplementary Fig. IC, D). A GTT was performed when mice were 13 weeks old, followed by an ITT the next week. Due to the low yield of the MSKO-ob/ob mice, we combined the data from three sets of mice by normalizing data to baseline values. Similar to the HF diet study, we saw no differences in the GTTs and ITTs for ob/ob and MSKO-ob/ob mice (Fig. 3C, D), indicating comparable systemic insulin resistance. Similar plasma insulin concentrations (Fig. 3E, F) further confirmed that insulin resistance did not differ between genotypes. Myeloid-specific ABCA1 deficiency does not worsen adipose tissue inflammation We next examined the inflammatory status in fat, an important insulin target tissue. We first measured expression of F4/80 and CD68 (macrophage markers) in epididymal fat from HF dietfed mice (17 weeks). Compared with chow, the HF diet significantly increased F4/80 and CD68 expression in adipose tissue, indicating macrophage infiltration into fat tissue (Fig. 3A). Unexpectedly, MSKO versus WT fat had significantly lower F4/80 expression and a trend toward less CD68 expression. Furthermore, immunohistochemistry staining of macrophages using CD68 antibody revealed that CD68 + cells in MSKO adipose tissues were greatly reduced compared with WT (Fig. 4B), indicating significantly less macrophage infiltration into fat. The comparable level of cleaved caspase-3 in adipose tissues (supplementary Fig. IIA, B) rules out a major role for apoptosis in the differential accumulation of macrophages between genotypes. Interestingly, this
9 difference was not observed with a longer period of HF diet feeding (24 weeks, supplementary Fig. IIIA). Obesity induces a phenotypic switch from M2-polarized state (alternatively activated) to an M1 (classically activated) pro-inflammatory state in adipose macrophages (31). We examined expression of the M1 type of inflammatory cytokine/chemokine and M2 macrophage markers in fat. Feeding the HF diet for 17 weeks significantly induced pro-inflammatory cytokine (TNF-α) or chemokine (MCP-1) expression in adipose tissues compared with chow (Fig. 4C). However, no genotypic differences were observed between the chow-fed or HF diet-fed mice (Fig. 4C). Not surprisingly, the expression of M2 macrophage markers did not show any difference between genotypes (Fig. 4D). Again, no significant difference was observed in TLR4 or CD36 expression between genotypes (supplementary Fig. IIIB). Increasing the duration of HF diet feeding from 17 to 24 weeks did not significantly increase adipose inflammation in MSKO versus WT mice (supplementary Fig. IIIC). Next, we examined the macrophage infiltration and inflammatory cytokine expression in ob/ob mouse adipose tissue. Flow cytometry revealed that chow-fed MSKO-ob/ob versus ob/ob mice had significantly less (F4/80 + ) and M1 (F4/80 + CD11C + ), but similar M2 (F4/80 + CD206 + ) macrophage infiltration into fat (Fig. 5A). Furthermore, MSKO-ob/ob versus ob/ob mice had similar levels of IL-1β, TNF-α, and MCP-1 mrna expression and a trend toward decrease in IL- 6 and IL-12p40 mrna expression in fat tissue (Fig. 5B). Note that similar to peritoneal macrophages, stromal vascular cells from MSKO-ob/ob fat had significantly higher FC (23% increase) and CE (254% increase) accumulation relative to their WT counterparts (Fig. 5C). Thus, our data suggest that myeloid cell-specific ABCA1 deletion enhances cholesterol accumulation in adipose macrophages; however, it does not enhance obesity-induced macrophage infiltration or chronic inflammation in adipose tissue. Myeloid cell-specific ABCA1 deficiency does not alter plasma and liver lipid profiles in HF diet-induced or genetically-induced obese mice To determine whether ABCA1 deficiency in myeloid cells affects lipid homeostasis in HF dietfed or chow-fed ob/ob obese mice, we examined the plasma and liver lipid concentrations. There was no genotypic difference in plasma total cholesterol (TC) and TG concentrations during the 24 weeks of HF diet feeding (Fig. 6A, C) or after mice were crossed into the ob/ob background (Fig. 6B, D). We also saw comparable levels of hepatic TC (Fig. 6E, F), FC, and CE (data not shown) accumulation between HF diet-fed WT and MSKO mice. WT and MSKO mice developed hepatic steatosis to a similar extent when fed a HF diet for 24 weeks (Fig. 6G) or crossed into the ob/ob background (Fig. 6H), respectively. Together, these data suggest that myeloid cell-specific ABCA1 deletion does not significantly affect plasma and liver lipid metabolism in the setting of obesity-induced insulin resistance. DISCUSSION Obesity and insulin resistance are chronic low-grade inflammatory diseases associated with macrophage accumulation and activation in adipose tissue (29, 30). We previously demonstrated that myeloid cell-specific deletion of ABCA1 results in increased macrophage inflammatory
10 response to TLR agonists (24), and human studies suggest an association between ABCA1 single nucleotide polymorphisms, leukocyte ABCA1 expression, and insulin resistance (17 22). However, whether there is a link between macrophage ABCA1-regulated innate immunity and insulin resistance is unknown. In the present study, HF diet-fed MSKO mice and chow-fed MSKOob/ob mice did not exhibit exacerbated obesity-associated chronic inflammation or insulin resistance compared with their WT counterparts, despite a significant increase in cholesterol content in both resident peritoneal macrophages and adipose stromal vascular cells that accompanies ABCA1 deletion. Our results suggest that increased dietary saturated fatty acid intake (i.e., HF diet feeding) is not sufficient to induce an augmented inflammatory response and insulin resistance in MSKO mice compared with their WT counterparts. Macrophages play a central role in HF diet-induced obesity and subsequent development of systemic insulin resistance through secretion of pro-inflammatory cytokines that act on local tissues or circulate to distal tissues. The inflammatory cytokines then block insulin receptor signaling by serine phosphorylation of insulin-like substrate, interfering with its ability to engage in insulin receptor signaling (32). During development of diet-induced obesity, saturated fatty acids can activate TLR4, triggering a low-grade inflammatory state in macrophages and other cell types, such as adipose tissue and muscle (7, 8). Deletion of TLR4 in bone marrow-derived cells blunts the macrophage and adipose tissue response to dietary fatty acids and protects mice from HF diet-induced insulin resistance (8). Deletion of hematopoietic cell JNK1 (a key component of the TLR4 signaling pathway) attenuates inflammation in metabolic tissues and improves insulin sensitivity in HF diet-fed mice (9). Moreover, myeloid-specific IKKβ deficiency improves systemic insulin sensitivity in mice on a HF diet (10), suggesting a causative link between increased macrophage inflammation and insulin resistance. Our previous studies reported that myeloid-specific ABCA1 deficiency exacerbates macrophage inflammatory response to a moderate LPS dose in vivo (3 mg/kg) and in vitro (100 ng/ml), and this response was mediated through a MyD88-dependent TLR4 pathway (24). Unstimulated resting macrophages were indistinguishable between WT and MSKO mice, except for a small ( 10%) but significant increase in cellular FC. Furthermore, MSKO mice cleared infection with L. monocytogenes more efficiently than WT mice (26). Collectively, these studies clearly establish a regulatory role for myeloid ABCA1 in acute macrophage inflammation and innate immunity. In addition, leukocyte ABCA1 expression protects against atherosclerosis, a chronic disease with well-characterized inflammatory components (33 35). Based on the causative association between macrophage inflammation and insulin resistance, we hypothesized that myeloid-specific ABCA1 deficiency would exacerbate obesity-associated chronic inflammation, worsening insulin resistance. However, MSKO and WT mice developed obesity, insulin resistance, and mild hepatic steatosis to a similar extent with HF diet feeding. Relative to chow, HF diet feeding markedly induced macrophage infiltration into adipose tissue and inflammatory cytokine expression. However, we did not observe enhanced macrophage infiltration and adipose inflammation in MSKO versus WT mice after a HF diet feeding. Similar data were obtained with chow-fed MSKO mice crossed into the ob/ob background. Consistent with our previous findings, macrophages from HF diet-fed MSKO versus WT mice accumulated significantly more cholesterol, but cytokine and chemokine gene expression was similar to WT macrophages. Therefore, myeloid-specific ABCA1 deficiency impairs cholesterol efflux, leading to intracellular cholesterol accumulation, but does
11 not exacerbate obesity-associated low-grade chronic inflammation and does not lead to insulin resistance. Our study shows that absence of myeloid cell ABCA1 expression is not sufficient to mount an exaggerated inflammatory response in the setting of obesity and does not result in insulin resistance. In our previous study, an increased macrophage inflammatory response in ABCA1- deficient versus WT macrophages was triggered by a moderate dose of LPS in vivo or in vitro, and not observed in nonstimulated macrophages (24). There may be several explanations for the lack of an exaggerated inflammatory response with HF diet feeding in our current study. First, the amount of saturated fatty acid necessary to differentially activate an inflammatory response in macrophages with and without ABCA1 expression may have been insufficient. Inflammatory stimulation of macrophages via TLR4 requires high concentrations of saturated fatty acids ( 500 μm) in vitro or intravenous infusion of high amounts of TGs ( 17 μmol/h) in vivo (7). Second, previous studies demonstrating TLR4-specific activation of inflammation by HF diets used mice lacking TLR4 globally (7) or in bone marrow cells (8), leaving open the possibility that cells other than macrophages could have contributed to the exacerbated inflammation and systemic insulin resistance. In fact, to our knowledge, there are only two examples in which myeloid-specific deletion of genes (IKK-β or JNK) involved in canonical inflammatory signaling pathways resulted in HF-induced systemic insulin resistance (10, 36). However, IKK-β and JNK are directly involved in inflammatory signaling, whereas ABCA1 indirectly affects TLR signaling by increasing membrane lipid raft content, resulting in more TLR4 in lipid rafts (25). In addition, the concept that saturated fatty acids activate TLR4 is not universally accepted. For example, functional TLR4 was necessary to attenuate trans fatty acid-induced obesity, hyperglycemia, and hyperinsulinemia (37). Similarly, mice lacking MyD88, a TLR4 signaling adaptor protein, were more susceptible to diet-induced insulin resistance, suggesting a protective role for MyD88 signaling in obesityassociated insulin resistance (38). Finally, in vitro studies suggest that TLR4 activation by fatty acids can potentially be confounded by LPS and lipopeptide contamination (39). In summary, myeloid-specific ABCA1 deficiency impairs cholesterol efflux, but does not significantly exacerbate macrophage infiltration and low-grade inflammation in adipose tissue, or insulin resistance in both HF diet-induced and genetically obese mouse models. However, because we examined advanced obesity resulting from 17 weeks of HF diet feeding, we cannot rule out the possibility that myeloid-specific ABCA1 deficiency might worsen adipose inflammation or metabolic abnormalities in the context of less advanced obesity. Acknowledgments The authors gratefully acknowledge Karen Klein (Translational Science Institute, Wake Forest University Health Sciences) for editing the manuscript. This study was supported by National Institutes of Health Grants HL-49373, HL (J.S.P.), and by American Heart Association Fellowship 09POST (X.Z.).
12 Abbreviations: BW body weight CE cholesteryl ester FC free cholesterol GTT glucose tolerance test HF high-fat IKKβ inhibitor of NF-κB-kinase β ITT insulin tolerance test JNK c-jun N-terminal kinase LPS lipopolysaccharide MCP-1 monocyte chemoattractant protein-1 MSKO myeloid cell-specific ABCA1 knockout MyD88 myeloid differentiation primary-response protein 88 SVF stromal vascular fraction TG triglyceride TLR4 Toll-like receptor 4 TNF-α tumor necrosis factor-α WT wild-type REFERENCES 1. Hotamisligil G. S., Arner P., Caro J. F., Atkinson R. L., Spiegelman B. M Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Invest. 95: [PMCID: PMC295872] [PubMed: ] 2. Kanda H., Tateya S., Tamori Y., Kotani K., Hiasa K., Kitazawa R., Kitazawa S., Miyachi H., Maeda S., Egashira K., et al MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 116: [PMCID: PMC ] [PubMed: ] 3. Hotamisligil G. S., Peraldi P., Budavari A., Ellis R., White M. F., Spiegelman B. M IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNF-alpha- and obesity-induced insulin resistance. Science. 271: [PubMed: ] 4. Wellen K. E., Hotamisligil G. S Obesity-induced inflammatory changes in adipose tissue. J. Clin. Invest. 112: [PMCID: PMC297006] [PubMed: ] 5. Hwang D Modulation of the expression of cyclooxygenase-2 by fatty acids mediated through toll-like receptor 4-derived signaling pathways. FASEB J. 15: [PubMed: ] 6. Lee J. Y., Sohn K. H., Rhee S. H., Hwang D Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J. Biol. Chem. 276: [PubMed: ]
13 7. Shi H., Kokoeva M. V., Inouye K., Tzameli I., Yin H., Flier J. S TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Invest. 116: [PMCID: PMC ] [PubMed: ] 8. Saberi M., Woods N. B., de Luca C., Schenk S., Lu J. C., Bandyopadhyay G., Verma I. M., Olefsky J. M Hematopoietic cell-specific deletion of toll-like receptor 4 ameliorates hepatic and adipose tissue insulin resistance in high-fat-fed mice. Cell Metab. 10: [PMCID: PMC ] [PubMed: ] 9. Solinas G., Vilcu C., Neels J. G., Bandyopadhyay G. K., Luo J. L., Naugler W., Grivennikov S., Wynshaw-Boris A., Scadeng M., Olefsky J. M., et al JNK1 in hematopoietically derived cells contributes to diet-induced inflammation and insulin resistance without affecting obesity. Cell Metab. 6: [PubMed: ] 10. Arkan M. C., Hevener A. L., Greten F. R., Maeda S., Li Z. W., Long J. M., Wynshaw-Boris A., Poli G., Olefsky J., Karin M IKK-beta links inflammation to obesity-induced insulin resistance. Nat. Med. 11: [PubMed: ] 11. Attie A. D., Kastelein J. P., Hayden M. R Pivotal role of ABCA1 in reverse cholesterol transport influencing HDL levels and susceptibility to atherosclerosis. J. Lipid Res. 42: [PubMed: ] 12. Oram J. F., Lawn R. M ABCA1. The gatekeeper for eliminating excess tissue cholesterol. J. Lipid Res. 42: [PubMed: ] 13. Bodzioch M., Orso E., Klucken J., Langmann T., Bottcher A., Diederich W., Drobnik W., Barlage S., Buchler C., Porsch-Ozcurumez M., et al The gene encoding ATP-binding cassette transporter 1 is mutated in Tangier disease. Nat. Genet. 22: [PubMed: ] 14. Brooks-Wilson A., Marcil M., Clee S. M., Zhang L. H., Roomp K., van Dam M., Yu L., Brewer C., Collins J. A., Molhuizen H. O., et al Mutations in ABC1 in Tangier disease and familial high-density lipoprotein deficiency. Nat. Genet. 22: [PubMed: ] 15. Rust S., Rosier M., Funke H., Real J., Amoura Z., Piette J. C., Deleuze J. F., Brewer H. B., Duverger N., Denefle P., et al Tangier disease is caused by mutations in the gene encoding ATP-binding cassette transporter 1. Nat. Genet. 22: [PubMed: ] 16. Serfaty-Lacrosniere C., Civeira F., Lanzberg A., Isaia P., Berg J., Janus E. D., Smith M. P., Jr, Pritchard P. H., Frohlich J., Lees R. S Homozygous Tangier disease and cardiovascular disease. Atherosclerosis. 107: [PubMed: ] 17. Daimon M., Kido T., Baba M., Oizumi T., Jimbu Y., Kameda W., Yamaguchi H., Ohnuma H., Tominaga M., Muramatsu M., et al Association of the ABCA1 gene polymorphisms with type 2 DM in a Japanese population. Biochem. Biophys. Res. Commun. 329: [PubMed: ] 18. Porchay I., Pean F., Bellili N., Royer B., Cogneau J., Chesnier M. C., Caradec A., Tichet J., Balkau B., Marre M., et al ABCA1 single nucleotide polymorphisms on high-density lipoprotein-cholesterol and overweight: the D.E.S.I.R. study. Obesity (Silver Spring). 14: [PubMed: ] 19. Villarreal-Molina M. T., Aguilar-Salinas C. A., Rodriguez-Cruz M., Riano D., Villalobos- Comparan M., Coral-Vazquez R., Menjivar M., Yescas-Gomez P., Konigsoerg-Fainstein M., Romero-Hidalgo S., et al The ATP-binding cassette transporter A1 R230C variant
14 affects HDL cholesterol levels and BMI in the Mexican population: association with obesity and obesity-related comorbidities. Diabetes. 56: [PubMed: ] 20. Villarreal-Molina M. T., Flores-Dorantes M. T., Arellano-Campos O., Villalobos-Comparan M., Rodriguez-Cruz M., Miliar-Garcia A., Huertas-Vazquez A., Menjivar M., Romero- Hidalgo S., Wacher N. H., et al Association of the ATP-binding cassette transporter A1 R230C variant with early-onset type 2 diabetes in a Mexican population. Diabetes. 57: [PubMed: ] 21. Patel D. C., Albrecht C., Pavitt D., Paul V., Pourreyron C., Newman S. P., Godsland I. F., Valabhji J., Johnston D. G Type 2 diabetes is associated with reduced ATP-binding cassette transporter A1 gene expression, protein and function. PLoS ONE. 6: e [PMCID: PMC ] [PubMed: ] 22. Albrecht C., Simon-Vermot I., Elliott J. I., Higgins C. F., Johnston D. G., Valabhji J Leukocyte ABCA1 gene expression is associated with fasting glucose concentration in normoglycemic men. Metabolism. 53: [PubMed: ] 23. Brunham L. R., Kruit J. K., Pape T. D., Timmins J. M., Reuwer A. Q., Vasanji Z., Marsh B. J., Rodrigues B., Johnson J. D., Parks J. S., et al Beta-cell ABCA1 influences insulin secretion, glucose homeostasis and response to thiazolidinedione treatment. Nat. Med. 13: [PubMed: ] 24. Zhu X., Lee J. Y., Timmins J. M., Brown J. M., Boudyguina E., Mulya A., Gebre A. K., Willingham M. C., Hiltbold E. M., Mishra N., et al Increased cellular free cholesterol in macrophage-specific Abca1 knock-out mice enhances pro-inflammatory response of macrophages. J. Biol. Chem. 283: [PMCID: PMC ] [PubMed: ] 25. Zhu X., Owen J. S., Wilson M. D., Li H., Griffiths G. L., Thomas M. J., Hiltbold E. M., Fessler M. B., Parks J. S Macrophage ABCA1 reduces MyD88-dependent Toll-like receptor trafficking to lipid rafts by reduction of lipid raft cholesterol. J. Lipid Res. 51: [PMCID: PMC ] [PubMed: ] 26. Zhu X., Westcott M. M., Bi X., Liu M., Gowdy K. M., Seo J., Cao Q., Gebre A. K., Fessler M. B., Hiltbold E. M., et al Myeloid cell-specific ABCA1 deletion protects mice from bacterial infection. Circ. Res. 111: [PMCID: PMC ] [PubMed: ] 27. Brown J. M., Halvorsen Y. D., Lea-Currie Y. R., Geigerman C., McIntosh M Trans- 10, cis-12, but not cis-9, trans-11, conjugated linoleic acid attenuates lipogenesis in primary cultures of stromal vascular cells from human adipose tissue. J. Nutr. 131: [PubMed: ] 28. Stewart C. R., Stuart L. M., Wilkinson K., van Gils J. M., Deng J., Halle A., Rayner K. J., Boyer L., Zhong R., Frazier W. A., et al CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 11: [PMCID: PMC ] [PubMed: ] 29. Weisberg S. P., McCann D., Desai M., Rosenbaum M., Leibel R. L., Ferrante A. W., Jr Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112: [PMCID: PMC296995] [PubMed: ] 30. Xu H., Barnes G. T., Yang Q., Tan G., Yang D., Chou C. J., Sole J., Nichols A., Ross J. S., Tartaglia L. A., et al Chronic inflammation in fat plays a crucial role in the
15 development of obesity-related insulin resistance. J. Clin. Invest. 112: [PMCID: PMC296998] [PubMed: ] 31. Lumeng C. N., Bodzin J. L., Saltiel A. R Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117: [PMCID: PMC ] [PubMed: ] 32. Hotamisligil G. S Inflammation and metabolic disorders. Nature. 444: [PubMed: ] 33. Aiello R. J., Brees D., Bourassa P. A., Royer L., Lindsey S., Coskran T., Haghpassand M., Francone O. L Increased atherosclerosis in hyperlipidemic mice with inactivation of ABCA1 in macrophages. Arterioscler. Thromb. Vasc. Biol. 22: [PubMed: ] 34. van Eck M., Bos I. S., Kaminski W. E., Orsó E., Rothe G., Twisk J., Böttcher A., Van Amersfoort E. S., Christiansen-Weber T. A., Fung-Leung W. P., et al Leukocyte ABCA1 controls susceptibility to atherosclerosis and macrophage recruitment into tissues. Proc. Natl. Acad. Sci. USA. 99: [PMCID: PMC122943] [PubMed: ] 35. Yvan-Charvet L., Ranalletta M., Wang N., Han S., Terasaka N., Li R., Welch C., Tall A. R Combined deficiency of ABCA1 and ABCG1 promotes foam cell accumulation and accelerates atherosclerosis in mice. J. Clin. Invest. 117: [PMCID: PMC ] [PubMed: ] 36. Han M. S., Jung D. Y., Morel C., Lakhani S. A., Kim J. K., Flavell R. A., Davis R. J JNK expression by macrophages promotes obesity-induced insulin resistance and inflammation. Science. 339: [PMCID: PMC ] [PubMed: ] 37. Vijay-Kumar M., Aitken J. D., Carvalho F. A., Ziegler T. R., Gewirtz A. T., Ganji V Loss of function mutation in toll-like receptor-4 does not offer protection against obesity and insulin resistance induced by a diet high in trans fat in mice. J. Inflamm. (Lond.). 8: 2. [PMCID: PMC ] [PubMed: ] 38. Hosoi T., Yokoyama S., Matsuo S., Akira S., Ozawa K Myeloid differentiation factor 88 (MyD88)-deficiency increases risk of diabetes in mice. PLoS ONE. 5: e [PMCID: PMC ] [PubMed: ] 39. Erridge C., Samani N. J Saturated fatty acids do not directly stimulate Toll-like receptor signaling. Arterioscler. Thromb. Vasc. Biol. 29: [PubMed: ]
16 Figures and Tables Fig. 1.
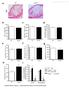
17 Myeloid-specific ABCA1 deficiency increases cholesterol accumulation but does not enhance obesity-associated inflammation in peritoneal macrophages. Mice were euthanized and resident peritoneal macrophages were isolated for quantification of cholesterol by gas-liquid chromatography or gene expression by quantitative real-time PCR. A, C: FC and CE content of resident peritoneal macrophages from WT and MSKO mice fed the HF diet for 24 weeks. B, D: FC and CE content in resident peritoneal macrophages from 14-week-old chow-fed ob/ob and MSKO-ob/ob mice. E, F: Cytokine and chemokine mrna expression in resident peritoneal macrophages from HF diet-fed WT and MSKO mice (E) and chow-fed ob/ob and MSKO-o/ob mice (F). G, H: TLR4 and CD36 mrna expression in resident peritoneal macrophages from HF diet-fed WT and MSKO mice (G) and chow-fed ob/ob and MSKO-o/ob mice (H). *P < 0.05; **P < 0.01; ***P < ns, no significance. +/+, WT; M/ M, MSKO.
18 Fig. 2. Myeloid-specific ABCA1 deficiency does not worsen insulin resistance in HF diet-fed mice. Male mice, 8 9 weeks old, were fed a HF diet consisting of 45% calories from lard and 0.015% cholesterol for 24 weeks. A: BW over the 24 week diet feeding period. B: Fasting plasma glucose was measured after 20 weeks of HF diet consumption, after a 5 h fast. GTTs (C) and ITTs (D) were performed after 20 weeks of HF diet feeding. Plasma insulin levels of fed (E) or fasted (overnight) (F) mice were measured after 20 weeks of HF diet feeding. +/+, WT; M/ M, MSKO.
19 Fig. 3. Myeloid-specific ABCA1 deficiency does not worsen insulin resistance in male ob/ob mice. MSKO mice were crossed with ob/ob mice to induce obesity. A, B: BW was measured at 8 and 14 weeks of age. GTTs (C) and ITTs (D) were performed at 13 and 14 weeks of age, respectively. Plasma insulin levels in fed (E) or overnight-fasted (F) mice were measured at 13 weeks of age.
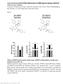
20 Fig. 4. Myeloid-specific ABCA1 deficiency does not worsen adipose macrophage infiltration and inflammation in HF diet-fed mice. A: Epididymal fat mrna expression of macrophage markers (F4/80 and CD68) from mice fed the HF diet for 17 weeks and mice fed the chow diet for 20 weeks. B: Macrophages in adipose tissue were visualized by immunohistochemistry staining using CD68 antibody (objective magnification 10). CD68+ cells were quantified by Image-Pro
21 software. Data are presented as percentage of total area that is CD68+. C, D: Epididymal fat mrna expression of TNF-α, MCP-1, arginase I, and Ch3l3 from mice fed chow for 20 weeks or HF diet for 17 weeks. Data are expressed as mean ± SEM. *P < ***P < ns, no significance. +/+, WT; M/ M, MSKO.
22 Fig. 5. Myeloid-specific ABCA1 deficiency does not worsen adipose macrophage infiltration and inflammation in ob/ob mice. A: Percentage of macrophages (F4/80 + ), M1 macrophages
23 (F4/80 + CD11C + ), or M2 macrophages (F4/80 + CD206 + ) in stromal vascular cells isolated from ob/ob epididymal fat. Data are expressed as mean ± SEM. B: Epididymal fat mrna expression of macrophage markers and inflammatory cytokines and chemokines from chow-fed 14-weekold MSKO-ob/ob and ob/ob mice. Data are expressed as mean ± SEM. C: FC and CE content in stromal vascular cells (SVF). Data are expressed as mean ± range. *P < ns, not significantly different.
24 Fig. 6.
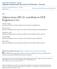
25 Myeloid-specific ABCA1 deficiency does not alter plasma and liver lipid homeostasis in the setting of obesity. A, C: Blood samples from HF diet-fed mice were collected periodically to assay total cholesterol (TC) and TGs by enzymatic assays. B, D: Blood samples were collected from 14-week-old MSKO-ob/ob and ob/ob mice to assay TC and TGs by enzymatic assays. E H: After 24 weeks HF diet consumption or at the age of 14 weeks for ob/ob mice, mice were euthanized and liver lipids were quantified by enzymatic assay and normalized to liver protein content. +/+, WT; M/ M, MSKO.
26 Supplemental Materials Myeloid Cell -Specific ABCA1 Deletion Does Not Worsen Insulin Resistance in High Fat Diet-Induced or Genetically Obese Mouse Models Xuewei Zhu 1, Soonkyu Chung 1, 2, Xin Bi 1, Chia-Chi Chuang 1, Amanda L. Brown 1, Mingxia Liu 1, Jeongmin Seo 1, Helen Cuffe 1, Abraham K. Gebre 1, Elena Boudyguina 1, John S. Parks 1, 3 1 Department of Pathology/Lipid Sciences; 3 Department of Biochemistry, Wake Forest School of Medicine, Winston-Salem, NC, USA; 2 Department of Food Science and Human Nutrition, The University of Florida, Gainesville, Florida, USA Supplemental Table I: High-fat diet composition Ingredient Grams/100 g Grams of fat Grams of Grams of protein carbohydrate Casein L-cysteine Dextrin 8.00 Maltodextrin Sucrose Cellulose Soybean oil Lard Hegsted Salts 5.0 Vitamin Mix MtS-50 (Vitamin E) 0.20 Total (calories) % of Calories 45% 35% 20% Supplemental Figure 1. Liver (A and C) and epididymal fat (B and D) mass from mice consuming the HF diet for 24 wks or chow-fed mice crossed into the ob/ob background (14 wks old). +/+= WT; -M/- M=MSKO. Supplemental Figure 2. Immunohistochemical staining and quantification of cleaved caspase-3 positive cells (apoptotic) in adipose tissues from mice fed a HF diet for 17 wks (objective magnification 10). Cleaved caspase-3 positive cells were quantified using Image Pro software. ns= not statistically significant. ns= not significant; p>0.05.
27 Supplemental Figure 3. (A) mrna expression of F4/80 and CD68 in epididymal fat from mice consuming a HF diet for 24 wks. (B) mrna expression of TLR4 and CD36 in epididymal fat from mice consuming chow (20 wks) or HF diet (17 wks). (C) mrna expression of inflammatory cytokines/chemokines in epididymal fat from mice consuming a HF diet for 24 wks. Gene expression was evaluated by quantitative real-time PCR. +/+= WT; -M/-M=MSKO. ns= not significant; p>0.05.
28 Supplemental Figure 1 A HF diet C ob/ob B D
29 Supplemental Figure 2 A -M/-M (HF) +/+ (HF) 100 um B
30 Supplemental Figure 3 A B C
Effect of High-fat or High-glucose Diet on Obesity and Visceral Adipose Tissue in Mice
 ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
Molecular Medicine. Myeloid Cell Specific ABCA1 Deletion Protects Mice From Bacterial Infection
 Molecular Medicine Myeloid Cell Specific ABCA1 Deletion Protects Mice From Bacterial Infection Xuewei Zhu, Marlena M. Westcott, Xin Bi, Mingxia Liu, Kymberly M. Gowdy, Jeongmin Seo, Qiang Cao, Abraham
Molecular Medicine Myeloid Cell Specific ABCA1 Deletion Protects Mice From Bacterial Infection Xuewei Zhu, Marlena M. Westcott, Xin Bi, Mingxia Liu, Kymberly M. Gowdy, Jeongmin Seo, Qiang Cao, Abraham
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Hematopoietic ABCA1 deletion promotes monocytosis and worsens diet-induced insulin resistance in mice
 Hematopoietic ABCA1 deletion promotes monocytosis and worsens diet-induced insulin resistance in mice Chongren Tang, 1 Yuhua Liu, Wendy Yang, Carl Storey, Tim S. McMillen, Barbara A. Houston, Jay W. Heinecke,
Hematopoietic ABCA1 deletion promotes monocytosis and worsens diet-induced insulin resistance in mice Chongren Tang, 1 Yuhua Liu, Wendy Yang, Carl Storey, Tim S. McMillen, Barbara A. Houston, Jay W. Heinecke,
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity
 Downloaded from http:// on December 17, 2017. https://doi.org/10.1172/jci.insight.87748 Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity Xianfeng Wang, 1
Downloaded from http:// on December 17, 2017. https://doi.org/10.1172/jci.insight.87748 Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity Xianfeng Wang, 1
Hepatic ABC transporters and triglyceride metabolism
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2012 Hepatic ABC transporters
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2012 Hepatic ABC transporters
The role of the ABCA1 transporter and cholesterol efflux in familial hypoalphalipoproteinemia
 The role of the ABCA1 transporter and cholesterol efflux in familial hypoalphalipoproteinemia G. Kees Hovingh,* Michel J. A. van Wijland, Alison Brownlie, Radjesh J. Bisoendial,* Michael R. Hayden,** John
The role of the ABCA1 transporter and cholesterol efflux in familial hypoalphalipoproteinemia G. Kees Hovingh,* Michel J. A. van Wijland, Alison Brownlie, Radjesh J. Bisoendial,* Michael R. Hayden,** John
Naohito AOKI, Erina ARAKAWA and Miyuki ITO. Department of Life Science, Graduate School of Bioresources, Mie University Tsu ABSTRACT
 Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Naohito AOKI, Erina ARAKAWA and Miyuki ITO Department of Life Science, Graduate School of Bioresources, Mie University Tsu 514-857 ABSTRACT C57BL/6J mice (male, 4wk old) were fed low fat diet (LF), high
Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice.
 Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
University of California, San Diego La Jolla CA 92093
 AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
Summary and concluding remarks
 Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Leptin deficiency suppresses progression of atherosclerosis in apoe-deficient mice
 Leptin deficiency suppresses progression of atherosclerosis in apoe-deficient mice Atherosclerosis, 2007 Chiba T, Shinozaki S, Nakazawa T, et al. Present by Sudaporn Pummoung Apolipoprotein E (apoe( apoe)
Leptin deficiency suppresses progression of atherosclerosis in apoe-deficient mice Atherosclerosis, 2007 Chiba T, Shinozaki S, Nakazawa T, et al. Present by Sudaporn Pummoung Apolipoprotein E (apoe( apoe)
We obtained Male C57BL/6J, ob/ob, and Cd8a-deficient mice from Charles River Japan
 Supplementary Methods Animal models We obtained Male C57BL/6J, ob/ob, and Cd8a-deficient mice from Charles River Japan or Jackson Laboratories. All mice were housed under a 12-h light-dark cycle and allowed
Supplementary Methods Animal models We obtained Male C57BL/6J, ob/ob, and Cd8a-deficient mice from Charles River Japan or Jackson Laboratories. All mice were housed under a 12-h light-dark cycle and allowed
Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity.
 Supplementary Figure 1 Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity. Male C57BL/6J mice were fed a normal chow (NC, 10% fat) or a high-fat diet (HFD,
Supplementary Figure 1 Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity. Male C57BL/6J mice were fed a normal chow (NC, 10% fat) or a high-fat diet (HFD,
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Protein extraction and western blot analysis Protein extraction was performed as
 ESM Methods Protein extraction and western blot analysis Protein extraction was performed as previously described [1]. 2 g protein was loaded on SDSPAGE and immunoblotted with antibodies to mouse AKT (1:1,
ESM Methods Protein extraction and western blot analysis Protein extraction was performed as previously described [1]. 2 g protein was loaded on SDSPAGE and immunoblotted with antibodies to mouse AKT (1:1,
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by. activating Syk and promoting MyD88 and TRIF degradation via cbl-b
 Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Nature Medicine doi: /nm.3150
 Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO
 Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Supplemental Table 1: Demographics and characteristics of study participants. Male, n (%) 3 (20%) 6 (50%) Age, years [mean ± SD] 33.3 ± ± 9.
![Supplemental Table 1: Demographics and characteristics of study participants. Male, n (%) 3 (20%) 6 (50%) Age, years [mean ± SD] 33.3 ± ± 9. Supplemental Table 1: Demographics and characteristics of study participants. Male, n (%) 3 (20%) 6 (50%) Age, years [mean ± SD] 33.3 ± ± 9.](/thumbs/90/103696086.jpg) SUPPLEMENTAL DATA Supplemental Table 1: Demographics and characteristics of study participants Lean (n=15) Obese (n=12) Male, n (%) 3 (20%) 6 (50%) Age, years [mean ± SD] 33.3 ± 9.5 44.8 ± 9.1 White, n
SUPPLEMENTAL DATA Supplemental Table 1: Demographics and characteristics of study participants Lean (n=15) Obese (n=12) Male, n (%) 3 (20%) 6 (50%) Age, years [mean ± SD] 33.3 ± 9.5 44.8 ± 9.1 White, n
alveolar macrophages (AMs) after 24 hours of in vitro culture in complete medium
 Online Supplement for: NF-κB ACTIVATION IN ALVEOLAR MACROPHAGES REQUIRES IκB KINASE-β, BUT NOT NF-κB INDUCING KINASE Supershift and Competition Assays for NF-κB Competition and supershift assays were performed
Online Supplement for: NF-κB ACTIVATION IN ALVEOLAR MACROPHAGES REQUIRES IκB KINASE-β, BUT NOT NF-κB INDUCING KINASE Supershift and Competition Assays for NF-κB Competition and supershift assays were performed
CHAPTER Cir. Res. 2010; 107(12): e
 CHAPTER 5 Enhanced foam cell formation, atherosclerotic lesion development, and inflammation by combined deletion of ABCA1 and SR-BI in bone marrow-derived cells in LDL receptor knockout mice on Western-type
CHAPTER 5 Enhanced foam cell formation, atherosclerotic lesion development, and inflammation by combined deletion of ABCA1 and SR-BI in bone marrow-derived cells in LDL receptor knockout mice on Western-type
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
SUPPLEMENTARY METHODS
 SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
SUPPLEMENTARY METHODS Histological analysis. Colonic tissues were collected from 5 parts of the middle colon on day 7 after the start of DSS treatment, and then were cut into segments, fixed with 4% paraformaldehyde,
Quantitative Real-Time PCR was performed as same as Materials and Methods.
 Supplemental Material Quantitative Real-Time PCR Quantitative Real-Time PCR was performed as same as Materials and Methods. Expression levels in the aorta were normalized to peptidylprolyl isomerase B
Supplemental Material Quantitative Real-Time PCR Quantitative Real-Time PCR was performed as same as Materials and Methods. Expression levels in the aorta were normalized to peptidylprolyl isomerase B
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle.
 Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
HVEM-deficient mice fed a high-fat diet are protected from adipose tissue inflammation and glucose intolerance
 FEBS Letters 585 (2011) 2285 2290 journal homepage: www.febsletters.org HVEM-deficient mice fed a high-fat diet are protected from adipose tissue inflammation and glucose intolerance Ha-Jung Kim a, Hong-Min
FEBS Letters 585 (2011) 2285 2290 journal homepage: www.febsletters.org HVEM-deficient mice fed a high-fat diet are protected from adipose tissue inflammation and glucose intolerance Ha-Jung Kim a, Hong-Min
Dietary Cholesterol Promotes Adipocyte Hypertrophy and Adipose Tissue Inflammation in Visceral, But Not Subcutaneous, Fat in Monkeys
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2014 Dietary Cholesterol
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2014 Dietary Cholesterol
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Supplementary Figure 1
 Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
 Glossary For TheFatNurse s For All Ages Series Adipocytes, also known as lipocytes and fat cells, are the cells that primarily compose adipose tissue, specialized in storing energy as fat. Apolipoprotein
Glossary For TheFatNurse s For All Ages Series Adipocytes, also known as lipocytes and fat cells, are the cells that primarily compose adipose tissue, specialized in storing energy as fat. Apolipoprotein
Supplemental Information. Increased 4E-BP1 Expression Protects. against Diet-Induced Obesity and Insulin. Resistance in Male Mice
 Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Primer sequences Target Sequence F Sequence R TNF-α (Tnfa) TCAGCCGATTTGCTATCTCAT A
 Supplementary Table 1. Q- and RT-PR primers used in this study. Primer sequences Target Sequence F Sequence R TNF-α (Tnfa) TGGTTTGTTTT GTTTGGGGTTG T hemokine (- motif) ligand 5 (cl5) GTGTTTGTTT TGGTGGTG
Supplementary Table 1. Q- and RT-PR primers used in this study. Primer sequences Target Sequence F Sequence R TNF-α (Tnfa) TGGTTTGTTTT GTTTGGGGTTG T hemokine (- motif) ligand 5 (cl5) GTGTTTGTTT TGGTGGTG
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Effect of BI-1 on insulin resistance through regulation of CYP2E1
 Effect of BI-1 on insulin resistance through regulation of CYP2E1 Geum-Hwa Lee 1, Kyoung-Jin Oh 2, 3, Hyung-Ryong Kim 4, Hye-Sook Han 2, Hwa-Young Lee 1, Keun-Gyu Park 5, Ki-Hoan Nam 6, Seung-Hoi Koo 2
Effect of BI-1 on insulin resistance through regulation of CYP2E1 Geum-Hwa Lee 1, Kyoung-Jin Oh 2, 3, Hyung-Ryong Kim 4, Hye-Sook Han 2, Hwa-Young Lee 1, Keun-Gyu Park 5, Ki-Hoan Nam 6, Seung-Hoi Koo 2
Gut microbiota, metabolic syndrome, obesity and the nutrient sensor pathways
 Gut microbiota, metabolic syndrome, obesity and the nutrient sensor pathways Department of Gastroenterology, Endocrinology & Metabolism Medical University Innsbruck Herbert Tilg Nothing to disclose Fig.
Gut microbiota, metabolic syndrome, obesity and the nutrient sensor pathways Department of Gastroenterology, Endocrinology & Metabolism Medical University Innsbruck Herbert Tilg Nothing to disclose Fig.
Stewart et al. CD36 ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer
 NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
ABCA1 mutants reveal an interdependency between lipid export function, apoa-i binding activity, and Janus kinase 2 activation
 ABCA1 mutants reveal an interdependency between lipid export function, apoa-i binding activity, and Janus kinase 2 activation Ashley M. Vaughan, Chongren Tang, and John F. Oram 1 Division of Metabolism,
ABCA1 mutants reveal an interdependency between lipid export function, apoa-i binding activity, and Janus kinase 2 activation Ashley M. Vaughan, Chongren Tang, and John F. Oram 1 Division of Metabolism,
Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes
 Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes T Jourdan, G Godlewski, R Cinar, A Bertola, G Szanda, J Liu, J Tam, T Han, B Mukhopadhyay,
Endocannabinoid-activated Nlrp3 inflammasome in infiltrating macrophages mediates β- cell loss in type 2 diabetes T Jourdan, G Godlewski, R Cinar, A Bertola, G Szanda, J Liu, J Tam, T Han, B Mukhopadhyay,
High density lipoprotein metabolism
 High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
Supplementary Figure 1
 Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
Novel function of NADPH oxidase in atherosclerosis. Yun Soo Bae Department of Life Science Ewha Womans University
 Novel function of NADPH oxidase in atherosclerosis Yun Soo Bae Department of Life Science Ewha Womans University Recent understanding of ROS: act as second messengers e e Catalase/peroxidase O 2 H 2 O
Novel function of NADPH oxidase in atherosclerosis Yun Soo Bae Department of Life Science Ewha Womans University Recent understanding of ROS: act as second messengers e e Catalase/peroxidase O 2 H 2 O
Adipose tissue ABCA1 contributes to HDL biogenesis in vivo
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2011 Adipose tissue ABCA1
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Nutrition and Health Sciences -- Faculty Publications Nutrition and Health Sciences, Department of 2011 Adipose tissue ABCA1
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
SUPPLEMENTARY INFORMATION. Supplemental Figure 1. Body weight and blood glucose parameters of chow-diet (CD)
 SUPPLEMENTARY INFORMATION LEGENDS Supplemental Figure. Body weight and blood glucose parameters of chow-diet (CD) fed and high-fat diet (HFD) fed mice. (A) Body weight was measured at the beginning of
SUPPLEMENTARY INFORMATION LEGENDS Supplemental Figure. Body weight and blood glucose parameters of chow-diet (CD) fed and high-fat diet (HFD) fed mice. (A) Body weight was measured at the beginning of
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas Summary
 Published in "" which should be cited to refer to this work. Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas http://doc.rero.ch Laboratory of Metabolic Stress Biology, Division
Published in "" which should be cited to refer to this work. Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas http://doc.rero.ch Laboratory of Metabolic Stress Biology, Division
Supplementary Figures
 Supplementary Figures mir-150 regulates obesityassociated insulin resistance by controlling B cell functions Wei Ying, Alexander Tseng, Richard Cheng-An Chang, Haiqing Wang, Yu-lieh Lin, Srikanth Kanameni,
Supplementary Figures mir-150 regulates obesityassociated insulin resistance by controlling B cell functions Wei Ying, Alexander Tseng, Richard Cheng-An Chang, Haiqing Wang, Yu-lieh Lin, Srikanth Kanameni,
Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. H-PGDS deficiency does not affect GI tract functions and anaphylactic reaction. (a) Representative pictures of H&E-stained jejunum sections ( 200 magnification;
Gene Polymorphisms and Carbohydrate Diets. James M. Ntambi Ph.D
 Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
well for 2 h at rt. Each dot represents an individual mouse and bar is the mean ±
 Supplementary data: Control DC Blimp-1 ko DC 8 6 4 2-2 IL-1β p=.5 medium 8 6 4 2 IL-2 Medium p=.16 8 6 4 2 IL-6 medium p=.3 5 4 3 2 1-1 medium IL-1 n.s. 25 2 15 1 5 IL-12(p7) p=.15 5 IFNγ p=.65 4 3 2 1
Supplementary data: Control DC Blimp-1 ko DC 8 6 4 2-2 IL-1β p=.5 medium 8 6 4 2 IL-2 Medium p=.16 8 6 4 2 IL-6 medium p=.3 5 4 3 2 1-1 medium IL-1 n.s. 25 2 15 1 5 IL-12(p7) p=.15 5 IFNγ p=.65 4 3 2 1
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
The relationship between coronary artery disease and low
 Cellular Phospholipid and Cholesterol Efflux in High-Density Lipoprotein Deficiency Michel Marcil, PhD; Rachel Bissonnette, MSc; Jérôme Vincent, DEA; Larbi Krimbou, DES; Jacques Genest, MD Background Prospective
Cellular Phospholipid and Cholesterol Efflux in High-Density Lipoprotein Deficiency Michel Marcil, PhD; Rachel Bissonnette, MSc; Jérôme Vincent, DEA; Larbi Krimbou, DES; Jacques Genest, MD Background Prospective
Inflammasome-mediated caspase-1 activity Gatekeeper of inflammation in the adipose tissue. Rinke Stienstra
 Inflammasome-mediated caspase-1 activity Gatekeeper of inflammation in the adipose tissue Rinke Stienstra Obesity promotes the development of insulin resistance and type 2 diabetes County-level Estimates
Inflammasome-mediated caspase-1 activity Gatekeeper of inflammation in the adipose tissue Rinke Stienstra Obesity promotes the development of insulin resistance and type 2 diabetes County-level Estimates
Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel
 Supplementary Figures 1-8 Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel Marc Schmidt 1,2, Badrinarayanan Raghavan 1,2, Verena Müller 1,2, Thomas Vogl 3, György
Supplementary Figures 1-8 Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel Marc Schmidt 1,2, Badrinarayanan Raghavan 1,2, Verena Müller 1,2, Thomas Vogl 3, György
MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity
 Research article MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity Hajime Kanda, 1 Sanshiro Tateya, 1 Yoshikazu Tamori, 1 Ko Kotani,
Research article MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity Hajime Kanda, 1 Sanshiro Tateya, 1 Yoshikazu Tamori, 1 Ko Kotani,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
902 Biomed Environ Sci, 2014; 27(11):
 902 Biomed Environ Sci, 2014; 27(11): 902-906 Letter to the Editor Curcuminoids Target Decreasing Serum Adipocyte-fatty Acid Binding Protein Levels in Their Glucose-lowering Effect in Patients with Type
902 Biomed Environ Sci, 2014; 27(11): 902-906 Letter to the Editor Curcuminoids Target Decreasing Serum Adipocyte-fatty Acid Binding Protein Levels in Their Glucose-lowering Effect in Patients with Type
Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ).
 Supplementary Figure 1 Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ). (b) Immunoblot analysis of TRIM29 in lung primary
Supplementary Figure 1 Trim29 gene-targeting strategy. (a) Genotyping of wildtype mice (+/+), Trim29 heterozygous mice (+/ ) and homozygous mice ( / ). (b) Immunoblot analysis of TRIM29 in lung primary
TD-BF01: Innate immunity to microorganisms
 TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
Nutrition and Age-Associated Inflammation :Role of Nutritional Intervention Simin Nikbin Meydani, DVM, PhD.
 Nutrition and Age-Associated Inflammation :Role of Nutritional Intervention Simin Nikbin Meydani, DVM, PhD simin.meydani@tufts.edu Outline 1) Role of immune and inflammatory responses in age-related diseases
Nutrition and Age-Associated Inflammation :Role of Nutritional Intervention Simin Nikbin Meydani, DVM, PhD simin.meydani@tufts.edu Outline 1) Role of immune and inflammatory responses in age-related diseases
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Obesity has reached epidemic proportions worldwide. In
 National Cholesterol Awareness Month Article Basic Sciences Dietary Cholesterol Promotes Adipocyte Hypertrophy and Adipose Tissue Inflammation in Visceral, but Not in Subcutaneous, Fat in Monkeys Soonkyu
National Cholesterol Awareness Month Article Basic Sciences Dietary Cholesterol Promotes Adipocyte Hypertrophy and Adipose Tissue Inflammation in Visceral, but Not in Subcutaneous, Fat in Monkeys Soonkyu
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ
 ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ ΘΩΜΑΣ ΠΑΠΑΔΟΠΟΥΛΟΣ, MD, PHD ΕΠΕΜΒΑΤΙΚΟΣ ΚΑΡΔΙΟΛΟΓΟΣ ΙΑΤΡΙΚΟ ΔΙΑΒΑΛΚΑΝΙΚΟ ΚΕΝΤΡΟ Inflammation as a cause of disease has entered the popular imagination. Diet ( macronutrients )
ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ ΘΩΜΑΣ ΠΑΠΑΔΟΠΟΥΛΟΣ, MD, PHD ΕΠΕΜΒΑΤΙΚΟΣ ΚΑΡΔΙΟΛΟΓΟΣ ΙΑΤΡΙΚΟ ΔΙΑΒΑΛΚΑΝΙΚΟ ΚΕΝΤΡΟ Inflammation as a cause of disease has entered the popular imagination. Diet ( macronutrients )
Supplementary Table I - Primers used for real-time quantitative PCR and RT-PCR
 Supplement Supplementary Table I - Primers used for real-time quantitative PCR and RT-PCR Gene Forward Primer (5-3 ) Reverse primer (5-3 ) Reference Human ST2 CTTGATTGATAAACAGAATG CTGATCCAGATACTGTTGAA
Supplement Supplementary Table I - Primers used for real-time quantitative PCR and RT-PCR Gene Forward Primer (5-3 ) Reverse primer (5-3 ) Reference Human ST2 CTTGATTGATAAACAGAATG CTGATCCAGATACTGTTGAA
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Monocyte/macrophage expression of ABCA1 has minimal contribution to plasma HDL levels
 Monocyte/macrophage expression of ABCA1 has minimal contribution to plasma HDL levels See related Commentary on pages 1273 1275. Mehrdad Haghpassand, Patricia-Ann K. Bourassa, Omar L. Francone, and Robert
Monocyte/macrophage expression of ABCA1 has minimal contribution to plasma HDL levels See related Commentary on pages 1273 1275. Mehrdad Haghpassand, Patricia-Ann K. Bourassa, Omar L. Francone, and Robert
INTRODUCTION. Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells
 Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells Galina Chipitsyna, Qiaoke Gong, Chance F. Gray et al. Endocrinology,
Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells Galina Chipitsyna, Qiaoke Gong, Chance F. Gray et al. Endocrinology,
* Author to whom correspondence should be addressed; Tel.: ; Fax:
 Int. J. Mol. Sci. 2014, 15, 6184-6223; doi:10.3390/ijms15046184 Review OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Obesity and Its Metabolic Complications:
Int. J. Mol. Sci. 2014, 15, 6184-6223; doi:10.3390/ijms15046184 Review OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Obesity and Its Metabolic Complications:
IL-6Rα IL-6RαT-KO KO. IL-6Rα f/f bp. f/f 628 bp deleted 368 bp. 500 bp
 STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
STD H 2 O WT KO IL-6Rα f/f IL-6Rα IL-6RαT-KO KO 1000 bp 500 bp f/f 628 bp deleted 368 bp Supplementary Figure 1 Confirmation of T-cell IL-6Rα deficiency. (a) Representative histograms and (b) quantification
METABOLISM of ADIPOSE TISSUE
 METABOLISM of ADIPOSE TISSUE 2. LF UK Prof. Rudolf Poledne, PhD. TYPES OF ADIPOSE TISSUE brown adipose tissue subcutaneous adipose tissue visceral adipose tissue ADIPOSE TISSUE FUNCTIONS: thermal isolation
METABOLISM of ADIPOSE TISSUE 2. LF UK Prof. Rudolf Poledne, PhD. TYPES OF ADIPOSE TISSUE brown adipose tissue subcutaneous adipose tissue visceral adipose tissue ADIPOSE TISSUE FUNCTIONS: thermal isolation
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats
 J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
Yiying Zhang, PhD Research Scientist. Research Summary:
 Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
3/20/2011. Body Mass Index (kg/[m 2 ]) Age at Issue (*BMI > 30, or ~ 30 lbs overweight for 5 4 woman) Mokdad A.H.
![3/20/2011. Body Mass Index (kg/[m 2 ]) Age at Issue (*BMI > 30, or ~ 30 lbs overweight for 5 4 woman) Mokdad A.H. 3/20/2011. Body Mass Index (kg/[m 2 ]) Age at Issue (*BMI > 30, or ~ 30 lbs overweight for 5 4 woman) Mokdad A.H.](/thumbs/82/84935173.jpg) U.S. Adults: 1988 Nineteen states with 10-14% 14% Prevalence of Obesity (*BMI > 30, or ~ 30 lbs overweight for 5 4 woman) Metabolic John P. Cello, MD Professor of Medicine and Surgery, University of California,
U.S. Adults: 1988 Nineteen states with 10-14% 14% Prevalence of Obesity (*BMI > 30, or ~ 30 lbs overweight for 5 4 woman) Metabolic John P. Cello, MD Professor of Medicine and Surgery, University of California,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/4/199/ra75/dc1 Supplementary Materials for Signaling by the Matrix Proteoglycan Decorin Controls Inflammation and Cancer Through PDCD4 and MicroRNA-21 Rosetta
www.sciencesignaling.org/cgi/content/full/4/199/ra75/dc1 Supplementary Materials for Signaling by the Matrix Proteoglycan Decorin Controls Inflammation and Cancer Through PDCD4 and MicroRNA-21 Rosetta
STAT4 Deficiency Reduces Obesity-Induced Insulin Resistance and Adipose Tissue Inflammation
 ORIGINAL ARTICLE STAT4 Deficiency Reduces Obesity-Induced Insulin Resistance and Adipose Tissue Inflammation Anca D. Dobrian, 1 Elena V. Galkina, 2 Qian Ma, 1 Margaret Hatcher, 1 Sabai Myo Aye, 1 Mathew
ORIGINAL ARTICLE STAT4 Deficiency Reduces Obesity-Induced Insulin Resistance and Adipose Tissue Inflammation Anca D. Dobrian, 1 Elena V. Galkina, 2 Qian Ma, 1 Margaret Hatcher, 1 Sabai Myo Aye, 1 Mathew
IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia
 Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Supplementary Figures IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia Yaming Wang, Kristy J. Szretter, William Vermi, Susan Gilfillan, Cristina
Taylor Yohe. Project Advisor: Dr. Martha A. Belury. Department of Human Nutrition at the Ohio State University
 Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Fig. S1. REGN1500 reduces plasma levels of cholesterol, TG and NEFA in WT and Ldlr -/- mice. (A) WT
 Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
LPS LPS P6 - + Supplementary Fig. 1.
 P6 LPS - - - + + + - LPS + + - - P6 + Supplementary Fig. 1. Pharmacological inhibition of the JAK/STAT blocks LPS-induced HMGB1 nuclear translocation. RAW 267.4 cells were stimulated with LPS in the absence
P6 LPS - - - + + + - LPS + + - - P6 + Supplementary Fig. 1. Pharmacological inhibition of the JAK/STAT blocks LPS-induced HMGB1 nuclear translocation. RAW 267.4 cells were stimulated with LPS in the absence
SR-BI inhibits ABCG1-stimulated net cholesterol efflux from cells to plasma HDL
 SR-BI inhibits ABCG1-stimulated net cholesterol efflux from cells to plasma HDL Laurent Yvan-Charvet, 1, * Tamara A. Pagler,* Nan Wang,* Takafumi Senokuchi,* May Brundert, Hongna Li,* Franz Rinninger,
SR-BI inhibits ABCG1-stimulated net cholesterol efflux from cells to plasma HDL Laurent Yvan-Charvet, 1, * Tamara A. Pagler,* Nan Wang,* Takafumi Senokuchi,* May Brundert, Hongna Li,* Franz Rinninger,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Targeting tumour associated macrophages in anti-cancer therapies. Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018
 Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
Supplementary Information
 Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
FAPESP Week /21/2017
 Texas Tech University Dr Naima Moustaid-Moussa Texas Tech University FAPESP Week 2017 09/21/2017 University of Sao Paulo Dr Sonia Jancar ICB-USP Theresa Ramalho MS PhD candidate, Immunology ICB-USP Dr
Texas Tech University Dr Naima Moustaid-Moussa Texas Tech University FAPESP Week 2017 09/21/2017 University of Sao Paulo Dr Sonia Jancar ICB-USP Theresa Ramalho MS PhD candidate, Immunology ICB-USP Dr
 Glossary For TheFatNurse s For All Ages Series Apolipoprotein B (APOB or ApoB) are the primary apolipoproteins of chylomicrons and low-density lipoproteins (LDL - known commonly by the misnomer "bad cholesterol"
Glossary For TheFatNurse s For All Ages Series Apolipoprotein B (APOB or ApoB) are the primary apolipoproteins of chylomicrons and low-density lipoproteins (LDL - known commonly by the misnomer "bad cholesterol"
Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy. Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L.
 Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L. Bartel 1 Ixmyelocel-T, an expanded, autologous multicellular therapy cultured
Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L. Bartel 1 Ixmyelocel-T, an expanded, autologous multicellular therapy cultured
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
New therapeutic targets for T2DM
 New therapeutic targets for T2DM Targeting inflammation: NF- B, salsalate Gwanpyo Koh Department of Internal Medicine Jeju National University School of Medicine Introduction Obesity is occurring at epidemic
New therapeutic targets for T2DM Targeting inflammation: NF- B, salsalate Gwanpyo Koh Department of Internal Medicine Jeju National University School of Medicine Introduction Obesity is occurring at epidemic
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
