Jang, H; Lee, GY; Selby, CP; Lee, G; Jeon, YG; Lee, JH; Cheng, KY; Titchenell, P; Birnbaum, MJ; Xu, A; Sancar, A; Kim, JB
|
|
|
- Lester Simpson
- 5 years ago
- Views:
Transcription
1 Title SREBPc- signalling represses hepatic glucose production by promoting FOXO degradation during refeeding Author(s) Jang, H; Lee, GY; Selby, CP; Lee, G; Jeon, YG; Lee, JH; Cheng, KY; Titchenell, P; Birnbaum, MJ; Xu, A; Sancar, A; Kim, JB Citation Nature Communications, 6, v. 7, p. 8:-4 Issued Date 6 URL Rights This work is licensed under a Creative Commons Attribution- NonCommercial-NoDerivatives 4. International License.
2 Received 7 Aug 5 Accepted 8 Jun 6 Published 4 Jul 6 DOI:.38/ncomms8 OPEN SREBPc- signalling represses hepatic glucose production by promoting FOXO degradation during refeeding Hagoon Jang, Gha Young Lee, Christopher P. Selby, Gung Lee, Yong Geun Jeon, Jae Ho Lee, Kenneth King Yip Cheng 3, Paul Titchenell 4, Morris J. Birnbaum 4, Aimin Xu 3, Aziz Sancar & Jae Bum Kim SREBPc is a key lipogenic transcription factor activated by insulin in the postprandial state. Although SREBPc appears to be involved in suppression of hepatic gluconeogenesis, the molecular mechanism is not thoroughly understood. Here we show that is activated by insulin-induced SREBPc and decreases hepatic gluconeogenesis through FOXO degradation, at least, at specific circadian time points. SREBPc / and / mice show higher blood glucose than wild-type (WT) mice in pyruvate tolerance tests, accompanied with enhanced expression of and genes. promotes degradation of nuclear FOXO by promoting its binding to the ubiquitin E3 ligase MDM. Although SREBPc fails to upregulate expression in db/db mice, overexpression of attenuates hyperglycaemia through reduction of hepatic FOXO protein and gluconeogenic gene expression. These data suggest that insulin-activated SREBPc downregulates gluconeogenesis through -mediated FOXO degradation and that dysregulation of hepatic SREBPc- signalling may contribute to hyperglycaemia in diabetic animals. School of Biological Sciences, Institute of Molecular Biology and Genetics, Center for Adipose Tissue Remodeling, Seoul National University, Seoul 5-74, Korea. Department of Biochemistry and Biophysics, University of North Carolina School of Medicine, CB # 76, Chapel Hill, North Carolina , USA. 3 State Key Laboratory of Pharmaceutical Biotechnology and Department of Medicine, The University of Hong Kong, Hong Kong, 99977, China. 4 The Institute for Diabetes, Obesity and Metabolism, Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania 94, USA. Correspondence and requests for materials should be addressed to J.B.K. ( jaebkim@snu.ac.kr). NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
3 NATURE COMMUNICATIONS DOI:.38/ncomms8 Insulin, which is released from pancreatic b-cells, plays a key role in the maintenance of the whole body energy homoeostasis by actively regulating glucose and lipid metabolism. In the postprandial state, insulin lowers blood glucose by stimulating glucose uptake in adipose tissues and muscles as well as by inhibiting hepatic glucose production,. Moreover, in the liver, insulin stimulates the conversion of excess glucose into glycogen (glycogenesis) and triacylglyceride (lipogenesis) for the long-term energy storage 35. Suppression of hepatic gluconeogenesis by insulin is an important process to inhibit hyperglycaemia. and are crucial enzymes that convert pyruvate to glucose, and their gene expression is regulated by several transcription factors such as Forkhead box O (FOXO), camp response elementbinding protein (CREB), hepatocyte nuclear factor 4 (HNF4), glucocorticoid receptor and peroxisome proliferator-activated receptor gamma coactivator -alpha (PGCa) 68. In the liver, FOXO is activated on fasting and gets inactivated by feeding, which is one of the essential mechanisms by which insulin rapidly and efficiently represses hepatic glucose production during postprandial periods 9. After insulin treatment, FOXO protein is phosphorylated by AKT and then moves to the cytoplasm, resulting in the decrease of gluconeogenic gene expression. Although the translocation of hepatic FOXO from the nucleus to the cytoplasm is a well-defined mechanism mediating a quick decrease in glucose production by insulin, it is largely unknown how insulin endows a sustainable inhibition of hepatic gluconeogenesis throughout the postprandial state. On the other hand, SREBPc has been proposed to be involved in the regulation of hepatic glucose metabolism. SREBPc is a basic-helix-loop-helix-leucine zipper (bhlh-lz) transcription factor that regulates de novo lipogenesis 37. Activation of SREBPc is mediated by AKT and mtorc on insulin signalling 8,9. SREBPc regulates lipogenic pathways by stimulating the expression of target genes such as those encoding fatty acid synthase (FASN), stearoyl-coa desaturase (SCD), and acetyl-coenzyme A carboxylase (ACC). In addition, SREBPc appears to regulate hepatic carbohydrate metabolism. For example, SREBPc affects the mrna levels of, and IRS- genes and inhibits the interaction between HNF4 and PGCa to suppress gluconeogenic genes 37. Although hepatic SREBPc has been reported to be upregulated in obese animals, the reason why increased SREBPc fails to repress hepatic gluconeogenesis is unknown. Thus, understanding the molecular mechanisms by which SREBPc could modulate gluconeogenesis under physiological and pathological conditions is important. is a member of the mammalian clock genes regulated by transcriptiontranslation feedback loop that also includes CLOCK, BMAL, PER, PER and CRY, to modulate rhythmic oscillations. CLOCK and BMAL form a heterodimer to activate PER and CRY genes and then elevated PER and CRY proteins act as transcriptional repressors that decrease the transcriptional activity of CLOCK and BMAL (refs 83). The hepatic circadian clock is regulated by food intake and by the expression of hormones such as insulin and glucagon, whereas the suprachiasmatic nucleus circadian clock is controlled by the lightdark cycle 3. Recently, it has been shown that hepatic circadian clock genes also contribute to glucose homoeostasis. For example, hepatic CRY proteins modulate glucose production by inhibiting the glucagon receptor signalling pathway and binding to glucocorticoid receptor 33,34. In addition, an agonist of CRY proteins has been reported to repress the expression of hepatic gluconeogenic genes, such as and 35. Furthermore, BMAL / mice exhibited disrupted hepatic glucose homoeostasis 36. However, the molecular mechanisms by which could repress hepatic glucose production during the postprandial state remain to be elucidated. The fact that SREBPc downregulates hepatic gluconeogenesis prompted us to investigate novel target genes of SREBPc by comparing SREBPc þ / þ and SREBPc / mice. Here we demonstrate that SREBPc attenuates hepatic glucose production via the activation of at certain circadian time points (particularly at ZT 3), eventually leading to degradation of the FOXO protein on insulin signalling. While hepatic FOXO is rapidly translocated from the nucleus into the cytoplasm by AKTmediated phosphorylation triggered by insulin, the SREBPc- signalling pathway durably represses the execution of gluconeogenic genes by decreasing nuclear FOXO protein for long-term insulin action. In addition, in the liver of diabetic db/db mice, overexpression of lowers blood glucose, accompanied with attenuated gluconeogenic gene expression and FOXO protein. Altogether, our data suggest that insulin activates the SREBPc- signalling pathway, resulting in FOXO degradation mediated by MDM, which is one of the crucial mechanisms to maintain hepatic glucose homoeostasis. Results is upregulated by feeding and insulin. In the liver, peripheral circadian clock genes are regulated by various nutritional and hormonal changes 37. Given that circadian clock genes are closely associated with energy homoeostasis, we sought to investigate which circadian clock genes could affect feedingdependent hepatic lipid and glucose metabolism. Since the expression of most circadian clock genes oscillates in a time dependent manner, the end points of the fasting and/or refeeding experiment were fixed at ZT 3. On refeeding, the expression of lipogenic genes including SREBPc, FASN and ACC was upregulated in the liver. In accordance with previous reports 384, the expression of gluconeogenic genes such as and was downregulated in the postprandial state (Fig. a, Supplementary Fig. A). Interestingly, the level of hepatic mrna was elevated in refed mice (Fig. a). Moreover, the expression of the protein was markedly increased in the liver of refed mice at ZT 3 (Fig. b, Supplementary Fig. B). Because is one of core circadian clock genes, we investigated the expression of gene at ZT and ZT, which are the peak and trough time point of expression, respectively 3. On refeeding, the expression of many circadian clock genes including, BMAL, CLOCK, PER, PER and Reverbs was significantly changed at ZT, but mostly not at ZT (Supplementary Fig. ). While basal levels of expression were differently regulated at ZT and ZT, refeeding increased mrna and protein levels at ZT, compared with ZT. These results imply that expression would be modulated by nutritional stimuli at certain ZT time points (Supplementary Fig. ). These findings prompted us to test whether insulin might elevate hepatic gene expression. In primary hepatocytes, the level of mrna was increased by insulin, similar to SREBPc mrna (Fig. c). Altogether, these data propose that hepatic expression would be upregulated by feeding and insulin. SREBPc regulates gene expression. To investigate which transcription factors regulate insulin-dependent gene expression, we analysed promoters in several species including monkey, cow, sheep, human, rat and mouse (Supplementary Fig. 3A). In the proximal promoter, there are several sterol regulatory element (SRE) motifs as well as an E-BOX (CANNTG) motif, which is also a target motif for BMAL and CLOCK, the core circadian clock proteins NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
4 NATURE COMMUNICATIONS DOI:.38/ncomms8 ARTICLE a b Lipogenesis SREBPc SREBPa SREBP FASN ACC Mouse liver Fasted Refed psrebp nsrebp c Circadian clock SREBPc FASN PER BMAL CLOCK CRY BMAL CRY Gluconeogenesis Fasted Refed PBS Insulin Figure is stimulated by feeding and exposure to insulin. (a,b) C57BL/6 mice were fasted for 4 h and then refed for h. Both fasted and refed mice were sacrificed at ZT 3. In the liver, levels of the mrna (a) and protein (b) were determined using qrt-pcr with normalization to TBP mrna levels and western blotting, respectively. psrebp, precursor SREBP; nsrebp, nuclear SREBP. Data are represented as mean ±s.d., N ¼ 3 for each group. Po.5, Po. (Student s t-test). (c) gene expression was measured in mouse primary hepatocytes following h of insulin exposure using qrt-pcr. The level of the TBP mrna was used for the qrt-pcr normalization. Data are represented as mean±s.d., N ¼ 3 for each group. Po.5, Po. (Student s t-test). See Supplementary Fig. 3 for original full immunoblot. (Supplementary Fig. 3A). Both SRE and E-BOX motifs are well-known binding targets of SREBPc with its dual DNA binding specificity 4,4. To examine whether SREBPc could regulate gene expression, SREBPc was overexpressed in mouse primary hepatocytes. As shown in Fig. a,b, the levels of hepatic mrna and protein were increased by SREBPc overexpression (Fig. a,b, Supplementary Fig. 4A), implying that SREBPc may be a key transcription factor that upregulates hepatic gene expression in the postprandial state. Next, the effect of ectopic expression of SREBPc on the promoter activity was examined. Expression of luciferase from a wild-type promoter was compared with the expression from a promoter with mutated SRE motifs (3XSRE) in HEK93T cells (Fig. c). We observed substantial loss of promoter activity with loss of the 3XSRE sequences but not E-BOX motif sequences (Fig. c, Supplementary Fig. 3B). In addition, SREBPc binding to the promoter was confirmed by a ChIP assay (Fig. d). Meanwhile, consistent with previous reports 35, hepatic SREBPc reduced and expression (Fig. a). To investigate whether SREBPc might promote gene expression on insulin, primary hepatocytes isolated from SREBPc þ / þ and SREBPc / mice were challenged with insulin. The level of protein in SREBPc þ / þ primary hepatocytes was prominently elevated by insulin whereas that of protein in SREBPc / primary hepatocytes was increased to a lesser extent by insulin (Fig. e). Moreover, in contrast to SREBPc þ / þ mice, refeeding failed to increase hepatic gene expression in SREBPc / mice (Fig. f). These data indicate that SREBPc is a key factor for the upregulation of hepatic gene expression in the postprandial state. SREBPc- pathway inhibits hepatic gluconeogenesis. Consistent with previous reports 5,33,34, SREBPc overexpression decreased glucose production in mouse primary hepatocytes (Fig. 3a). In addition, the optical in vivo imaging analysis revealed that hepatic SREBPc overexpression remarkably repressed the promoter activity of the gene in vivo (Fig. 3b). Accordingly, the expression of and genes was suppressed in the liver of SREBPc overexpressing mice (Supplementary Fig. 4B). These findings led us to investigate the effect of SREBPc on blood glucose level in vivo. Pyruvate tolerance test revealed that the adenoviral overexpression of SREBPc significantly decreased the level of blood glucose following pyruvate injection (Fig. 3c,d). Also, SREBPc / mice showed higher blood glucose than SREBPc þ / þ mice (Fig. 3e,f). Thus, these results suggest that hepatic SREBPc would suppress gluconeogenesis, potentially by modulating gluconeogenic gene expression. To examine whether, a novel target gene of SREBPc, might modulate hepatic gluconeogenic gene expression, we suppressed expression via small interfering RNA (sirna) in rat hepatoma H4IIE cells. Downregulation of increased the expression of and genes (Fig. 3g, Supplementary Fig. 5A), which are crucial for hepatic gluconeogenesis. On the contrary, hepatic overexpression decreased the expression of and genes in mouse primary hepatocytes (Fig. 3h, Supplementary Fig. 5B). To confirm these observations, we measured pyruvate-induced blood glucose level from þ / þ and / mice. As shown in Fig. 3i,j, / mice exhibited higher blood glucose level than þ / þ mice. To test whether would be a key mediator of the inhibition of hepatic gluconeogenesis by SREBPc, glucose production assays were performed in primary hepatocytes from þ / þ and / mice. As indicated in Fig. 3k and Supplementary Fig. 5C, SREBPc overexpression attenuated glucose production in þ / þ primary hepatocytes while SREBPc failed to suppress glucose production in / primary hepatocytes. To establish whether the SREBPc- signalling pathway could indeed repress hepatic glucose production in vivo, was adenovirally overexpressed in the liver of NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8 3
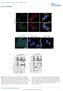
5 NATURE COMMUNICATIONS DOI:.38/ncomms8 a Ad-SREBPc CRY BMAL FASN b 4 4 Ad-SREBPc SREBPc (FLAG) c promoter Luci SRE TSS SRE SRE3 Luci SRE TSS SRE SRE3 MOCK SREBPc Relative luciferase unit (RLU) d Fold enrichment in promoter.5.5 lgg SREBP e 4 SREBPc / SREBPc / Control Insulin Control Insulin nsrebp f SREBPc / Fasted SREBPc SREBPc / Refed SREBPc / SREBPc / Figure SREBPc directly activates gene expression. (a,b) Mouse primary hepatocytes were adenovirally infected with or Ad-SREBPc, as indicated. The levels of the mrna (a) and protein (b) were determined using qrt-pcr with normalization to TBP mrna levels and western blotting, respectively. Data are represented as mean ±s.d., N ¼ 3 for each group. Po.5, Po. (Student s t-test). (c) Luciferase activity of the WT promoter and 3XSRE mutant promoter were measured following co-transfection with expression plasmids encoding either SREBPc or MOCK in HEK93T cells. Luciferase activity was normalized by b-gal activity. TSS, Transcription Start Site; SRE, Sterol Regulatory Elements. Data are represented as mean ±s.d., N ¼ 5 for each group. Po. (Student s t-test). (d) ChIP assay, performed as described in Methods section, showing promoter occupancy by SREBP in H4IIE cells. (e) Mouse primary hepatocytes were isolated from SREBPc / and SREBPc þ / þ mice. With insulin ( nm), the levels of SREBPc and protein were determined using western blotting. (f) SREBPc / and SREBPc þ / þ mice were fasted for 4 h and then refed for h. Both fasted and refed mice were sacrificed at ZT 3. The levels of SREBPc and mrnas were determined by qrt-pcr and normalized to TBP mrna levels. Data are represented as mean ±s.d., N ¼ 34 for each group. Po.5, Po. (Student s t-test). See Supplementary Fig. 3 for original full immunoblot. SREBPc / mice. While SREBPc / mice showed higher blood glucose level than SREBPc þ / þ mice during the pyruvate tolerance test, SREBPc / mice with overexpression decreased blood glucose level (Fig. 3l,m, Supplementary Fig. 6A,B). These data strongly indicate that the SREBPc- signalling pathway could inhibit hepatic gluconeogenesis in vivo. regulates FOXO protein levels. To decipher the underlying mechanism(s) by which insulin-induced could repress hepatic gluconeogenesis, we focused on FOXO, as its regulatory effects on insulin signalling and gluconeogenesis are well established. In mouse primary hepatocytes, the level of FOXO protein was decreased by overexpression (Fig. 4a) while FOXO mrna levels were not altered (Fig. 4b). These data indicated that might modulate the level of FOXO protein, probably, independent of FOXO mrna. Furthermore, the level of FOXO protein was enhanced in suppressed hepatocytes (Fig. 4c,d). In accordance with these in vitro data, the level of FOXO protein was higher in the liver of / mice than in the liver of þ / þ mice, whereas the levels of FOXO mrna were not different between þ / þ and / mice (Fig. 4e,f). Compared with SREBPc þ / þ mice, the levels of FOXO protein and and mrnas were augmented in the liver of SREBPc / mice whereas that of protein was reduced (Fig. 4g, Supplementary Fig. 7). To verify that could inhibit hepatic gluconeogenesis via FOXO, the effects of and/or FOXO suppression on gluconeogenic gene expression were examined. Increased expression of and genes by suppression was abolished when FOXO gene was downregulated by sirna (Fig. 4h), implying that FOXO might be a downstream factor of to regulate gluconeogenesis. These in vivo and in vitro data imply that would alleviate hepatic gluconeogenesis through FOXO. 4 NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
6 NATURE COMMUNICATIONS DOI:.38/ncomms8 ARTICLE a b c d Relative glucose production Blood glucose (mg dl ) Ad-SREBPc Ad-SREBPc Pro-luci PTT SREBPc / ( 4 ) / 3.8 SREBPc Time (min) AUC glucose (PTT) (mg dl X min) Photon density SREBPc / ( 8 ) Ad-SREBPc SREBPc / Relative mrna levels Blood glucose (mg dl ) PTT ( 4 ) Ad-SREBPc Time (min) e f g h sicon si Relative mrna levels AUC glucose (PTT) (mg dl X min) Ad-SREBPc Ad- i j k PTT / ( 4 ) 6 / Time (min) Blood glucose (mg dl ) AUC glucose (PTT) (mg dl X min) / / Relative glucose production / NS ### / Ad-SREBPc l Blood glucose (mg dl ) PTT # ### # ### # # Time (min) SREBPc /, SREBPc /, SREBPc /, Ad- m AUC glucose (PTT) (mg dl X min) ( 4 ) ### SREBPc /, SREBPc /, SREBPc /, Ad- Figure 3 The SREBPc- signalling pathway regulates hepatic glucose production. (a) Mouse primary hepatocytes were infected with or Ad-SREBPc. Relative glucose production was measured using a glucose oxidase (GO) kit as described in Methods. Data represent mean ±s.d., N ¼ 3 for each group. Po.5 (Student s t-test). (b) C57BL/6 mice were infected with Ad--luc and either or Ad-SREBPc. promoter activity was measured by optical in vivo imaging and photon density. (c,d) C57BL/6 mice were infected with or Ad-SREBPc and performed pyruvate tolerance test (c). All mice were fasted at ZT and performed PTT at ZT 3. Results were converted to area-under-the curve (AUC) (d). Data represent mean ±s.d., N ¼ 5 for each group. Po.5, Po. (Student s t-test). (e,f) Pyruvate tolerance test (e) was performed in SREBPc / and SREBPc þ / þ mice. All mice were fasted at ZT and performed PTTat ZT 3. Results were converted to AUC (f). Data represent mean ±s.d., N ¼ 5 for each group. Po.5, Po. (Student s t-test). (g) H4IIE cells were transfected with sicon or si. were determined by qrt-pcr and normalized to cyclophilin mrna levels. Data represent mean ±s.d., N ¼ 3 for each group. Po.5, Po. (Student s t-test). (h) Mouse primary hepatocytes were infected with or Ad-. were determined by qrt-pcr and normalized to TBP mrna levels. Data represent mean ±s.d., N ¼ 3 for each group. Po.5 (Student s t-test). (i,j) Pyruvate tolerance test (i) was performed in / and þ / þ mice. All mice were fasted at ZT and performed PTT at ZT 3. Results were converted to AUC (j). Data represent mean ±s.d., N ¼ 7 for each group. Po.5, Po. (Student s t-test). (k) Mouse primary hepatocytes isolated from þ / þ and / mice were infected with or Ad-SREBPc. Relative glucose production was measured using a glucose oxidase (GO) kit. Data represent mean ±s.d., N ¼ 8 for each group. Po. versus, ### Po. versus þ / þ (Student s t-test). (l,m) Pyruvate tolerance test (l) insrebpc þ / þ mice injected with and in SREBPc / mice injected with either or Ad-. Results were converted to AUC (m). All mice were fasted at ZT and performed PTT at ZT 3. Data represent mean ±s.d., N ¼ 7 for each group. Po., Po. versus SREBPc þ / þ,, # Po.5, ### Po. versus SREBPc /, (Student s t-test). NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8 5
7 NATURE COMMUNICATIONS DOI:.38/ncomms8 a b c 4 d 4 e 4 Ad- Cell lysates sicon si Liver lysates FOXO FOXO / / FOXO f Relative mrna levels Relative mrna levels / / Ad- FOXO FOXO DAPI FOXO Merge g 3 4 sicon sicry Liver lysates SREBPc / SREBPc / psrebp nsrebp FOXO h Relative mrna levels ## sicon si sifoxo si sifoxo # sicon si sifoxo si sifoxo sicon si sifoxo si sifoxo FOXO sicon si sifoxo si sifoxo Figure 4 regulates FOXO protein level. (a,b) Mouse primary hepatocytes were adenovirally infected with or Ad-. The expression profiles of FOXO were analysed at the protein level (a) using western blotting and at the mrna level (b) using qrt-pcr. Data are represented as mean ±s.d., N ¼ 4 for each group. Po.5 (Student s t-test). (c,d) H4IIE cells were transfected with sicon or si. Immunocytochemical analysis (c) of endogenous FOXO. DAPI, 4, 6-diamidino--phenylindole. Scale bars, mm. Endogenous FOXO and protein levels were analysed using western blotting (d). (e,f) The expression patterns of FOXO protein in the liver of þ / þ and / mice were analysed by western blotting (e) and qrt-pcr (f). were determined using qrt-pcr and normalized to the levels of the TBP mrna. Data are represented as mean ±s.d., N ¼ 3 for each group. Po.5, (Student s t-test). (g) Expression of and FOXO proteins in the liver of SREBPc þ / þ and SREBPc / mice was analysed by western blotting. (h) H4IIE cells were co-transfected with si and/or sifoxo. were determined using qrt-pcr and normalized to the cyclophilin mrna level. Data are represented as mean ±s.d., N ¼ 3 for each group. # Po.5, ## Po. versus si, Po.5, Po., Po. versus sicon (Student s t-test). See Supplementary Fig. 3 for original full immunoblot. FOXO protein is decreased by insulin-activated. FOXO translocation from the nucleus to the cytoplasm by AKT is a well-known mechanism by which insulin acutely inhibits hepatic glucose production 43. As insulin upregulates that, in turn, downregulates the FOXO protein (Figs c and 4a), we investigated the time course of these events by examining the expression profiles of FOXO and proteins in insulin-treated primary hepatocytes. As shown in Fig. 5a, phosphorylation of AKT and FOXO was clearly induced in cells treated with insulin for a relatively short periods (4 h). Consistent with previous report 44, the level of FOXO protein was decreased by short-term insulin treatment (B4 h), which would be mediated by FOXO ubiquitination and degradation in cytosol. Furthermore, the phosphorylation levels of AKT and FOXO were gradually and substantially decreased by a longterm incubation (8 h) with insulin. It appears that FOXO translocation from the nucleus to the cytoplasm by AKT might be more pronounced after a short exposure (4 h) to insulin rather than following a long-term insulin treatment (8 h). Intriguingly, in hepatocytes treated with insulin for long periods (8 h), the level of the protein was markedly increased, while that of the total FOXO protein was continuously decreased, implying that the amount of the protein appears to be inversely related to the total quantity of the FOXO protein. Accordingly, the expression of and genes was downregulated after either a long-term (8 h) or a shortterm (4 h) insulin treatment (Fig. 5b). These data suggest that sustainable reduction of the FOXO protein might be involved in the suppression of hepatic gluconeogenesis for long-term insulin action. Next, we explored whether could indeed modulate the FOXO protein in insulin-treated hepatocytes. To address this, we have tested þ / þ and / primary hepatocytes. As shown in Fig. 5c, the level of FOXO protein was decreased in insulin-treated þ / þ primary hepatocytes while that of FOXO protein was not continuously suppressed in insulintreated / primary hepatocytes. In the late phase of insulin action, decreased levels of and mrna were slightly but substantially increased in / primary hepatocytes (Fig. 5d). Moreover, insulin failed to suppress glucose production in long-term (8 h) insulin-treated / primary hepatocytes. (Fig. 5e). Altogether, these data indicate that could repress the expression of hepatic gluconeogenic genes with reduced FOXO protein for the long-term insulin action (8 h). 6 NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
8 NATURE COMMUNICATIONS DOI:.38/ncomms8 ARTICLE a Insulin: h.5 h h 4 h 8 h h pakt AKT pfoxo FOXO b Insulin: 4 h.5 h h 4 h 8 h h h.5 h h 4 h 8 h PBS Insulin h c Insulin: h h 4 h 8 h h h h 4 h 8 h h / / pakt.4..8 AKT.6 pfoxo.4. FOXO Insulin: 4 d Relative mrna leevels h h 4 h 8 h h / / h h 4 h 8 h h e Insulin: Relative glucose production h / / h 4 h 8 h h f Insulin: AKT VIII: pakt AKT pfoxo FOXO Ad- 4 g h i Blood glucose (mg dl ) vehicle MK6 Ad- MK6 PTT ## ## ### ## Time (min) AUC glucose (PTT) (mg dl X min) ( 4 ) vehicle MK6 Ad- MK6 ### FOXO short exposure Long exposure Ad- Vehicle MK6 MK6 4 Figure 5 Long-term insulin treatment stimulates expression and inhibits hepatic gluconeogenesis. (a,b) Mouse primary hepatocytes were treated with nm insulin for different periods. Protein levels (a) were determined with western blotting. The mrna levels (b) were analysed by qrt-pcr. Data are represented as mean ±s.d., N ¼ 3 for each group. Po.5, Po. (Student s t-test). (ce) Mouse primary hepatocytes isolated from þ / þ and / mice were treated with nm insulin for different periods. Protein levels (c) were analysed by western blotting, and relative mrna levels (d) were determined by qrt-pcr and normalized to the TBP mrna level. Data are represented as mean ±s.d., N ¼ 3 for each group. Po.5, Po., Po. versus þ / þ control (Student s t-test). Relative glucose production (e) was measured using a glucose oxidase (GO) kit. Data are represented as mean ±s.d., N ¼ 5 for each group. Po.5, Po., versus þ / þ control (Student s t-test). (f) Mouse primary hepatocytes were infected with and Ad-, and then treated with insulin ( nm) or insulin ( nm) and AKTVIII (5 mm) for h. Protein levels were determined with western blotting. (gi) C57BL/6 mice were infected with or Ad- and subjected to the pyruvate tolerance test (g) with or without the AKT inhibitor MK6. MK6 (3 mg kg ) was given by oral gavage min before the pyruvate tolerance test. All mice were fasted at ZT and performed PTT at ZT 3. Results were converted to AUC values (h). After the pyruvate tolerance test, hepatic protein levels (i) were analysed by western blotting. Data are represented as mean ±s.d., N ¼ 57 for each group. ## Po., ### Po. versus þ vehicle control, Po.5, Po., versus þ MK6 control (Student s t-test). See Supplementary Fig. 3 for original full immunoblot. To test whether enhanced could suppress hepatic gluconeogenesis even in the absence of a short-term insulin action (B4 h), we employed AKT inhibitors. In primary hepatocytes, insulin increased phosphorylation levels of both AKT and FOXO, while a co-treatment with the AKT inhibitor AKTVIII blocked phosphorylation of both proteins, as expected (Fig. 5f). However, in -overexpressing hepatocytes, the total FOXO level was decreased in insulin and/or the AKT inhibitor NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8 7
9 NATURE COMMUNICATIONS DOI:.38/ncomms8 treated cells, implying that could downregulate FOXO protein independent of FOXO phosphorylation (Fig. 5f). To confirm this observation in vivo, we tested another AKT inhibitor, MK6, in mice. As shown in Fig. 5g,h, administration of MK6 significantly increased blood glucose level on pyruvate challenge; however, adenoviral overexpression in mice significantly attenuated blood glucose level even in the presence of MK6. It is noteworthy that the level of FOXO protein was greatly augmented by MK6, whereas elevation suppressed FOXO protein expression in vivo (Fig. 5i). Taken together, these data clearly indicate that -dependent FOXO reduction may contribute to the suppression of hepatic gluconeogenesis independent of AKT activation. stimulates proteasomal degradation of FOXO. Since overexpression decreased the level of the FOXO protein, but not the FOXO mrna, we investigated whether the downregulation of the FOXO protein might proceed via proteasomal degradation. As shown in Fig. 6a, the reduction of the FOXO protein by overexpression was alleviated by MG3 treatment, indicating that the regulation of FOXO protein by may be, at least in part, dependent on proteasomal degradation. When we tested physical interaction between FOXO and proteins, co-immunoprecipitation assays revealed that could associate with FOXO protein (Fig. 6b). Then, we examined whether might induce FOXO degradation via the ubiquitination-proteasome pathway. As shown in Fig. 6c, overexpression dramatically promoted FOXO poly-ubiquitination, implying that could potentiate FOXO degradation, probably, through protein protein interactions. To further explore the subcellular location of FOXO degradation induced by, the levels of the nuclear and cytosolic FOXO protein were investigated. As shown in Fig. 6d, the nuclear fraction of the FOXO protein was decreased by overexpression whereas incubation with MG3 blocked this decrease. At the same time, the levels of cytosolic FOXO were not altered on overexpression, irrespective of the presence of MG3. Consistent with these results, polyubiquitination of the nuclear form of the FOXO mutant protein (nfoxo-myc) was greatly augmented by (Fig. 6e). Therefore, it is plausible that the degradation of the FOXO protein via poly-ubiquitination is stimulated by in the nucleus. is involved in MDM-mediated FOXO degradation. Among several ubiquitin E3 ligases of the FOXO protein 45,46,we found that the MDM ubiquitin E3 ligase appeared to be involved in the -mediated FOXO degradation. As shown in Fig. 7a, MDM suppression markedly rescued the level of the FOXO protein in -overexpressing cells, implying that MDM may participate in the -dependent FOXO reduction. To study the role of in MDM-mediated FOXO degradation, we tested whether might regulate the subcellular localization of MDM. Wild-type and cytosolic (DNLS-) did not change the subcellular location of the nuclear MDM (Supplementary Fig. 8). Instead, we revealed that potentiates the association between FOXO and MDM (Fig. 7b). a FOXO (MYC) Ad- Vehicle MG3 4 b MOCK GFP- FOXO-MYC (GFP) FOXO (MYC) (GFP) FOXO (MYC) 4 IP:MYC Input c d e MOCK FOXO-MYC Ub-HA GFP- Ub-FOXO FOXO (MYC) IP:FOXO(MYC) 3 4 Ad- MG3 FOXO POL II FOXO GAPDH 35 Nuclear Cytosol MOCK nfoxo-myc Ub-HA GFP- Ub-nFOXO nfoxo (MYC) IP:nFOXO(MYC) 3 Figure 6 accelerates ubiquitin-mediated FOXO degradation. (a) Mouse primary hepatocytes were adenovirally infected with or Ad-. The cells were treated with mm MG3 or vehicle for 4 h. Total cell lysates were analysed by western blotting with indicated antibodies. (b) HEK93T cells were transfected with GFP- and/or FOXO-MYC expression vectors. Co-immunoprecipitation with an anti-myc antibody and western blotting were performed with the indicated antibodies. IP, immunoprecipitation. (c) COS- cells were co-transfected with plasmids encoding FOXO-MYC, GFP-, and Ubiquitin-HA. After transfection, the cells were treated with MG3 ( mm) for 6 h and then the cell lysates were subjected to immunoprecipitation with an anti-myc antibody followed by western blotting with indicated antibodies. IP, immunoprecipitation. (d) Mouse primary hepatocytes were infected with or Ad-. After infection, the cells were treated with MG3 ( mm) for 4 h. Nuclear and cytosolic fractions were isolated and analysed by western blotting with indicated antibodies. (e) COS- cells were co-transfected with plasmids encoding nfoxo-myc, GFP-, and Ubiquitin-HA. After transfection, the cells were challenged with MG3 ( mm) for 6 h. The cell lysates were subjected to immunoprecipitation with an anti-myc antibody. IP, immunoprecipitation. See Supplementary Fig. 3 for original full immunoblot. 8 NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
10 NATURE COMMUNICATIONS DOI:.38/ncomms8 ARTICLE a Adeno : sicon simdm MOCK MOCK b MOCK nfoxo-myc FLAG-MDM GFP- FOXO (MYC) MDM 4 MDM (FLAG) (GFP) nfoxo (MYC) MDM (FLAG) (GFP) nfoxo (MYC) 4 IP:MYC Input c MOCK nfoxo-myc Ub-HA FLAG-MDM FLAG- d MOCK nfoxo-myc Ub-HA FLAG-MDM si IP:nFOXO(MYC) Input 3 4 Ub-nFOXO IgG nfoxo(myc) MDM (FLAG) nfoxo(myc) IP:nFOXO(MYC) Input 3 4 Ub-nFOXO IgG nfoxo(myc) MDM (FLAG) nfoxo(myc) Figure 7 is involved in MDM-mediated FOXO ubiquitination. (a) Mouse primary hepatocytes were infected with or Ad- and/or sicon or simdm. Total cell lysates were analysed by western blotting with indicated antibodies. (b) HEK93T cells were transfected with FLAG-MDM, nfoxo-myc, and GFP- expression vectors. Total cell lysates were subjected to co-immunoprecipitation with an anti-myc antibody followed by western blotting with indicated antibodies. IP, immunoprecipitation. (c) COS- cells were co-transfected with plasmids encoding nfoxo-myc, FLAG-MDM, FLAG-, and Ubiquitin-HA. After transfection, the cells were challenged with MG3 ( mm) for 6 h. Cell lysates were subjected to immunoprecipitation with an anti-myc antibody. IP, immunoprecipitation (d) COS- cells were co-transfected with plasmids encoding nfoxo-myc, FLAG-MDM, Ubiquitin-HA, and si. Cells were treated with MG3 ( mm) for 6 h. Cell lysates were subjected to immunoprecipitation with an anti-myc antibody. IP, immunoprecipitation. See Supplementary Fig. 3 for original full immunoblot. In another experiment, we explored whether could modulate MDM-mediated FOXO degradation. As shown in Fig. 7c,d, overexpression promoted MDM-mediated poly-ubiquitination of the nuclear form of FOXO protein (Fig. 7c), whereas suppression attenuated FOXO polyubiquitination by MDM (Fig. 7d). These data indicate that would participate in MDM-induced FOXO degradation and repress FOXO-mediated hepatic glucose production. mitigates hyperglycaemia in diabetic mouse models. Streptozotocin (STZ)-treated insulin-deficient mice elevated blood glucose as well as gluconeogenic programs including FOXO protein levels, accompanied with increased expression of and genes (Fig. 8ac). Since the SREBPc- axis was upregulated by insulin (Figs c,e), we overexpressed in the liver of STZ-treated insulin-deficient mice. As shown in Fig. 8d, overexpression diminished blood glucose levels in STZ-treated mice. In addition, reduced not only the expression of and genes but also the level of FOXO protein (Fig. 8e,f). These data suggest that would be one of important factors to suppress hepatic glucose production on insulin. Intriguingly, in the liver of obese animals such as db/db and diet-induced obesity mice, SREBPc level is elevated while hepatic gluconeogenesis is not repressed 475. To explore which process(es) might be dysregulated in the regulation of hepatic gluconeogenesis, we have examined several mrna and protein levels for SREBPc- axis and gluconeogenic genes in diabetic animals. Similar to previous reports 475, the mrna levels of SREBPc and gluconeogenic genes were elevated in db/db mice (Fig. 8g). However, hepatic protein was substantially decreased and the level of MDM protein was not significantly changed in db/db mice (Fig. 8h). Similarly, diet-induced obesity mice exhibited elevated expression of SREBPc and gluconeogenic genes whereas was not activated (Supplementary Fig. 9A,B). To test the idea that dysregulated protein might mediate hyperglycaemia with enhanced FOXO protein in diabetic animals, was adenovirally overexpressed in the liver of db/db mice. As shown in Fig. 8i, the levels of pyruvate-induced blood glucose were decreased in -overexpressing db/db mice. Moreover, the levels of feeding blood glucose were diminished by overexpression (Fig. 8j). In addition, ectopic expression reduced the levels of FOXO protein as well as gluconeogenic gene expression in db/db NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8 9
11 NATURE COMMUNICATIONS DOI:.38/ncomms8 a b c Blood glucose (mg dl ) Control STZ Control STZ nsrebp FOXO Control STZ FOXO d e f Blood glucose (mg dl ) g Relative mrna levels Blood glucose (mg dl ) STZ, STZ, Ad- AUC glucose (PTT) (mg dl X min) STZ, STZ, Ad SREBPc db/ db/db PTT Time (min) db/ db/db ( 4 ) 7 db/ db/db Blood glucose (mg dl ) FOXO db/ db/db i j k l Ad- Ad- Ad Ad- Ad Ad- Ad STZ Ad- db/ db/db db/db Ad- FOXO nsrebp Figure 8 alleviates gluconeogenesis in diabetic mouse models. (ac) Eight-week-old male mice were injected STZ (5 mg kg ) and sacrificed in ZT 3 after week later. Blood glucose levels (a) were measured in ad libitum at ZT 3. After all of the mice were sacrificed at ZT 3, hepatic protein levels (b) were analysed by western blotting. The mrna levels (c) were determined by qrt-pcr analyses and normalized to the TBP mrna level. Data represent mean ±s.d., N ¼ 3 for each group. Po.5, Po., Po. (Student s t-test). (df) STZ-injected mice were infected with adenovirus encoding MOCK or (adenoviral dose of 9 viral particles per mouse). The blood glucose levels (d) were measured in ad libitum at ZT 3. After all of the mice were sacrificed at ZT 3, the mrna levels (e) were determined by qrt-pcr analyses and normalized to the TBP mrna level. Hepatic protein levels (f) were analysed by western blotting. Data represent mean ±s.d., N ¼ 4 for each group. Po.5, Po. (Student s t-test). (g,h) Ten-week-old male db/ þ and db/db mice were sacrificed in ad libitum at ZT 3. The relative mrna levels of various hepatic genes (g) were determined by qrt-pcr analyses and normalized to the TBP mrna level. Data represent mean ±s.d., N ¼ 4 for each group. Po.5 (Student s t-test). Protein levels (h) were determined with western blotting. (il) Ten-week-old male db/db mice were infected through the tail vein with adenovirus encoding MOCK or (adenoviral dose of viral particles per mouse) and subjected to the pyruvate tolerance test (i). All mice were fasted at ZT and performed PTT at ZT 3. Results were converted to AUC. The blood glucose levels (j) were measured in ad libitum at ZT 3. After all of the mice were sacrificed at ZT 3, hepatic protein levels (k) were analysed by western blotting. The mrna levels (l) were determined by qrt-pcr analyses and normalized to the TBP mrna level. Data represent mean ±s.d., N ¼ 5 for each group. Po.5, Po., Po. (Student s t-test). See Supplementary Fig. 3 for original full immunoblot. h FOXO MDM FOXO NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
12 NATURE COMMUNICATIONS DOI:.38/ncomms8 ARTICLE mice (Fig. 8k,l). These results indicate that could ameliorate hyperglycaemia by repressing the level of FOXO protein in db/db mice. Discussion As a major anabolic hormone, insulin stimulates lipogenesis and represses gluconeogenesis in the liver. Following insulin exposure, lipogenesis is upregulated by SREBPc, and the expression of SREBPc target genes such as FASN, SCD and ELOVL6 is induced,5,5. In contrast, insulin blocks hepatic gluconeogenesis through AKT-mediated phosphorylation of FOXO and PGCa 53,54, both are major regulators of gluconeogenic genes including and. Here we propose that the SREBPc- signalling pathway plays an important role to inhibit hepatic gluconeogenesis under anabolic state. Our data from hepatic gluconeogenic gene expression, in vitro glucose output assays, time kinetics of insulin signalling cascades, and pyruvate tolerance test, which reflects both hepatic glucose output and peripheral glucose disposal, have consistently suggested the idea that maintenance of SREBPc-induced is crucial to prevent unnecessary hepatic gluconeogenesis during insulin action. In this regard, it has been reported that single-nucleotide polymorphisms of SREBPc and genes are associated with hyperglycaemia in human,56. Similar to previous reports 47,48,5, the expression of SREBPc and gluconeogenic genes was increased in the liver of diabetic db/db mice (Fig. 8g,h). Unexpectedly, the level of hepatic protein was downregulated in db/db mice. Moreover, in the liver of db/db mice, diurnal expression of protein level was overall reduced whereas FOXO protein levels were elevated compared to db/ þ mice (Supplementary Fig. A,B). In this work, we have demonstrated that ectopic overexpression of in db/db mice alleviated hepatic gluconeogenesis by reducing FOXO protein (Fig. 8il). Also, we have shown that hepatic could attenuate the level of blood glucose by decreasing FOXO protein, independent of AKT activity (Fig. 5fi). Although it remains to be elucidated how elevated SREBPc fails to increase in the liver of db/db mice, it is very likely that increased FOXO protein results from reduced hepatic protein in db/db mice. It has been reported that seems to suppress hepatic glucose production by interfering with glucocorticoid receptor signalling and the glucagon signalling pathway Insulin interacts with glucocorticoid receptor 34 and the a-subunit of the glucagon receptor 33, which are involved in the regulation of gluconeogenesis. Nonetheless, specific roles of during the nutrient-rich state after exposure to insulin have not been clearly elucidated. To test whether might repress gluconeogenesis by inhibiting glucagon signalling and/or glucocorticoid receptordependent pathway, -overexpressing primary hepatocytes were treated with forskolin, db-camp or dexamethasone to mimic the conditions for glucagon or glucocorticoid stimulation. In primary hepatocytes, partially repressed gluconeogenic gene expression in the presence of forskolin, db-camp or dexamethasone (Supplementary Fig. AC), implying that in addition to the glucagon and glucocorticoid signalling pathways, there may be another signalling cascade regulated by, which could suppress hepatic glucose production. Thus, appears to be involved in multiple regulatory pathways that control hepatic gluconeogenesis in response to various hormones. Activation of FOXO-mediated gluconeogenesis is inhibited by insulin. AKT, a key downstream molecule of the insulin-activated signalling, phosphorylates FOXO, which then is translocated from the nucleus to the cytoplasm through its association with the protein 57. In primary hepatocytes, FOXO phosphorylation was rapidly increased by insulin. However, hepatic gluconeogenic programing is persistently and efficiently suppressed regardless of the decreased FOXO phosphorylation at the late stage of insulin action. Intriguingly, hepatic expression was enhanced at relatively late periods of insulin action (Fig. 5a). Furthermore, in primary hepatocytes, a longterm insulin treatment downregulated FOXO expression, while deficiency rescued FOXO protein levels as well as gluconeogenic gene expression (Fig. 5c,d). It is of interest to note that -overexpressing mice showed a decrease of blood glucose level as well as of FOXO protein when AKT activity was pharmacologically repressed with AKT inhibitor MK6 (Fig. 5gi). Collectively, our in vitro and in vivo data suggest that the -dependent FOXO degradation would be one of the crucial mechanisms in attenuating hepatic gluconeogenesis for the long-term insulin action. Therefore, these observations prompted us to propose that the AKT-mediated FOXO phosphorylation provides an acute response during early insulin response, whereas SREBPc-mediated regulation would be a more durable process leading to the repression of futile hepatic gluconeogenesis throughout the anabolic state (Fig. 9). Insulin Cytosol Proteasomal degradation Ub P P P Ub FOXO O Ub Cytosol Nucleus AKT P P P FOXO O Nucleus SREBPc AKT MDM Ub Ub Ub MDM FOXO Proteasomal degradation Suppression of gluconeogenesis in early phase of insulin action Suppression of gluconeogenesis in late phase of insulin action Figure 9 SREBPc- axis suppresses hepatic gluconeogenesis by promoting FOXO degradation. For long-term insulin action, SREBPc- axis suppresses hepatic gluconeogenesis through nuclear FOXO degradation. During anabolic state, increased accelerates MDM-mediated FOXO degradation and thereby insulin sustainably represses hepatic glucose production. NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
13 NATURE COMMUNICATIONS DOI:.38/ncomms8 As one of the key players for circadian rhythmic modulation, expression is affected by various transcription factors and epigenetic regulation. Although our in vivo and in vitro data suggested that expression was promoted by insulin and SREBPc, mrna expression was not elevated by refeeding at ZT, which is the trough point of oscillation (Supplementary Fig. B). These data propose that increased SREBPc might not be sufficient to activate at ZT. Conversely, the levels of mrna and proteins were robustly increased by refeeding at ZT, which is the peak point of its oscillation, implying that SREBPc would augment expression, probably, accompanied with other circadian regulatory factors. Nonetheless, it remains to be elucidated whether as well as other circadian clock genes may contribute to modulate hepatic gluconeogenesis at ZT. On the other hand, we asked the question whether SREBPc deficiency would change hepatic circadian clock gene oscillation. As shown in Supplementary Fig., hepatic circadian clock gene oscillations were not significantly different in SREBPc þ / þ and SREBPc / mice. Of course, we cannot exclude the possibility that SREBPc-induced might contribute to minor roles for hepatic circadian oscillation in SREBPc / mice because it has been reported that / mice exhibit fewer changes in circadian oscillations, compared with / CRY / double-mutant mice 58. Furthermore, it is also possible that remaining SREBPa and/or SREBP activity in SREBPc / mice might maintain intact circadian clock gene oscillations and this homoeostatic regulation needs to be addressed in future studies,59. Nonetheless, hepatic gene expression is clearly upregulated by feeding/insulin-mediated SREBPc. Consistent with previous reports 3, hepatic was increased at night time (ZT 4) when feeding behaviour dominantly occurs in nocturnal animals (Supplementary Fig. A). Although SREBPc / mice did not show any significant difference of circadian clock oscillation (Supplementary Fig. ), SREBPc expression was also increased at night time (ZT 4; Supplementary Fig. ). Unlike the expression profiles of SREBPc and, the level of FOXO protein appeared to be reduced at night time, implying that circadian oscillations of SREBPc and may contribute to repress diurnal glucose production by the regulation of FOXO. As SREBPc could simultaneously regulate both gluconeogenesis and lipogenesis, it is plausible to speculate that hepatic SREBPc would effectively coordinate the anabolic pathways by upregulating fatty acid metabolism and downregulating glucose metabolism on insulin signalling with different target genes. Our study is the first report to reveal the role of SREBPc in activation, which would be crucial in the regulation of hepatic glucose metabolism in the anabolic state. On the basis of the circadian oscillatory gene expression profile in SREBPc / mice, it appears that SREBPc does not actively govern the hepatic circadian clock. Rather, increased expression of hepatic during the postprandial state is primarily regulated, in a clock-gated manner, by insulin-activated SREBPc, which eventually leads to the suppression of glucose production via FOXO degradation (Fig. 9). Although the roles of hepatic in energy metabolism need to be investigated further, our data provide an important clue to understand the molecular mechanisms that link hepatic SREBPc and glucose homoeostasis in physiological and pathological conditions. Methods Animals. C57BL/6 male mice were purchased from SAMTACO (Seoul, South Korea) and housed in colony cages. db/ þ and db/db male mice were obtained from Central Lab (Seoul, Korea). SREBPc / mice were generously provided from Dr J. Horton at the University of Texas Southwestern Medical Center and bred in isolated cages. All animals were maintained under h light/ h dark cycle in a pathogen-free animal facility. Following dissection, mouse tissue specimens were immediately stored at 8 C until further analysis. All experiments with mice were approved by the Seoul National University Institutional Animal Care and Use Committee (SNUIACUC) and the Institutional Animal Care and Use Committee at the University of North Carolina. In vivo imaging system. Ten-week-old C57BL/6 male mice were injected with adenoviruses encoding green fluorescent protein (GFP) (), SREBPc (Ad-SREBPc) and luciferase (Ad--luc) through the tail vein. After 5 days, adenovirus-infected mice were injected intraperitoneally with mg kg sterile firefly D-luciferin. After 5 min, mice were anaesthetised and imaged using an IVIS imaging system (Xenogen, Alameda, CA, USA) as described 6. Pyruvate tolerance tests. For the pyruvate tolerance test, mice were fasted for 6 h and then injected intraperitoneally with pyruvate ( g kg body weight for mice). Blood glucose levels were measured in tail vein blood samples at the indicated time points by using a Free-Style blood glucose metre (TheraSense, Sweden). Materials and plasmids. MG3 was purchased from Calbiochem (San Diego, CA, USA). MK6 was purchased from Selleckchem (S78). AKTVIII was purchased from Santa Cruz Biotechnology (sc-48). Antibodies against MYC (Cell Signalling, 76, :, dilution), HA (Cell Signalling, 374, :, dilution), FOXO (Cell Signalling, 88, :, dilution), phosphor-foxo- Ser56 (Cell Signalling, 946, :, dilution), AKT (Cell Signalling, 97, :, dilution), and phosphor-akt-ser473 (Cell Signalling, 97, :, dilution), FLAG (Sigma-Aldrich, F365, :, dilution), (Sigma-Aldrich, A536, :, dilution), (Santa Cruz Biotechnology, sc-33839, :5 dilution), POLII (Santa Cruz Biotechnology, sc-899, :, dilution), GFP (Santa Cruz Biotechnology, sc-9996, :, dilution), GAPDH (LabFrontier, Co., LF-PA8, :, dilution), SREBP (BD Bioscience, 36, :, dilution), MDM (Abcam, ab6895, :, dilution) and (lab made antibody from Dr Aziz Sancar 3, : dilution) were used. GFP- was cloned into the pegfp-n vector and FLAG-MDM was cloned into pcmv-3 FLAG. Mouse promoter was cloned into the pgl3-basic vector. Cell-based ubiquitination assays. COS- cells (ATCC, CRL65) were transfected with plasmids encoding FOXO WT-MYC, nfoxo (ADA)-MYC, GFP- (or FLAG-), FLAG-MDM and Ubiquitin-HA in the presence of mm MG3 for 4 h. Total cell lysates were prepared using the TGN buffer. FOXO WT-MYC and nfoxo (ADA)-MYC were immunoprecipitated with an anti-myc antibody, and after washing in the TGN buffer, proteins were separated by SDSPAGE followed by western blotting analyses with an anti-ha antibody. ChIP assays. Cross-linking and chromatin immunoprecipitation assays with H4IIE cells (ATCC, CRL548) were performed as described previously 6. Extracted proteins from total cell lysates were immunoprecipitated with anti-srebp (BD Bioscience) or IgG (Santa Cruz) for h. Precipitated DNA fragments were analysed by qrt-pcr using primer sets that encompassed the proximal ( to þ base pairs) region of the rat promoter and negative control ( þ 96 to þ 989 base pairs) region. The sequences of ChIP assay primers were as follows: sense, 5 -GTCCGAGCCAGCGTAGTAAA-3, antisense, 5 -GGATAGCGCGGG CTAGAG-3 ; negative control primer sense, 5 -CCAGCCACTTTGCTGAAGTT-3 and antisense, 5 -CTAGACAAGGCTGCCCACTC-3. Preparation of recombinant adenovirus. The adenovirus plasmid was constructed as previously described 6.RatSREBPc and mouse cdnas were incorporated into the AdTrack-CMV shuttle vector and a recombinant vector was generated using the Ad-Easy adenoviral vector system. Adenoviruses were amplified in HEK93A cells and isolated by caesium chloride density centrifugation. The GFP was co-expressed from an independent promoter with inserted cdna. For in vivo experiments, mice were injected with 5 9 p.f.u. of adenovirus (db/db mice, p.f.u.) in ml PBS through the tail vein. Empty virus expressing only the gene for GFP served as the control (MOCK). Mouse primary hepatocytes cultures. Mouse primary hepatocytes were isolated from -week-old male mice using collagenase perfusion method 63 and plated with medium 99 (Invitrogen, Carlsbad, CA, USA). After 6 h of attachment, cells were infected with adenovirus or transfected with sirna. For adenoviral infection, isolated hepatocytes were incubated for 4 h with adenovirus at PFU per cell with the serum-free medium, which was subsequently replaced by the % FBS-containing M99 medium. Cell lysis and immunoprecipitation. After washing in cold PBS, cells were treated either with the TGN buffer (5 mm Tris-HCl, ph 7.5, 5 mm NaCl, % Tween- NATURE COMMUNICATIONS 7:8 DOI:.38/ncomms8
ACC ELOVL MCAD. CPT1α 1.5 *** 0.5. Reverbα *** *** 0.5. Fasted. Refed
 Supplementary Figure A 8 SREBPc 6 5 FASN ELOVL6.5.5.5 ACC.5.5 CLOCK.5.5 CRY.5.5 PPARα.5.5 ACSL CPTα.5.5.5.5 MCAD.5.5 PEPCK.5.5 G6Pase 5.5.5.5 BMAL.5.5 Reverbα.5.5 Reverbβ.5.5 PER.5.5 PER B Fasted Refed
Supplementary Figure A 8 SREBPc 6 5 FASN ELOVL6.5.5.5 ACC.5.5 CLOCK.5.5 CRY.5.5 PPARα.5.5 ACSL CPTα.5.5.5.5 MCAD.5.5 PEPCK.5.5 G6Pase 5.5.5.5 BMAL.5.5 Reverbα.5.5 Reverbβ.5.5 PER.5.5 PER B Fasted Refed
Supplementary Table 1. Metabolic parameters in GFP and OGT-treated mice
 Supplementary Table 1. Metabolic parameters in GFP and OGT-treated mice Fasted Refed GFP OGT GFP OGT Liver G6P (mmol/g) 0.03±0.01 0.04±0.02 0.60±0.04 0.42±0.10 A TGs (mg/g of liver) 20.08±5.17 16.29±0.8
Supplementary Table 1. Metabolic parameters in GFP and OGT-treated mice Fasted Refed GFP OGT GFP OGT Liver G6P (mmol/g) 0.03±0.01 0.04±0.02 0.60±0.04 0.42±0.10 A TGs (mg/g of liver) 20.08±5.17 16.29±0.8
Supplementary Information
 Supplementary Information Akt regulates hepatic metabolism by suppressing a Foxo1 dependent global inhibition of adaptation to nutrient intake Mingjian Lu 1, Min Wan 1, Karla F. Leavens 1, Qingwei Chu
Supplementary Information Akt regulates hepatic metabolism by suppressing a Foxo1 dependent global inhibition of adaptation to nutrient intake Mingjian Lu 1, Min Wan 1, Karla F. Leavens 1, Qingwei Chu
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat. day, cells were transduced with adenoviruses expressing GFP, MKP-3 or shgfp,
 SUPPLEMENTAL METHODS Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat hepatocytes Primary rat hepatocytes were seeded as described in experimental procedures. The next day, cells
SUPPLEMENTAL METHODS Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat hepatocytes Primary rat hepatocytes were seeded as described in experimental procedures. The next day, cells
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA. 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT. Acc1 AGCAGATCCGCAGCTTG ACCTCTGCTCGCTGAGTGC
 Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
Supplementary Table 1. Quantitative PCR primer sequences Gene symbol Sequences (5 to 3 ) Forward Reverse 18s AAACGGCTACCACATCCAAG CCTCCAATGGATCCTCGTTA 36b4 GTTCTTGCCCATCAGCACC AGATGCAGCAGATCCGCAT Acc1
AAV-TBGp-Cre treatment resulted in hepatocyte-specific GH receptor gene recombination
 AAV-TBGp-Cre treatment resulted in hepatocyte-specific GH receptor gene recombination Supplementary Figure 1. Generation of the adult-onset, liver-specific GH receptor knock-down (alivghrkd, Kd) mouse
AAV-TBGp-Cre treatment resulted in hepatocyte-specific GH receptor gene recombination Supplementary Figure 1. Generation of the adult-onset, liver-specific GH receptor knock-down (alivghrkd, Kd) mouse
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
CIRCADIAN SIGNALING NETWORKS
 Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P. Matthias and RG Clerc Roger G. Clerc 07.05.2014 CIRCADIAN SIGNALING NETWORKS Master pacemaker SCN «Slave clocks»
Transcription Regulation And Gene Expression in Eukaryotes Cycle G2 (lecture 13709) FS 2014 P. Matthias and RG Clerc Roger G. Clerc 07.05.2014 CIRCADIAN SIGNALING NETWORKS Master pacemaker SCN «Slave clocks»
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
Salt-inducible kinase 2 links transcriptional coactivator p300 phosphorylation to the prevention of ChREBP-dependent hepatic steatosis in mice
 Salt-inducible kinase 2 links transcriptional coactivator p300 phosphorylation to the prevention of ChREBP-dependent hepatic steatosis in mice Julien Bricambert,, Catherine Postic, Renaud Dentin J Clin
Salt-inducible kinase 2 links transcriptional coactivator p300 phosphorylation to the prevention of ChREBP-dependent hepatic steatosis in mice Julien Bricambert,, Catherine Postic, Renaud Dentin J Clin
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Predictive PP1Ca binding region in BIG3 : 1,228 1,232aa (-KAVSF-) HEK293T cells *** *** *** KPL-3C cells - E E2 treatment time (h)
 Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna
 Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna targeted for hamster PAQR3. At 24 h after the transfection,
Supplementary Figure 1. PAQR3 knockdown inhibits SREBP-2 processing in CHO-7 cells CHO-7 cells were transfected with control sirna or a sirna targeted for hamster PAQR3. At 24 h after the transfection,
SUPPLEMENTAL FIGURE LEGENDS
 SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1: Endogenous interaction between RNF2 and H2AX: Whole cell extracts from 293T were subjected to immunoprecipitation with anti-rnf2 or anti-γ-h2ax antibodies
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh-
 1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
Sterol regulatory element binding proteins
 STEATOHEPATITIS/METABOLIC LIVER DISEASE Ring Finger Protein20 Regulates Hepatic Lipid Metabolism Through Protein Kinase A-Dependent Sterol Regulatory Element Binding Protein1c Degradation Jae Ho Lee, 1
STEATOHEPATITIS/METABOLIC LIVER DISEASE Ring Finger Protein20 Regulates Hepatic Lipid Metabolism Through Protein Kinase A-Dependent Sterol Regulatory Element Binding Protein1c Degradation Jae Ho Lee, 1
Supplementary Information
 Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
Supplementary Figure 1
 Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original
 Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
KSRP is critical in governing hepatic lipid metabolism
 KSRP is critical in governing hepatic lipid metabolism through controlling Per2 expression Chu-Fang Chou, 1, * Xiaolin Zhu, 1, Yi-Yu Lin, * Karen L. Gamble, W. Timothy Garvey, and Ching-Yi Chen 2, * Department
KSRP is critical in governing hepatic lipid metabolism through controlling Per2 expression Chu-Fang Chou, 1, * Xiaolin Zhu, 1, Yi-Yu Lin, * Karen L. Gamble, W. Timothy Garvey, and Ching-Yi Chen 2, * Department
(A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939 (5 µm)
 Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies
 Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
Supplementary Information. Glycogen shortage during fasting triggers liver-brain-adipose. neurocircuitry to facilitate fat utilization
 Supplementary Information Glycogen shortage during fasting triggers liver-brain-adipose neurocircuitry to facilitate fat utilization Supplementary Figure S1. Liver-Brain-Adipose neurocircuitry Starvation
Supplementary Information Glycogen shortage during fasting triggers liver-brain-adipose neurocircuitry to facilitate fat utilization Supplementary Figure S1. Liver-Brain-Adipose neurocircuitry Starvation
Eukaryotic transcription (III)
 Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
Eukaryotic transcription (III) 1. Chromosome and chromatin structure Chromatin, chromatid, and chromosome chromatin Genomes exist as chromatins before or after cell division (interphase) but as chromatids
SREBPs suppress IRS-2-mediated insulin signalling in the liver
 SREBPs suppress -mediated insulin signalling in the liver Tomohiro Ide 1,Hitoshi Shimano 2,,Naoya Yahagi 2,Takashi Matsuzaka 1,Masanori Nakakuki 1, Takashi Yamamoto 1,Yoshimi Nakagawa 2,Akimitsu Takahashi
SREBPs suppress -mediated insulin signalling in the liver Tomohiro Ide 1,Hitoshi Shimano 2,,Naoya Yahagi 2,Takashi Matsuzaka 1,Masanori Nakakuki 1, Takashi Yamamoto 1,Yoshimi Nakagawa 2,Akimitsu Takahashi
Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance
 Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Cell Metabolism, Volume 11 Supplemental Information Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance Ling Yang, Ping Li, Suneng Fu, Ediz S. Calay, and Gökhan S. Hotamisligil
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
The role of TXNIP in the hepatic glucose homeostasis
 The role of TXNIP in the hepatic glucose homeostasis Seong Ho Jo Department of Medical Science The Graduate School, Yonsei University 1 The role of TXNIP in the hepatic glucose homeostasis Directed by
The role of TXNIP in the hepatic glucose homeostasis Seong Ho Jo Department of Medical Science The Graduate School, Yonsei University 1 The role of TXNIP in the hepatic glucose homeostasis Directed by
Supplementary Materials
 Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
Supplementary Materials Supplementary Figure S1 Regulation of Ubl4A stability by its assembly partner A, The translation rate of Ubl4A is not affected in the absence of Bag6. Control, Bag6 and Ubl4A CRISPR
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
Supplementary Figure 1
 VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
Requires Signaling though Akt2 Independent of the. Transcription Factors FoxA2, FoxO1, and SREBP1c
 Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
supplementary information
 DOI: 1.138/ncb1 Control Atg7 / NAC 1 1 1 1 (mm) Control Atg7 / NAC 1 1 1 1 (mm) Lamin B Gstm1 Figure S1 Neither the translocation of into the nucleus nor the induction of antioxidant proteins in autophagydeficient
DOI: 1.138/ncb1 Control Atg7 / NAC 1 1 1 1 (mm) Control Atg7 / NAC 1 1 1 1 (mm) Lamin B Gstm1 Figure S1 Neither the translocation of into the nucleus nor the induction of antioxidant proteins in autophagydeficient
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary Information. Cryptochrome Mediates Circadian Regulation of camp. Signalling and Hepatic Gluconeogenesis
 Supplementary Information Cryptochrome Mediates Circadian Regulation of camp Signalling and Hepatic Gluconeogenesis Eric E. Zhang 1,2*, Yi Liu 3,5*, Renaud Dentin 3, Pagkapol Y. Pongsawakul 1, Andrew C.
Supplementary Information Cryptochrome Mediates Circadian Regulation of camp Signalling and Hepatic Gluconeogenesis Eric E. Zhang 1,2*, Yi Liu 3,5*, Renaud Dentin 3, Pagkapol Y. Pongsawakul 1, Andrew C.
Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients.
 Supplementary Materials Supplementary Figures Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients. Figure S2. Expression level of podocyte
Supplementary Materials Supplementary Figures Figure S1. Reduction in glomerular mir-146a levels correlate with progression to higher albuminuria in diabetic patients. Figure S2. Expression level of podocyte
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
Supplementary Figure 1
 Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used in qpcr
 Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
m 6 A mrna methylation regulates AKT activity to promote the proliferation and tumorigenicity of endometrial cancer
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
Gene Polymorphisms and Carbohydrate Diets. James M. Ntambi Ph.D
 Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Figure S1. Clinical significance of ZNF322A overexpression in Caucasian lung cancer patients. (A) Representative immunohistochemistry images of ZNF322A protein expression in tissue
SUPPLEMENTARY FIGURES Figure S1. Clinical significance of ZNF322A overexpression in Caucasian lung cancer patients. (A) Representative immunohistochemistry images of ZNF322A protein expression in tissue
FOR REVIEW. BMB Reports - Manuscript Submission. Manuscript Draft. Manuscript Number: BMB
 BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
BMB Reports - Manuscript Submission Manuscript Draft Manuscript Number: BMB-18-095 Title: Insulin Receptor Substrate 2:A Bridge between Hippo and AKT Pathways Article Type: Perspective (Invited Only) Keywords:
Supplementary Figure 1.
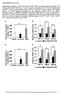 Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Effect of BI-1 on insulin resistance through regulation of CYP2E1
 Effect of BI-1 on insulin resistance through regulation of CYP2E1 Geum-Hwa Lee 1, Kyoung-Jin Oh 2, 3, Hyung-Ryong Kim 4, Hye-Sook Han 2, Hwa-Young Lee 1, Keun-Gyu Park 5, Ki-Hoan Nam 6, Seung-Hoi Koo 2
Effect of BI-1 on insulin resistance through regulation of CYP2E1 Geum-Hwa Lee 1, Kyoung-Jin Oh 2, 3, Hyung-Ryong Kim 4, Hye-Sook Han 2, Hwa-Young Lee 1, Keun-Gyu Park 5, Ki-Hoan Nam 6, Seung-Hoi Koo 2
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
SUPPLEMENTARY INFORMATION
 Glucose is a ligand for the oxysterol receptor LXR Supplementary Figure 1 Schematic representation of pathways influencing glucose fate in the liver. Glucose induces insulin secretion, suppressing hepatic
Glucose is a ligand for the oxysterol receptor LXR Supplementary Figure 1 Schematic representation of pathways influencing glucose fate in the liver. Glucose induces insulin secretion, suppressing hepatic
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Functional characterisation of hepatitis B viral X protein/microrna-21 interaction in HBVassociated hepatocellular carcinoma
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Functional characterisation of hepatitis B viral X protein/microrna-21 interaction in HBVassociated hepatocellular carcinoma CH Li, SC Chow, DL Yin,
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Functional characterisation of hepatitis B viral X protein/microrna-21 interaction in HBVassociated hepatocellular carcinoma CH Li, SC Chow, DL Yin,
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Supplementary Figure 1 Validation of Per2 deletion in neuronal cells in N Per2 -/- mice. (a) Western blot from liver extracts of mice held under ad
 Supplementary Figure 1 Validation of Per2 deletion in neuronal cells in N Per2 -/- mice. (a) Western blot from liver extracts of mice held under ad libitum conditions detecting PER2 protein in brain and
Supplementary Figure 1 Validation of Per2 deletion in neuronal cells in N Per2 -/- mice. (a) Western blot from liver extracts of mice held under ad libitum conditions detecting PER2 protein in brain and
Supplementary Information for
 Supplementary Information for Involvement of urinary bladder Connexin43 and the circadian clock in the coordination of diurnal micturition rhythm Hiromitsu Negoro, 1,2 Akihiro Kanematsu, 1,3 Masao Doi,
Supplementary Information for Involvement of urinary bladder Connexin43 and the circadian clock in the coordination of diurnal micturition rhythm Hiromitsu Negoro, 1,2 Akihiro Kanematsu, 1,3 Masao Doi,
Expanded View Figures
 Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
SUPPLEMENTARY FIGURES AND TABLE
 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Characterization of IRE1α mutants. A. U87-LUC cells were transduced with the lentiviral vector containing the GFP sequence (U87-LUC Tet-ON GFP).
SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Characterization of IRE1α mutants. A. U87-LUC cells were transduced with the lentiviral vector containing the GFP sequence (U87-LUC Tet-ON GFP).
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table
 Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table Title of file for HTML: Peer Review File Description: Innate Scavenger Receptor-A regulates
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table Title of file for HTML: Peer Review File Description: Innate Scavenger Receptor-A regulates
AN ABSTRACT OF THE DISSERTATION OF. Sasmita Tripathy for the Doctor of Philosophy in Nutrition presented on July 11, 2012.
 AN ABSTRACT OF THE DISSERTATION OF Sasmita Tripathy for the Doctor of Philosophy in Nutrition presented on July 11, 2012. Title: Polyunsaturated Fatty Acid Synthesis and Type 2 Diabetes Complications Abstract
AN ABSTRACT OF THE DISSERTATION OF Sasmita Tripathy for the Doctor of Philosophy in Nutrition presented on July 11, 2012. Title: Polyunsaturated Fatty Acid Synthesis and Type 2 Diabetes Complications Abstract
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
ab SREBP-2 Translocation Assay Kit (Cell-Based)
 ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Supplemental Data. TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim.
 Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
A Central Role of MG53 in Metabolic Syndrome. and Type-2 Diabetes
 A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
Loss of Calreticulin Uncovers a Critical Role for Calcium in Regulating Cellular Lipid Homeostasis
 SUPPLEMENTARY MATERIAL Loss of Calreticulin Uncovers a Critical Role for Calcium in Regulating Cellular Lipid Homeostasis Wen-An Wang 1, Wen-Xin Liu 1, Serpen Durnaoglu 2, Sun-Kyung Lee 2, Jihong Lian
SUPPLEMENTARY MATERIAL Loss of Calreticulin Uncovers a Critical Role for Calcium in Regulating Cellular Lipid Homeostasis Wen-An Wang 1, Wen-Xin Liu 1, Serpen Durnaoglu 2, Sun-Kyung Lee 2, Jihong Lian
Hepatic insulin signaling regulates VLDL secretion and atherogenesis in mice
 Hepatic insulin signaling regulates VLDL secretion and atherogenesis in mice Seongah Han,, Domenico Accili, Alan R. Tall J Clin Invest. 2009;119(4):1029-1041. https://doi.org/10.1172/jci36523. Research
Hepatic insulin signaling regulates VLDL secretion and atherogenesis in mice Seongah Han,, Domenico Accili, Alan R. Tall J Clin Invest. 2009;119(4):1029-1041. https://doi.org/10.1172/jci36523. Research
control kda ATGL ATGLi HSL 82 GAPDH * ** *** WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi iwat gwat ibat
 body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
Nature Genetics: doi: /ng.3731
 Supplementary Figure 1 Circadian profiles of Adarb1 transcript and ADARB1 protein in mouse tissues. (a) Overlap of rhythmic transcripts identified in the previous transcriptome analyses. The mouse liver
Supplementary Figure 1 Circadian profiles of Adarb1 transcript and ADARB1 protein in mouse tissues. (a) Overlap of rhythmic transcripts identified in the previous transcriptome analyses. The mouse liver
Molecular Medicine. Angiotensin II Induces Skeletal Muscle Atrophy by Activating TFEB-Mediated MuRF1 Expression
 Molecular Medicine Angiotensin II Induces Skeletal Muscle Atrophy by Activating TFEB-Mediated MuRF1 Expression Philipp Du Bois, Cristina Pablo Tortola, Doerte Lodka, Melanie Kny, Franziska Schmidt, Kunhua
Molecular Medicine Angiotensin II Induces Skeletal Muscle Atrophy by Activating TFEB-Mediated MuRF1 Expression Philipp Du Bois, Cristina Pablo Tortola, Doerte Lodka, Melanie Kny, Franziska Schmidt, Kunhua
(A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and a
 Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
Supplementary figure legends Supplementary Figure 1. Expression of Shh signaling components in a panel of gastric cancer. (A) RT-PCR for components of the Shh/Gli pathway in normal fetus cell (MRC-5) and
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643
 Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth.
 Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information. Induction of p53-independent apoptosis by ectopic expression of HOXA5
 Supplementary Information Induction of p53-independent apoptosis by ectopic expression of in human liposarcomas Dhong Hyun Lee 1, *, Charles Forscher 1, Dolores Di Vizio 2, 3, and H. Phillip Koeffler 1,
Supplementary Information Induction of p53-independent apoptosis by ectopic expression of in human liposarcomas Dhong Hyun Lee 1, *, Charles Forscher 1, Dolores Di Vizio 2, 3, and H. Phillip Koeffler 1,
Supplement Figure S1. Real Time PCR analysis of mrna levels of C/EBPα and PU.1 in wild type (WT) and NQO1-null (NQO1-/-) mice.
 competes with 20S proteasome for binding with C/EBP leading to its stabilization and Relative mrna levels Supplement Figure S1. Real Time PCR analysis of mrna levels of C/EBPα and PU.1 in wild type (WT)
competes with 20S proteasome for binding with C/EBP leading to its stabilization and Relative mrna levels Supplement Figure S1. Real Time PCR analysis of mrna levels of C/EBPα and PU.1 in wild type (WT)
FH- FH+ DM. 52 Volunteers. Oral & IV Glucose Tolerance Test Hyperinsulinemic Euglycemic Clamp in Non-DM Subjects ACADSB MYSM1. Mouse Skeletal Muscle
 A 52 Volunteers B 6 5 4 3 2 FH- FH+ DM 1 Oral & IV Glucose Tolerance Test Hyperinsulinemic Euglycemic Clamp in Non-DM Subjects ZYX EGR2 NR4A1 SRF target TPM1 ACADSB MYSM1 Non SRF target FH- FH+ DM2 C SRF
A 52 Volunteers B 6 5 4 3 2 FH- FH+ DM 1 Oral & IV Glucose Tolerance Test Hyperinsulinemic Euglycemic Clamp in Non-DM Subjects ZYX EGR2 NR4A1 SRF target TPM1 ACADSB MYSM1 Non SRF target FH- FH+ DM2 C SRF
TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer
 Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Information TRAF6 ubiquitinates TGFβ type I receptor to promote its cleavage and nuclear translocation in cancer Yabing Mu, Reshma Sundar, Noopur Thakur, Maria Ekman, Shyam Kumar Gudey, Mariya
Supplementary Figure 1. Expression of the inducible tper2 is proportional to Dox/Tet concentration in Rosa-DTG/Per2 Per2-luc/wt MEFs.
 Supplementary Figure 1. Expression of the inducible tper2 is proportional to Dox/Tet concentration in Rosa-DTG/Per2 Per2-luc/wt MEFs. (a) Dose-responsive expression of tper2 by Dox. Note that there are
Supplementary Figure 1. Expression of the inducible tper2 is proportional to Dox/Tet concentration in Rosa-DTG/Per2 Per2-luc/wt MEFs. (a) Dose-responsive expression of tper2 by Dox. Note that there are
