Butyrate attenuates lipolysis in adipocytes co-cultured with macrophages through non-prostaglandin E2 mediated and prostaglandin E2 mediated pathways
|
|
|
- Laura Mason
- 5 years ago
- Views:
Transcription
1 Ohira et al. Lipids in Health and Disease (216) 15:213 DOI /s RESERCH Open ccess utyrate attenuates lipolysis in adipocytes co-cultured with macrophages through non-prostaglandin E2 mediated and prostaglandin E2 mediated pathways Hideo Ohira, Wao Tsutsui, Rie Mamoto, Sayaka Yamaguchi, Masako Nishida, Miki Ito and Yoshio Fujioka bstract ackground: Interactions between adipocytes and macrophages are associated with metabolic disorders. Production of pro-inflammatory mediators and the release of free fatty acids (FFs) increase when these cells are co-cultured; butyrate significantly diminishes these effects by suppressing both the macrophage inflammatory and adipocyte lipolysis pathways. utyrate is known to up-regulate the expression of prostaglandin E2 (PGE2). Therefore, we hypothesized that PGE2 is associated with the suppression of lipolysis by butyrate in co-culture. Methods: Using contact or transwell co-culture methods with differentiated -L1 adipocytes and RW264.7 macrophages, we investigated the effects of butyrate on the release of PGE2 into the medium and on lipolysis in adipocytes. To elucidate the underlying mechanism, we examined the effects of butyrate on cyclooxygenase-2 (COX2) and phospholipase 2 (PL2) in co-cultured cells, and cyclic adenine monophosphate (cmp) and protein kinase type 1-α regulatory subunit (PRKR1) in co-cultured adipocytes. Silent interfering (si)rn targeting of G-protein coupled receptor (GPR)41 and 19 was employed to examine the effect on lipolysis in TNF-α stimulated adipocytes. Results: Co-culture increased PGE2 release into the medium, compared with cells cultured separately. utyrate significantly increased PGE2 production. Co-culture elevated COX2 expression in macrophages and adipocytes, and butyrate further enhanced this effect. Co-culture enhanced cytosolic PL2 activity in macrophages, which was further enhanced by butyrate. s for lipolysis, co-culture increased the release of FFs and free glycerol into the medium, whereas butyrate (and to a lesser extent, PGE2) suppressed FFs and free glycerol release. n inhibition study using a prostaglandin E receptor 3 selective antagonist suggested that approximately 4% of the suppressive effect of butyrate depends on the PGE2-mediated pathway, whereas 6% depends on a non-pge2 mediated pathway. Co-culture increased cmp and PRKR1 levels in adipocytes, whereas butyrate restored the levels to those of the control. Similarly, in TNF-α stimulated adipocytes, butyrate reduced FFs and free glycerol release. sirn inhibition of GPR41 and GPR19 suggested that the GPR19-mediated pathway predominates, but the GPR41-mediated pathway also regulates the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells. Conclusions: utyrate attenuates lipolysis in adipocytes co-cultured with macrophages via non-pge2 mediated and PGE2-mediated pathways. Keywords: utyrate, dipocyte, Macrophage, Lipolysis, Prostaglandin E2 Correspondence: oohira@nutr.kobegakuin.ac.jp Faculty of Nutrition, Kobe Gakuin University, 518 rise, Ikawadani-cho, Nishi-ku, Kobe , Japan The uthor(s). 216 Open ccess This article is distributed under the terms of the Creative Commons ttribution 4. International License ( which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver ( applies to the data made available in this article, unless otherwise stated.
2 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 2 of 17 ackground Recent studies have demonstrated a strong relationship between obesity (particularly visceral fat accumulation) and the development of cardiovascular diseases [1, 2]. Products derived from interactions between adipocytes and macrophages that may cause metabolic disorders leading to the formation of atherosclerotic lesions include cytokines, chemokines, and adipokines [3 5]. Suganami et al. proposed that the paracrine loop involving free fatty acids (FFs) derived from adipocytes and monocyte chemoattractant protein-1 (MCP-1) and tumor necrosis factor-α (TNF-α) produced by macrophages form a vicious cycle that accelerates hypertrophy of adipocytes [6]. Recently, we confirmed that co-culturing adipocytes and macrophages results in a marked up-regulation of the expression of various pro-inflammatory mediators, including TNF-α, MCP-1, and interleukin (IL)-6. In addition, butyrate significantly reduces the expression of these mediators by suppressing both the macrophage inflammatory and adipocyte lipolysis pathways [7]. Prostaglandin E2 (PGE2), an adipocyte and macrophage product, is known to be involved in a variety of cell signaling pathways [8 1].ProductionofPGE2requires a supply of arachidonic acid derived from phospholipids released by phospholipase 2 (PL2) to serve as the substrate for cyclooxygenase-2 (COX2) [8 1]. Short-chain fatty acids (SCFs), such as acetic acid, propionic acid, and butyric acid, are produced by anaerobic bacterial fermentation of undigested carbohydrates in the colon [11] and are rapidly absorbed to provide energy for the colorectal epithelium. Levels of SCFs are increased in rectal mucosal blood [12]. The SCF sodium butyrate (butyrate) modulates the production of inflammatory mediators [13 15]. Reports have demonstrated that butyrate up-regulates the expression of PL2, COX2, and PGE2 in Kupffer cells [16] and COX2 and PGE2 in human peripheral blood mononuclear cells [17]. Whether PGE2 is associated with the suppressive role of butyrate on the vicious cycle is an important question. In order to clarify the anti-lipolytic effect of butyrate in the interactions between macrophages and adipocytes, in the present study, we examined the effect of butyrate on the production of PGE2 and the expression of PL2 and COX2. We also examined the effect of butyrate on components of the lypolytic process, including the TNF-α/nuclear factor-kappa (NF-κ) pathway and PGE2-associated signaling factors such as cyclic adenosine monophosphate (cmp) and protein kinase type 1-α regulatory subunit (PRKR1), in co-cultured adipocytes. Results Effect of butyrate on PGE2 production in co-cultured cells Preliminary experiments using the MTT assay confirmed that incubating RW264.7 cells with >2. mmol/l butyrate and -L1 cells with >1. mmol/l butyrate for 24 h resulted in excessive toxicity (data not shown). Therefore, the maximum concentration of butyrate used in all subsequent experiments was 1. mmol/l. We first examined the effect of butyrate on PGE2 production. The concentration of PGE2 in the co-culture medium increased markedly when cells were cultured using the contact method, whereas the concentration was very low when cells were cultured separately (Fig. 1a). Production of PGE2 by cells co-cultured using the contact method in the presence of butyrate increased significantly in a dose-dependent manner up to a butyrate concentration of.5 mmol/l. similar effect of butyrate was observed in cells co-cultured using the transwell method, although the peak PGE2 value was lower than that produced by cells co-cultured using the contact method (Fig. 1b). When cultured separately, -L1 cells produced more PGE2 than RW264.7 cells (Fig. 1a, b). To examine which cells are the predominant PGE2 producers under co-culture conditions with and without butyrate, we examined PGE2 production in cells cultured separately in the presence of conditioned medium (CM) of the other cell type. -L1 cells produced more PGE2 when cultured in RW264.7-CM, and butyrate significantly increased PGE2 production, by approximately 2-fold compared with cells cultured separately (Fig. 1c). Similarly, RW264.7 cells produced more PGE2 when cultured in -L1-CM; the addition of butyrate significantly increased PGE2 production, by approximately 8-fold compared with cells cultured separately (Fig. 1d). Thus, both cell types produce PGE2, and the presence of butyrate under co-culture conditions stimulates PGE2 production, particularly in RW264.7 cells. Effect of pertussis toxin (PTX) on PGE2 production in co-cultured cells Previously, we found that PTX, a Gi signaling pathway inhibitor, blocks the anti-lipolytic effect of butyrate in cocultured -L1 cells by blocking G-protein coupled receptor (GPR) 41-mediated signaling [7]. Here, as expected, PTX completely suppressed the up-regulation of PGE2 production by cells co-cultured using the contact method with.2 mmol/l butyrate (Fig. 2). GPR19, a nicotinic acid receptor regulated by Gi signaling pathways, was recently identified as a receptor for SCFs [18, 19]. These data suggest that butyrate may stimulate PGE2 production via GPR41- and/or GPR19-mediated pathways. COX2 expression in co-cultured cells To clarify the mechanism by which butyrate enhances the production of PGE2, we monitored the expression of COX2 in RW264.7 cells co-cultured using the transwell method. lthough the levels of COX2 protein and mrn expression in control and butyrate-treated RW264.7 cells
3 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 3 of 17 PGE2 (ng/ml) PGE2 (ng/ml) Contact method co-culture RW RW C.8 D.8 PGE2 (ng/ml).6.4 PGE2 (ng/ml) L1 RW264.7-CM RW L1-CM Fig. 1 Effect of butyrate on the production of PGE2 induced in cells co-cultured using the contact method for 24 h (a), in cells co-cultured using the transwell method for 24 h (b), in -L1 cells incubated with RW264.7-CM for 24 h (c), and in RW264.7 cells incubated with -L1-CM for 24 h (d). The concentration of PGE2 in the co-culture medium was determined by ELIS. The concentration of PGE2 produced by non co-cultured -L1 or RW cells served as the control. Values from five independent experiments are expressed as the mean ± SD. p <.5,p <.1, and p <.1 versus co-culture, as determined by NOV and the Tukey-Kramer test cultured separately were very low, butyrate did induce a marked and significant dose-dependent increase in COX2 protein and mrn expression in co-cultured RW264.7 cells (Fig. 3a-c). Similar butyrate-induced increases in the expression of COX2 protein and mrn were observed in co-cultured -L1 cells (Fig. 3d-f). Thus, increased COX2 expression induced by butyrate may up-regulate PGE2 production in both RW264.7 and -L1 cells in co-culture. ctivities of calcium-dependent cytosolic PL2 (cpl2) and secretory PL2 (spl2) in co-cultured cells Next, we examined whether butyrate treatment increases the activities of cpl2 and spl2 in RW264.7 and 2 ### ### PGE2 (ng/ml) 15 1 ### 5 RW RW Contact method co-culture PTX (1 ng/ml) Fig. 2 Effect of pertussis toxin (PTX) on the production of PGE2 induced by 24 h of co-culture using the contact method. The concentration of PGE2 in the co-culture medium was determined by ELIS. The concentration of PGE2 in the medium of non co-cultured -L1 or RW cells served as the control. Values from five independent experiments are expressed as the mean ± SD. p <.1 versus co-culture, and ### p <.1 versus co-culture with PTX, as determined by NOV and the Tukey-Kramer test
4 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 4 of 17 RW RW COX2 D COX ctin ctin COX2/ctin (ratio of control) ## ## E COX2/ctin (ratio of control) ## ## RW RW C COX2 mrn levels (ratio of control) ### ### ### F COX2 mrn levels (ratio of control) # ### ### RW RW Fig. 3 Effects of butyrate on COX2 protein and mrn expression in RW264.7 (a, b, c) and -L1 (d, e, f) cells co-cultured using the transwell method for 24 h. COX2 and actin proteins were detected by western blotting (a, d). The expression ratio of COX2 to actin was determined based on quantification of the relative density of western blotting bands (b, e). COX2 and β-actin mrns were detected using qrt-pcr (c, f), as described in the Methods. For western blotting and protein expression, n = 3, and for qrt-pcr, n = 5. Values are expressed as the mean ± SD. p <.5, p <.1, and p <.1 versus the control, and # p <.5, ## p <.1,and ### p <.1 versus co-culture, as determined by NOV and the Tukey-Kramer test -L1 cells co-cultured using the transwell method. utyrate induced a significant dose-dependent increase in cpl2 activity in co-cultured RW264.7 cells (Fig. 4a), whereas no cpl2 activity was detected in cocultured -L1 cells (data not shown). The activity of spl2 increased in both RW264.7 and -L1 cells co-cultured using the transwell method. However, butyrate did not enhance the spl2 activity in either cell type in co-culture (Fig. 4b, c). dipose-specific PL2 (dpl) expression in co-cultured -L1 cells dpl2, which was recently discovered, is highly expressed only in adipocytes, where it catalyzes the release of sn-2 fatty acids from phosphatidylcholine [1]. In the present study, we examined the expression of dpl2 in -L1 cells. The expression of dpl2 protein and mrn were enhanced in cells co-cultured using the transwell method compared with cells cultured separately (Fig. 5a-c). utyrate treatment had no additional effect on dpl2 protein and mrn expression in either cocultured -L1 cells or cells cultured separately. These results indicate that co-culture enhances dpl2 expression, but butyrate has no effect on the expression of dpl2 protein or mrn. Effectoftrichostatin(TS)onPGE2productionin co-cultured cells utyrate is known to modulate gene transcription by inducing hyperacetylation of histones through inhibition of histone deacetylation (HDC) [2]. To elucidate the underlying mechanism of butyrate-induced up-regulation of PGE2 production, we examined whether TS, a HDC inhibitor, can mimic the effect of butyrate on PGE2 production. Production of PGE2 by cells co-cultured using both the contact and transwell methods in the presence of TS (25 1 nmol/l) increased significantly in a dosedependent manner (Fig. 6a, b). Thus, inhibition of HDC may be one mechanism by which butyrate increases the production of PGE2.
5 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 5 of 17 cpl2 activity (%) # 8 RW spl2 activity (%) C spl2 activity (%) RW Fig. 4 Effects of butyrate on cpl2 activity in RW264.7 cells (a) and spl2 activity in RW264.7 (b) and -L1 (c) cells co-cultured using the transwell method for 24 h. The activities of intracellular cpl2 and spl2 were measured using an assay kit, as described in the Methods. s the control, activity was measured in non co-cultured cells. Values from five independent experiments are expressed as mean ± SD. p <.5,p <.1, and p <.1 versus the control, and # p <.5 versus co-culture, as determined by NOV and the Tukey-Kramer test Trans-well method co-culture dpl2 ctin C dpl2 / ctin (ratio of control) pla2g16 (dpl2) mrn levels (ratio of control) Trans-well method co-culture Fig. 5 Effects of butyrate on dpl protein (a, b)andmrn(c) expression in -L1 cells co-cultured using the transwell method for 24 h. dpl and actin proteins were detected by western blotting (a). The expression ratio of dpl to actin was determined based on quantification of the relative density of western blotting bands (b). la2g16 and β-actin mrns were detected using qrt-pcr (C), as described in the Methods. For western blotting and protein expression, n = 3, and for qrt-pcr, n = 5. Values are expressed as the mean ± SD. p <.5, and p <.1,p <.1 versus the control, as determined by NOV and the Tukey-Kramer test
6 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 6 of 17 PGE2 (ng/ml) PGE2 (ng/ml) Contact method co-culture TS (nmol/l) RW TS (nmol/l) RW Fig. 6 Effect of TS on the production of PGE2 induced in cells co-cultured using the contact method for 24 h (a) and in cells co-cultured using the transwell method for 24 h (b). The concentration of PGE2 in the co-culture medium was determined by ELIS. The concentration of PGE2 produced by non co-cultured -L1 or RW cells served as the control. Values from five independent experiments are expressed as the mean ± SD. p <.5, p <.1, and p <.1 versus co-culture, as determined by NOV and the Tukey-Kramer test Effect of butyrate and prostaglandin E receptor 3 (EP3)- selective antagonist on lipolysis in co-cultured -L1 cells Previously, we showed that butyrate significantly reduces the release of FFs and free glycerol by diminishing total lipase activity, including that of hormone-sensitive lipase (HSL) and adipose triglyceride lipase (TGL), in cocultured -L1 cells [7]. In this study, we wanted to clarify whether butyrate-associated enhanced PGE2 production reduces lipolysis. PGE2 binds 4 cognate receptors, designated EP1 through EP4. Compared with other EP receptors in adipose tissue, Gαi-coupled EP3 is reportedly expressed at >1-fold markedly higher levels [1]. fter binding to EP3, PGE2 down-regulates lipolysis by inhibiting cmp production in adipocytes [1, 21]. We therefore examined whether treatment with the EP3-selective antagonist L79816 could attenuate the PGE2- and butyratedependent inhibition of lipolysis in adipocytes. The release of both FFs and free glycerol into the medium was increased in cells co-cultured using the contact method. utyrate suppressed FFs and free glycerol release from co-cultured cells (by approximately 5% of the level observed in cells co-cultured using the contact method); exogenous PGE2 also suppressed FFs and free glycerol release, although to a lesser extent (by approximately 2% of the level observed in cells co-cultured using the contact method) (Fig. 7a, b). Exogenous PGE2 had no Free fatty acids ( mol/l) # # Free glycerol ( gl/ml) # # Contact method co-culture PGE2 (5. ng/ml) utyrate (.5 mmol/l) L79816 (1 mol/l) Fig. 7 Effect of butyrate with PGE2 and/or EP3 receptor antagonist (L79816) on lipolysis in cells co-cultured using the contact method for 24 h. Concentrations of FFs (a) and free glycerol (b) in the co-culture medium were determining using an assay kit. s the control, the concentrations of FFs and free glycerol were determined in the medium of untreated and non co-cultured -L1 cells. Values from five independent experiments are expressed as the mean ± SD. p <.5,p <.1 and p <.1 versus co-culture, and # p <.5 versus co-culture with butyrate (.5 mmol/l), as determined by NOV and the Tukey-Kramer test; p <.5 for co-culture with PGE2 (+) and L79816 ( ) versus co-culture with PGE2 (+) and L79816 (+), as determined by paired t-test
7 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 7 of 17 significant additional suppressive effect on butyratetreated cells co-cultured using the contact method. These results suggest that approximately 4% of the effect of butyrate depends on the PGE2-mediated pathway. The EP3-selective antagonist L79816 completely reversed the increase in FFs and free glycerol release from exogenous PGE2-treated cells co-cultured using the contact method. ddition of L79816 with butyrate significantly suppressed FFs and free glycerol release (by approximately 35% of the level observed in cells co-cultured using the contact method). This result suggests that approximately 6% of the effect of butyrate depends on the non-pge2 mediated pathway. Thus, PGE2 inhibits lipolysis via EP3, and butyrate inhibits lipolysis through a mechanism involving both PGE2-mediated and non-pge2 mediated pathways. Effects of PGE2 and butyrate on cmp levels and lipolysis in co-cultured -L1 cells It is known that cmp signaling via protein kinase (PK) is important for lipolysis [22]. To determine whether butyrate inhibits lipolysis by modulating cmp levels, we examined the effects of PGE2 and butyrate on cmp levels in -L1 cells co-cultured using the transwell method. s shown in Fig. 8a, cmp levels significantly increased in -L1 cells co-cultured using the transwell method compared with -L1 cells cultured separately. Consistent with this result, lipolysis increased in -L1 cells cocultured using the transwell method (Fig. 8b-e). PTX treatment increased cmp above the levels observed in -L1 cells co-cultured using the transwell method (Fig. 8a) and increased lipolysis in cells co-cultured using the contact method (Fig. 8b, c). These results are also consistent with previously reported data [7]. Under treatment with PTX, we did not observe any additional effects of butyrate, L79816, and PGE2. We also examined whether the adenylyl cyclase selective inhibitor SQ22536 suppresses cmp production and lipolysis in co-cultured cells. s shown in Fig. 8a, SQ22536 completely suppressed cmp production in -L1 cells co-cultured using the transwell method. SQ22536 inhibited the release of FFs and free glycerol in cells co-cultured using the contact method by approximately 7% of the level observed in co-culture (Fig. 8d, e). Treatment with both SQ22536 and butyrate synergistically suppressed cmp production and the release of FFs and free glycerol. Thus, these results suggest that PGE2 and butyrate inhibit lipolysis by modulating cmp levels. Next, to determine whether butyrate reduces activation of PK after decreasing cmp, we examined the expression of PRKR1 in -L1 cells. utyrate treatment resulted in a dose-dependent decrease in PRKR1 expression (Fig. 9). Effects of NF-κ- and COX2-selective inhibitors on lipolysis in TNF-α stimulated -L1 cells TNF-α exerts a lipolytic effect via an NF-κ mediated pathway in adipocytes [23, 24]. Previously, we showed that co-culture increases TNF-α production, whereas butyrate reduces the production of TNF-α in cocultured cells and inhibits NF-κ activity in co-cultured macrophages, and we also demonstrated that either anti TNF-α antibody or butyrate reduces the release of both FFs and free glycerol from TNF-α stimulated -L1 cells [7]. Here, we examined the importance of PGE2 in the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells. TNF-α enhanced FFs and free glycerol release, and the NF-κ selective inhibitor Y completely suppressed TNF-α enhanced release of FFs and free glycerol (Fig. 1a, b). utyrate reduced the release of FFs and free glycerol from TNFα stimulated -L1 cells by approximately 4% of the level of inhibition observed with Y Exogenous PGE2 reduced the release of FFs and free glycerol by approximately 2% of the level of inhibition observed with Y In contrast, the COX2-selective inhibitor SC reduced TNF-α enhanced release of FFs and free glycerol to a greater degree than butyrate. This result suggests that other COX2 metabolites in addition to PGE2 are associated with lipolysis. Effect of silent interference (si)rns targeting GPR41 or 19 on lipolysis in TNF-α stimulated -L1 cells The results shown in Fig. 2 suggest that butyrate stimulates PGE2 production via GPR41- and/or GPR19-mediated pathways. Here, we examined which is the principal pathway regulating the effect of butyrate on lipolysis using TNF-α stimulated -L1 cells. Using sirns targeting either GPR41 or GPR19, we blunted the expression of GPR41 or GPR19, respectively (Fig. 11a). Using sirn targeting GPR41, the inhibitory effect of butyrate on the release of FFs and glycerol was reduced by half (Fig. 11b, c). Using sirn targeting GPR19, the inhibitory effect of butyrate on the release of FFs and free glycerol was almost completely abrogated (Fig. 11b, c). s shown in Fig. 11d, cmp levels significantly increased in TNF-α stimulated -L1 cells. utyrate reduced cmp levels, and exogenous PGE2 further reduced the cmp levels. These results suggest that PGE2 inhibits lipolysis through a mechanism involving both butyrate-related and nonbutyrate related pathways. Treatment with sirn targeting GPR19 attenuated the inhibitory effect of butyrate on cmp levels to a greater extent than treatment with sirn targeting GPR41 (Fig. 11d). These results correspond to the results of Fig. 11b and c. Thus, the GPR19-mediated pathway may predominate, but the
8 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 8 of 17 cmp (% of control) PGE2 (2. ng/ml) utyrate (.5 mmol/l) PTX (1 ng/ml) SQ22536 (1 mol/l) 8 D 8 Free fatty acids ( mol/l) Free fatty acids ( mol/l) C Free glycerol ( gl/ml) E Free glycerol ( gl/ml) Contact method co-culture utyrate (.5 mmol/l) PTX (1. ng/ml) L79816 (1 mol/l) PGE2 (5. ng/ml) Contact method co-culture utyrate (.5 mmol/l) SQ22536 (1 mol/l) Fig. 8 The effects of butyrate, PGE2, PTX, and adenylyl cyclase selective inhibitor (SQ22536) on cmp formation in -L1 cells co-cultured using the transwell method for 24 h (a). The concentration of intracellular cmp was measured using an assay kit. s the control, the concentration of intracellular cmp was determined in untreated and non co-cultured -L1 cells. The effects of butyrate, PGE2, PTX, and L79816 on lipolysis in cells co-cultured using the contact method for 24 h (a). Concentrations of FFs (b) and free glycerol (c) in the co-culture medium were determined using an assay kit. s the control, the concentrations of FFs and free glycerol were determined in the medium of untreated and non co-cultured -L1 cells. The effects of butyrate and SQ22536 on lipolysis in cells co-cultured using the contact method for 24 h. Concentrations of FFs (d) and free glycerol (e)in the co-culture medium were determined using an assay kit. s the control, the concentrations of FFs and free glycerol were determined in the medium of untreated and non co-cultured -L1 cells. Values from five independent experiments are expressed as the mean ± SD. p <.5, p <.1, p <.1 versus co-culture, as determined by NOV and the Tukey-Kramer test; p <.5 for co-culture without butyrate and with SQ22536 versus co-culture with both butyrate and SQ22536, as determined by paired t-test GPR41-mediated pathway also plays a role in regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells. In the case of both control cells and GPR41 or GPR19 knockdown cells, exogenous PGE2 caused a further significant reduction in the release of FFs and free glycerol compared with butyrate. These results, in conjunction with those shown in Fig. 1, suggest that PGE2 inhibits lipolysis in TNF-α stimulated -L1 cells via a mechanism involving both butyrate-mediated and non-butyrate mediated pathways, although exogenous PGE2 had no significant additional suppressive effect on butyrate-treated cells co-cultured using the contact method (Fig. 7a, b).
9 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 9 of 17 Trans-well method co-culture PRKR1 ctin Fig. 9 Effect of butyrate on the expression of PRKR1 and actin proteins in -L1 cells co-cultured using the transwell method for 24 h. Expression of PRKR1 and actin was detected by western blotting Discussion In this study, we clarified the mechanism underlying the anti-lipolytic effect of butyrate in the interaction between macrophages and adipocytes. utyrate up-regulated cpl2 activity in co-cultured macrophages and COX2 expression in co-cultured macrophages and adipocytes, resulting in enhanced production of PGE2 and subsequent reductions in the levels of cmp and PRKR1, leading to suppression of lipolysis. We first examined the levels of PGE2 in the medium of cells co-cultured using both the contact and transwell methods. PGE2 levels increased markedly in the medium of cells co-cultured using the contact method, whereas PGE2 levels were very low in the medium of cells cultured separately. Interestingly, an additional dose-dependent increase in PGE2 production was observed when cells were treated with butyrate at concentrations up to.5 mmol/l (Fig. 1a). The peak level of PGE2 production was higher when cells were co-cultured using the contact method than when they were cultured using the transwell method (Fig. 1b). oth cell types produced PGE2, and under coculture conditions, butyrate stimulated PGE2 production, particularly in macrophages (Fig. 1c, d). These results suggest that in addition to the effects of other stimulators such as cytokines, direct cell-to-cell contact may play a significant role in PGE2 production. These data were similar to our previously reported data regarding TNF-α, MCP-1, and IL-6 production [7]. Furthermore, Iyer et al. reported that PGE2 concentrations in plasma and white adipose tissues are elevated in obese rats fed a highcarbohydrate/high-fat diet compared with cornstarch-fed normal rats and that increased PGE2 concentrations are correlated with increased adiposity [25]. Our results appear to support their data. In this study, we chose the transwell method for the separate culture of RW264.7 and -L1 cells. Using this approach, we found that cpl2 activity in macrophages, spl2 activity in adipocytes and macrophages, and the expression of dpl2 protein and mrn in adipocytes were up-regulated in co-culture, and butyrate treatment elevated cpl2 activity to a greater degree in Free fatty acids ( mol/l) Free glycerol ( g/ml) L1 TNF- (1 ng/ml) PGE2 (5. ng/ml) Y (1 mol/l) SC (1 mol/l) utyrate (.5 mmol/l) Fig. 1 Effect of PGE2, butyrate, NF-κ selective inhibitor (Y11-782), or COX2-selective inhibitor (SC 58635) on lipolysis in TNF-α stimulated -L1 cells after 24 h. Concentrations of FFs (a) and free glycerol (b) in the co-culture medium were determined using an assay kit. s the control, the concentrations of FFs and free glycerol were determined in the medium of untreated and non co-cultured -L1 cells. Values from five independent experiments are expressed as the mean ± SD. p <.5 and p <.1 versus TNF-α stimulated -L1 cells, as determined by NOV and the Tukey-Kramer test; p <.5, p <.1 for TNF-α (+) and PGE2 ( ) and SC (+) versus TNF-α (+) and PGE2 (+) and SC (+), as determined by paired t-test
10 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 1 of 17 -L1 5 Negative Control sirn GPR41 sirn GPR19 sirn GPR41 GPR19 -ctin Free fatty acids ( mol/l) ## ## ## 1 D cmp (% of control) # # # C Free glycerol ( gl/ml) ## ## ## -L1 TNF- utyrate (.5 mmol/l) PGE2 (5. ng/ml) -L1 TNF- utyrate (.5 mmol/l) PGE2 (5. ng/ml) sirn NC sirn GPR41 sirn GPR19 sirn NC sirn GPR41 sirn GPR19 Fig. 11 Treatment with NC, GPR41, and 19 sirn and detection of the expression of GPR41 and 19 RN in -L1 cells (a). Effect of butyrate, PGE2, and NC, GPR41, and 19 sirn treatment on lipolysis assayed in TNF-α stimulated -L1 cells after 24 h. Concentrations of FFs (b) and free glycerol (c) in the medium were determined using an assay kit. s the control, the concentrations of FFs and free glycerol were determined in the medium of untreated -L1 cells. The effects of butyrate, PGE2, and NC, GPR41, and 19 sirn treatment on cmp accumulation in TNF-α stimulated -L1 cells after 24 h (d). The concentration of intracellular cmp was measured using an assay kit. s the control, the concentration of intracellular cmp was determined in untreated -L1 cells. Values from five independent experiments are expressed as the mean ± SD. p <.5, p <.1, p <.1 versus TNF-α stimulated -L1 cells, as determined by NOV and the Tukey-Kramer test; and # p <.5and ## p <.1 versus TNF-α stimulated -L1 cells treated with butyrate (.5 mmol/l), as determined by NOV and the Tukey-Kramer test; p <.5forTNF-α stimulated -L1 cells treated with GPR41 sirn and butyrate (+) versus -L1 cells treated with GPR19 sirn and butyrate (+), as determined by paired t-test macrophages (Figs. 4 and 5). The observation that PGE2 was produced primarily by macrophages was consistent with those results. utyrate may enhance interactions between cpl2, COX2, and PGE2 in co-cultured macrophages. utyrate modulates gene transcription by inducing hyperacetylation of histones through inhibition of histone deacetylase [2]. utyrate combined with lipopolysaccharide synergistically increases COX2 gene expression in macrophages through both acetylation and phosphorylation of histone H3 at the promoter site [26]. Consistent with previous reports [16, 17], in the present study, butyrate increased the expression of both COX2 protein and mrn in co-cultured adipocytes and macrophages (Fig. 3). lthough we did not examine detailed mechanisms in both cell types, we hypothesize that butyrate treatment increases COX2 gene and protein expression in both adipocytes and macrophages by inhibiting HDC. Furthermore, TS mimicked the up-regulating effect of butyrate on PGE2 production (Fig. 6). lthough our results did not provide direct evidence, taken together, they suggest that the inhibition of HDC is a mechanism by which butyrate increases the production of PGE2. Co-culture with macrophages increased the release of FFs and free glycerol from adipocytes into the culture medium, and butyrate suppressed this release. PGE2 also suppressed the release of FFs and free glycerol, although to a lesser extent than butyrate. Complementarily, the EP3-selective antagonist L79816 attenuated the PGE2- and butyrate-dependent inhibition of lipolysis (Fig. 7). We determined that approximately 4% of the suppressive effect of butyrate depends on the PGE2- mediated pathway, whereas 6% depends on the non- PGE2 mediated pathway. TNF-α exerts a lipolytic effect via an NF-κ mediated pathway in adipocytes [23, 24]. SCFs inhibit the
11 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 11 of 17 activation of NF-κ, leading to suppression of the multiple responses in both immune cells and various other cell types [27]. Previously, we showed that coculture increases TNF-α production, whereas butyrate decreases TNF-α production by reducing TNF-α mrn expression in co-cultured both cells and inhibiting NFκ activity in co-cultured macrophages [7]. We thus speculated that butyrate reduces lipolysis via a PGE2- mediated pathway by suppressing the production of TNF-α. In this study, we examined the importance of PGE2 in regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells (Fig. 1). utyrate reduced the release of FFs and free glycerol by approximately 4% of the level of inhibition observed with the NF-κ selective inhibitor, Y11-782; exogenous PGE2 reduced the release of FFs and free glycerol by approximately 2% of that level. Taken together, these results suggest that butyrate contributes at least in part to the effect on lipolysis via an NF-κ mediated pathway and that additional non-pge2 mechanisms also appear to be relevant. The COX2-selective inhibitor SC reduced NFκ mediated lipolysis to a greater degree than butyrate, suggesting that the mechanism involves other COX2 metabolites in addition to PGE2 and butyrate (Fig. 1). Previously, we showed that butyrate significantly reduces FFs release in TNF-α stimulated -L1 cells by diminishing total lipase activity by suppressing the expression of TGL and the phosphorylation of HSL (pser 66 ) in co-cultured -L1 cells [7]. Exogenous TNF-α induces an increase in intracellular cmp production in adipocytes [28]. Elevation of intracellular cmp activates cmp-dependent protein kinase, with subsequent expression of TGL and phosphorylation of HSL in adipocytes [29, 3]. PGE2 down-regulates lipolysis by inhibiting cmp production in adipocytes [1, 21]. To further elucidate the underlying mechanism by which butyrate inhibits lipolysis in adipocytes, we therefore examined the effects of PGE2 and butyrate on cmp levels in co-cultured -L1 cells. We found that cmp levels were significantly higher in cocultured -L1 cells compared with -L1 cells cultured separately, and butyrate treatment restored cmp levels, lipolysis, and PRKR1 expression (Figs. 8 and 9). Thus, the present data appear to support our previous results. It is unclear why PTX increased cmp levels to a greater degree than co-culture in adipocytes and why treatment with both SQ22536 and butyrate synergistically suppressed cmp levels and lipolysis. There may be other mechanisms regulating cmp levels in adipocytes. GPR41, GPR43, and GPR19 exhibit affinity for SCFs [19, 31 33]. PTX, which is known to inactivate Gi/ o proteins, negates the effect of GPR41 and GPR19 but not that of GPR43 [33, 34]. The observation that PTX completely reversed the up-regulated production of PGE2 induced by butyrate (Fig. 2) suggests that the effect of butyrate on PGE2 production may involve GPR41- and/or GPR19-mediated signaling. We therefore sought to determine which pathway plays the greatest role in regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells. Using sirns targeting GPR41 or GPR19, we found that the GPR19-mediated pathway predominates, but the GPR41-mediated pathway also plays a role in regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells. Interestingly, inflammation stimulates GPR19 expression in adipocytes and macrophages [35]. TNF-α also enhances GPR19 expression in adipocytes, leading to an acceleration of the suppressive effect of butyrate on lipolysis. Several hypothetical pathways are shown in Fig. 12. Co-culture elevates PGE2; butyrate increases PGE2 levels further by elevating cpl2 activity in macrophages and COX2 expression in both types of cells in co-culture (Fig. 12a). Co-culture also increases cmp and PK levels in adipocytes and increases the release of FFs and free glycerol into the medium (lipolysis). utyrate suppresses cmp and PK levels, and exogenous PGE2 has a lesser effect in this regard than butyrate. These suppressive effects may reduce the activities of lipases, including TGL and HSL, resulting in inhibition of lipolysis (Fig. 12b). Coculture increases TNF-α production. TNF-α in turn increases cmp levels, leading to increased lipolysis. nti TNF-α antibody, butyrate, or exogenous PGE2 decrease cmp levels and reduce lipolysis in TNF-α stimulated -L1 cells. The GPR19-mediated pathway thus appears to predominate in regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells (Fig. 12c). Rumberger et al. recently reported that butyrate increases lipolysis in -L1 cells [36]. However, their methods differed from ours in several respects. First, they used -L1 cells but not co-culture. In addition, they used a higher concentration of butyrate than we did (5 mmol/l versus.1 1. mmol/l). Finally, they used cells at 3 1 days post-differentiation, whereas we used cells at 2 days post-differentiation. Similarly, in this study, we found that PGE2 has a small additional effect on TNF-α stimulated -L1 cells compared with butyrate, but this effect was not observed with -L1 cells co-cultured with RW264.7 cells. Thus, differences in the condition of the cells could have affected the action of butyrate. To fully elucidate the suppressive effect of butyrate on lipolysis in adipose tissues, therefore, further in vitro and in vivo studies will be necessary. Finally, many other nutritional supplements, such as other SCFs, glucose, or fructose, may regulate lipolysis in -L1 cells. Further investigations are thus needed to elucidate the interactions between adipocytes and macrophages in metabolic disorders.
12 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 12 of 17 C Fig. 12 (See legend on next page.)
13 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 13 of 17 (See figure on previous page.) Fig. 12 Hypothetical pathways based on the results of the present study and our previous data. a The effect of butyrate on PGE2 production in the interaction between co-cultured macrophages and adipocytes. Co-culture elevates cpl2 activity in macrophages, spl2 activity in adipocytes and macrophages, and the expression of dpl2 protein and mrn in adipocytes. utyrate elevates cpl2 activity to a greater degree in macrophages. Co-culture elevates COX2 expression in both cells, and butyrate further enhances COX2 expression in both cells. Thus, butyrate increases PGE2 production more than co-culture alone. b The effects of butyrate and PGE2 on lipolysis in co-cultured adipocytes. Co-culture increases cmp and PK (PRKR1) levels in adipocytes and increases the release of FFs and free glycerol into the medium (lipolysis). utyrate suppresses cmp and PK levels, and exogenous PGE2 has a lesser effect than butyrate. These suppressive effects may reduce the activities of lipases, including TGL and HSL, thus resulting in inhibition of lipolysis. c The effects of butyrate and exogenous PGE2 on cmp- and NF-κ mediated lipolysis in TNF-α stimulated -L1 adipocytes. Co-culture increases TNF-α production. TNF-α increases cmp, leading to increased lipolysis. nti TNF-α antibody, butyrate, or exogenous PGE2 decrease cmp levels and reduce lipolysis in TNF-α stimulated -L1 cells. The GPR19-mediated pathway may be the predominant pathway regulating the effect of butyrate on lipolysis in TNF-α stimulated -L1 cells Conclusions utyrate attenuates lipolysis in adipocytes co-cultured with macrophages via non-pge2 mediated and PGE2- mediated pathways. Methods Reagents and cells utyric acid and FF-free bovine serum albumin (S) were purchased from Wako Chemicals (Osaka, Japan). ll other chemicals were obtained from Nacalai Tesque (Kyoto, Japan). RW264.7 mouse macrophages and - L1 mouse pre-adipocytes (merican Type Culture Collection, Manassas, V) were cultured in DMEM (Nissui Pharmaceutical Co., Ltd., Tokyo, Japan) with 292 μg/ml L-glutamine, 1 units/ml penicillin, 1 μg/ml streptomycin, and 1% fetal bovine serum (FS) at 37 C under 5% CO 2. Differentiation of -L1 pre-adipocytes was induced by addition of.5 mmol/l 13-isobutyl-1-methylxanthine, 2.5 μmol/l dexamethasone, and 1 μg/ml insulin (Wako Chemical) in DMEM containing 1% FS, beginning 2 days after the cells reached confluence in a 24-well plate. The medium was then replaced with DMEM containing 1% FS and 5. μg/ml insulin and was changed every 2 or 3 days. Twenty days after induction of differentiation, the hypertrophied -L1 adipocytes were used in experiments. Cell viability was assessed using an MTT cell viability assay kit (R&D Systems, Minneapolis, MN). Co-culture of adipocytes and macrophages dipocytes and macrophages were co-cultured using two different methods, the contact method and the transwell method [6, 7]. In the contact method, serum-starved differentiated -L1 cells ( cells/well) were cultured in a 24-well plate, and RW264.7 cells ( cells/well) were then plated onto the -L1 cells. The cells were cultured in contact with each other for 24 h in the presence of butyrate (,.1,.2,.5, or 1. mmol/l) in 2% FF-free S DMEM and then harvested. In the transwell method, cells were co-cultured using 24-well transwell plate inserts with a.4-μm porous membrane (Corning, New York, NY), which separated the serumstarved -L1 cells ( cells/well) in the lower chamber from the serum-starved RW264.7 ( cells/well) cells in the upper chamber. fter incubation with butyrate (,.1,.2,.5, or 1. mmol/l) in 2% FFfree S for 24 h, both the RW264.7 cells in the upper chamber and the -L1 cells in the lower chamber were harvested. We used the contact method for experiments with culture medium, whereas we used the transwell method for experiments with each cell type separately, such as for the collection of mrn or detection of cmp, according to our previous report [7]. Measurement of PGE2 The concentration of PGE2 in the culture medium was determined using a Prostaglandin E2 Express EI kit (Cayman Chemical Co., nn rbor, MI) in accordance with the manufacturer s instructions. Levels of PGE2 secreted by RW264.7 or -L1 cells cultured separately were determined as a control. -L1 and RW264.7 CM Hypertrophied -L1 adipocytes were cultured in 2% FF-free S in a 24-well plate for 24 h, after which the supernatant was collected as -CM and stored at 8 C until use. RW264.7 macrophages were cultured in 2% FF-free S in a 24-well plate for 24 h, after which the supernatant was collected as RW-CM and stored at 8 C until use. fter incubation of -L1 cells with RW-CM or incubation of RW264.7 cells with -CM for 24 h with or without.5 mmol/l butyrate, PGE2 produced by both types of cells was measured. Effect of TS on PGE2 production in co-culture Cells were cultured using the contact method for 24 h in the presence of TS (Cayman Chemical) (, 25, 5, 1 nmol/l) in 2% FF-free S culture medium and then harvested. TS was dissolved in.5% (v/v) dimethylsulfoxide (DMSO). Effect of treatment with PTX Using the contact method, -L1 adipocytes were pretreated with 1 ng/ml of PTX at 37 C for 6 min. The
14 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 14 of 17 cells were then cultured for 24 h in the presence of PTX and butyrate (,.1,.2,.5, or 1. mmol/l) in 2% FFfree S culture medium and then harvested. Lipolysis assay Differentiated -L1 cells were incubated overnight using the contact method in serum-free DMEM with 2% FF-free S. The cells were then co-cultured with RW264.7 cells in the same medium for 24 h with butyrate (,.5 mmol/l) and PGE2 (, 5 ng/ml), after which the culture medium was harvested. The levels of FFs in the co-culture medium were measured using an acyl-coenzyme oxidase based colorimetric assay method, which enables detection of C8 (octane) and longer-chain fatty acids. The assay was conducted in accordance with the manufacturer s instructions [37]. Levels of free glycerol in the medium were measured using an dipolysis ssay kit (Cayman Chemical). To examine the effects of Y and SC 58635, - L1 cells were pretreated by culturing at 37 C for 6 min in serum-free DMEM with.5% (v/v) DMSO (vehicle), 5. ng/ml PGE2 (Cayman Chemical) dissolved in DMSO, 1 μmol/l Y (Santa Cruz iotechnology, Santa Cruz, C) dissolved in DMSO, or 1 μmol/l SC (R&D Systems) dissolved in DMSO. -L1 cells were then stimulated with TNF-α (with or without butyrate) in 2% FF-free S medium and cultured for 24 h, after which levels of FFs and free glycerol released into the medium were measured. Quantitative reverse transcription polymerase chain reaction (qrt-pcr) Total RN was extracted from RW264.7 cells in the transwell plate upper chamber and from -L1 cells in the lower chamber using TRIzol reagent (Invitrogen, Carlsbad, C). Single-stranded complementary DN (cdn) was synthesized from 1 μg of total RN using a ReverTrace-α first-strand cdn synthesis kit (Toyobo, Osaka, Japan). Samples were then incubated at 37 C for 6 min. The temperature of the reaction was then raised to 94 C for 5 min in order to inactivate the enzyme, after which the temperature was reduced to 4 C. cdns were synthesized using a Gene mp PCR System 97 (pplied iosystems, Carlsbad, C). To quantify mrn expression, real-time PCR was performed using a LightCycler System (Roche Diagnostics, Mannheim, Germany) and LightCycler Fast Start DN Master Plus SYR Green 1 (Roche Diagnostics), as described previously [7]. The linearity of amplification was determined for COX2 and β- actin cdns from RW264.7 or -L1 cells and for pla2g16 (dpl) cdns from -L1 cells. Primers for the different genes are listed in Table 1. The real-time PCR program was as follows: for COX2, dpl, and β- actin, 45 cycles of 95 C for 1 s, 55 C for 1 s, and 72 C for 1 s; for LPL, 45 cycles of 95 C for 1 s, 6 C for 1 s, and 72 C for 1 s. The level of β-actin mrn was adopted as an internal standard for the determination of target mrn levels. Samples were analyzed five times. Ct values (the cycle at which the emitted fluorescence signal was significantly higher than the background level and inversely proportional to the initial template copy number) were calculated by the LightCycler software. Finally, the relative expression level of each gene was calculated according to the 2 -ΔΔCt method. Reverse transcription-polymerase chain reaction (RT-PCR) Total RN was extracted from the hypertrophied - L1 adipocytes in 24 well plate using TRIzol reagent (Invitrogen). Single-stranded cdn was synthesized from 1 μg of total RN using a ReverTra ce-α, first strand cdn synthesis kit (Toyobo). Incubation was carried out at 37 C for 6 min. The temperature of the reaction was then raised to 94 C for 5 min in order to inactivate the enzyme and was then reduced to 4 C. To detetect GPR41, GPR19 and β-actin mrn expression, PCR amplification was performed using a Gene mp PCR System 97 (pplied iosystems). Primers for the different genes are listed in Table 1. RN interference Mouse GPR41 sirn duplex and negative control (NC) sirn were purchased from Santa Cruz iotechnology. Mouse GPR19 sirn duplexes were purchased from Origene Technologies (Rockville, MD). Hypertrophied -L1 adipocytes were seeded in 24-well plates and transfected with each targeting sirn at 1 μmol/l (final concentration 5 pmol/well). The sirn and lipofectamine RN imx reagent (Invitrogen) were diluted separately in Opti-MEM Medium (Invitrogen), then the two solutions were gently mixed and incubated for 5 min at room temperature. -L1 cells were transfected with sirn-lipofectamine complexes and incubated for 48 h at 37 C under 5% CO 2 and then used in the experiments. Western blotting Cells were co-cultured using the transwell method. Protein was extracted from RW264.7 cells cultured in the upper chamber and from -L1 cells cultured in the lower chamber. For western blot analysis, cells were lysed using M-PER Mammalian Protein Extraction reagent (Pierce, Rockford, IL) with a protease and phosphatase inhibitor cocktail (Nacalai Tesque). Protein concentration was determined using a protein assay reagent (io-rad Laboratories, Hercules, C). Denatured proteins were separated using SDS-polyacrylamide gel electrophoresis on a 1% polyacrylamide gel and then transferred onto Immobilon-P membranes (Millipore, illerica, M).
15 Ohira et al. Lipids in Health and Disease (216) 15:213 Page 15 of 17 Table 1 Primer sequences used for qrt-pcr or RT-PCR Gene Forward 5 3 Reverse 5 3 Geneank accesion number COX2 GGGGCCTTCTCCCC GCCCGGTCCTCGCTT NM_ pla2g16 (dpl) TCGGCTGCCGCGGCT CCCTCCGCGTGCCTCCG C GPR41 TCCGTCTCCTGTTGGTGTTCC CTCTCTGTCGTCGGCGG NM_ GPR19 TTCTCCTGTCTCTGCCTGCC TGTCTGTCCTCTGTCTCTGCC NM_371.3 β-ctin CTGGCCCCGTGG CCGGGCTCGGGC NM_ COX2 cyclooxygenase-2, dpl adipose specific PL2, LPL lipoprotein lipase The following antibodies were used: rabbit anti-mouse HRSL3 (dpl) (1:1; D iosciences, Franklin Lakes, NJ), rabbit anti-mouse COX2 (1:1; Santa Cruz iotechnology), rabbit anti-mouse PRKR1 (1:1; GeneTex, Irvine, C), rabbit anti-mouse actin (1:1; Santa Cruz iotechnology), and anti-rabbit horseradish peroxidase conjugated immunoglobulin G (1:2; GE Healthcare, ucks, UK). The blots were developed using ECL (GE Healthcare). Relative protein expression levels were determined by dividing the band intensity of the product of interest by that of the actin control band. The intensity of the level of objective protein expression was analyzed using ImageJ software ( [38]. Calcium-dependent cpl2 and spl2 activity Protein was extracted from -L1 cells in the lower chamber and RW264.7 cells in the upper chamber of the transwell plate. Cell lysates were prepared after 24 h of co-culture with butyrate treatment. The protein concentration was adjusted to 1 mg/ml and cpl2 and spl2 activity was assayed five times using cpl2 and spl2 colorimetric assay kit (Cayman Chemical), respectively, according to the manufacturer s instructions. Measurement of intracellular cmp concentration Differentiated -L1 cells cultured using the transwell method were treated with butyrate. The intracellular cmp concentration was determined using a cmp Total EI kit (rbor ssay Co., nn rbor, MI) in accordance with the manufacturer s instructions. The results were standardized by dividing the cmp concentration by the intracellular total protein concentration (1 mg/ml). Effect of butyrate and EP3 antagonist on lipolysis in co-culture Differentiated -L1 cells were pretreated for 1 h with.5% (v/v) DMSO alone (vehicle), 1 μmol/l of the EP3- selective antagonist L79816 (Sigma-ldrich, St. Louis, MO), or 5. ng/ml of PGE2 dissolved in DMSO and diluted in serum-free DMEM. The cells were then cocultured for 24 h using the contact method in DMEM with 2% FF-free S. Cells were also co-cultured in the same manner with and.5 mmol/l butyrate and and 5. ng/ml PGE2. Concentrations of FFs in the coculture medium were measured using an acyl-coenzyme oxidase based colorimetric assay kit, and free glycerol levels were measured using an dipolysis ssay kit. Effect of butyrate on cmp levels in -L1 cells Differentiated -L1 cells were pretreated for 1 h with.5% (v/v) DMSO alone (vehicle) or 1 μmol/l of the adenylyl cyclase selective inhibitor SQ22536 (bcam, Cambridge, UK) dissolved in DMSO and diluted in serum-free DMEM. Differentiated -L1 cells cultured using the transwell method were treated with butyrate, PGE2, PTX, or SQ22536 for 24 h. Protein was extracted from -L1 cells cultured in the lower chamber, and cmp levels were determined using a cmp Total EI kit in accordance with the manufacturer s instructions. The results were standardized by dividing the cmp concentration by the intracellular total protein concentration (1 mg/ml). Effect of butyrate and an adenylyl cyclase selective inhibitor on lipolysis in co-cultured cells Differentiated -L1 cells were pretreated for 1 h with.5% (v/v) DMSO alone (vehicle) or 1 μmol/l SQ22536 dissolved in DMSO and diluted in serum-free DMEM. The cells were then co-cultured for 24 h using the contact method in DMEM with 2% FF-free S and treated with butyrate or SQ22536 in 24 h. Concentrations of FFs in the co-culture medium were measured using an acyl-coenzyme oxidase based colorimetric assay kit, and free glycerol levels were measured using an dipolysis ssay kit. Statistical analysis Data are expressed as mean ± SD. We used a paired t- test for comparisons between two groups or NOV with a Tukey-Kramer post-hoc test for comparisons between more than three groups. Statistical analyses were performed using GraphPad Prism software, ver. 5. (GraphPad Software Inc., San Diego, C). p value <.5 in two-tailed tests was considered to indicate statistical significance.
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
www.sciencesignaling.org/cgi/content/full/9/439/ra78/dc1 Supplementary Materials for Small heterodimer partner mediates liver X receptor (LXR) dependent suppression of inflammatory signaling by promoting
Department of Pharmaceutical Sciences, School of Pharmacy, Northeastern University, Boston, MA 02115, USA 2
 Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Pancreatic Cancer Cell Exosome-Mediated Macrophage Reprogramming and the Role of MicroRNAs 155 and 125b2 Transfection using Nanoparticle Delivery Systems Mei-Ju Su 1, Hibah Aldawsari 2, and Mansoor Amiji
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
The p38 MAPK inhibitor SB differentially modulates LPS-induced interleukin 6 expression in macrophages
 Experimental immunology DOI: 1.5114/ceji.215.54586 The p38 MPK inhibitor S2358 differentially modulates LPS-induced interleukin 6 expression in macrophages Qinghai Shi, Liping Cheng, Zhengxiang Liu, Keyan
Experimental immunology DOI: 1.5114/ceji.215.54586 The p38 MPK inhibitor S2358 differentially modulates LPS-induced interleukin 6 expression in macrophages Qinghai Shi, Liping Cheng, Zhengxiang Liu, Keyan
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Supplemental Material:
 Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
Supplemental Material: MATERIALS AND METHODS RNA interference Mouse CHOP sirna (ON-TARGETplus SMARTpool Cat# L-062068-00) and control sirna (ON-TARGETplus Control) were purchased from Dharmacon. Transfection
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplementary Fig. 1. Identification of acetylation of K68 of SOD2
 Supplementary Fig. 1. Identification of acetylation of K68 of SOD2 A B H. sapiens 54 KHHAAYVNNLNVTEEKYQEALAK 75 M. musculus 54 KHHAAYVNNLNATEEKYHEALAK 75 X. laevis 55 KHHATYVNNLNITEEKYAEALAK 77 D. rerio
Supplementary Fig. 1. Identification of acetylation of K68 of SOD2 A B H. sapiens 54 KHHAAYVNNLNVTEEKYQEALAK 75 M. musculus 54 KHHAAYVNNLNATEEKYHEALAK 75 X. laevis 55 KHHATYVNNLNITEEKYAEALAK 77 D. rerio
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments:
 Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
Supplementary Figure S1 Targeted disruption and overexpression of Gpr43 in mice. (a) A targeting vector was constructed by ligation of 3 fragments: the 5' and 3' homology recombination arms and a fragment
INTRODUCTION. Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells
 Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells Galina Chipitsyna, Qiaoke Gong, Chance F. Gray et al. Endocrinology,
Induction of Monocyte Chemoattractant Protein-1 (MCP-1) Expression by Angiotensin II (AngII) in the Pancreatic Islets and Beta Cells Galina Chipitsyna, Qiaoke Gong, Chance F. Gray et al. Endocrinology,
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Validation & Assay Performance Summary
 Validation & Assay Performance Summary LanthaScreen IGF-1R GripTite Cells Cat. no. K1834 Modification Detected: Phosphorylation of Multiple Tyr Residues on IGF-1R LanthaScreen Cellular Assay Validation
Validation & Assay Performance Summary LanthaScreen IGF-1R GripTite Cells Cat. no. K1834 Modification Detected: Phosphorylation of Multiple Tyr Residues on IGF-1R LanthaScreen Cellular Assay Validation
Advances in Computer Science Research, volume 59 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016)
 7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
7th International Conference on Education, Management, Computer and Medicine (EMCM 2016) Expression of Beta-Adrenergic Receptor in Glioma LN229 Cells and Its Effect on Cell Proliferation Ping Wang1, Qingluan
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR
 Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR Gene Forward Primer (5-3 ) Reverse Primer (5-3 ) cadl CTTGGGGGCGCGTCT CTGTTCTTTTGTGCCGTTTCG cyl-coenzyme Dehydrogenase, very
Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR Gene Forward Primer (5-3 ) Reverse Primer (5-3 ) cadl CTTGGGGGCGCGTCT CTGTTCTTTTGTGCCGTTTCG cyl-coenzyme Dehydrogenase, very
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
JLR Papers In Press. Published on October 16, 2003 as Manuscript D JLR200
 JLR Papers In Press. Published on October 16, 2003 as Manuscript D300024-JLR200 A method of direct measurement for the enzymatic determination of cholesterol esters Toshimi Mizoguchi 1, Toshiyuki Edano,
JLR Papers In Press. Published on October 16, 2003 as Manuscript D300024-JLR200 A method of direct measurement for the enzymatic determination of cholesterol esters Toshimi Mizoguchi 1, Toshiyuki Edano,
Supplementary Figure 1
 Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplemental Figure 1. Western blot analysis indicated that MIF was detected in the fractions of
 Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
Supplemental Figure Legends Supplemental Figure 1. Western blot analysis indicated that was detected in the fractions of plasma membrane and cytosol but not in nuclear fraction isolated from Pkd1 null
RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using
 Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle.
 Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supporting Information
 Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
In The Name Of God. In The Name Of. EMRI Modeling Group
 In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein
![2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein](/thumbs/86/94743397.jpg) Supplementary Information 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked amino-modification products by acrolein Ayumi Tsutsui and Katsunori Tanaka* Biofunctional Synthetic Chemistry Laboratory, RIKEN
Supplementary Information 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked amino-modification products by acrolein Ayumi Tsutsui and Katsunori Tanaka* Biofunctional Synthetic Chemistry Laboratory, RIKEN
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy
 Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration
 Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Programmed necrosis, not apoptosis, is a key mediator of cell loss and DAMP-mediated inflammation in dsrna-induced retinal degeneration The Harvard community has made this article openly available. Please
Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit
 Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Amersham ECL Prime Western blotting reagent
 GE Healthcare Life Sciences Data file 28-9857-23 AA Western blotting reagents Amersham ECL Prime Western blotting reagent Since its introduction in 199, the enhanced chemiluminescence (ECL) Western Blotting
GE Healthcare Life Sciences Data file 28-9857-23 AA Western blotting reagents Amersham ECL Prime Western blotting reagent Since its introduction in 199, the enhanced chemiluminescence (ECL) Western Blotting
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Doctoral Degree Program in Marine Biotechnology, College of Marine Sciences, Doctoral Degree Program in Marine Biotechnology, Academia Sinica, Taipei,
 Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
For the rapid, sensitive and accurate measurement of Glycerol in cell cultures.
 ab185433 Lipolysis Assay Kit (Colorimetric) Instructions for Use For the rapid, sensitive and accurate measurement of Glycerol in cell cultures. This product is for research use only and is not intended
ab185433 Lipolysis Assay Kit (Colorimetric) Instructions for Use For the rapid, sensitive and accurate measurement of Glycerol in cell cultures. This product is for research use only and is not intended
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Table S1. Primers and fluorescent probes used for qrt-pcr analysis of relative expression levels of PPP family phosphatases. gene name forward primer, 5-3 probe, 5-3 reverse primer,
SUPPLEMENTARY MATERIAL Table S1. Primers and fluorescent probes used for qrt-pcr analysis of relative expression levels of PPP family phosphatases. gene name forward primer, 5-3 probe, 5-3 reverse primer,
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS
 Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Protein extraction and western blot analysis Protein extraction was performed as
 ESM Methods Protein extraction and western blot analysis Protein extraction was performed as previously described [1]. 2 g protein was loaded on SDSPAGE and immunoblotted with antibodies to mouse AKT (1:1,
ESM Methods Protein extraction and western blot analysis Protein extraction was performed as previously described [1]. 2 g protein was loaded on SDSPAGE and immunoblotted with antibodies to mouse AKT (1:1,
For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin
 Materials and Methods Pair Feeding Experiment For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin (0.005%), which is average daily food intake of mice fed control HFD ad libitum at week
Materials and Methods Pair Feeding Experiment For pair feeding, mice were fed 2.7g of HFD containing tofogliflozin (0.005%), which is average daily food intake of mice fed control HFD ad libitum at week
TITLE: Effect of COX-2 (PGE2) and IL-6 on Prostate Cancer Bone Mets
 AD Award Number: W81XWH-05-1-0166 TITLE: Effect of COX-2 (PGE2) and IL-6 on Prostate Cancer Bone Mets PRINCIPAL INVESTIGATOR: Alice C. Levine, M.D. CONTRACTING ORGANIZATION: Mount Sinai School of Medicine
AD Award Number: W81XWH-05-1-0166 TITLE: Effect of COX-2 (PGE2) and IL-6 on Prostate Cancer Bone Mets PRINCIPAL INVESTIGATOR: Alice C. Levine, M.D. CONTRACTING ORGANIZATION: Mount Sinai School of Medicine
Quantitative Real-Time PCR was performed as same as Materials and Methods.
 Supplemental Material Quantitative Real-Time PCR Quantitative Real-Time PCR was performed as same as Materials and Methods. Expression levels in the aorta were normalized to peptidylprolyl isomerase B
Supplemental Material Quantitative Real-Time PCR Quantitative Real-Time PCR was performed as same as Materials and Methods. Expression levels in the aorta were normalized to peptidylprolyl isomerase B
Chapter 10. Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002
 Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis
 212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
212 66 4 329334 Effect of Taurine on Acinar Cell Apoptosis and Pancreatic Fibrosis in Dibutyltin Dichloride-induced Chronic Pancreatitis a,c a* b b a a b a a b c 33 66 4 ʼ 6 6 6 28 6 5 5 5 28 45 ʼ ʼ ʼ
Male 30. Female. Body weight (g) Age (weeks) Age (weeks) Atg7 f/f Atg7 ΔCD11c
 ody weight (g) ody weight (g) 34 3 Male 3 27 Female 26 24 22 18 7 9 11 13 15 17 19 21 23 21 18 15 7 9 11 13 15 17 19 21 23 Age (weeks) Age (weeks) Supplementary Figure 1. Lean phenotypes in mice regardless
ody weight (g) ody weight (g) 34 3 Male 3 27 Female 26 24 22 18 7 9 11 13 15 17 19 21 23 21 18 15 7 9 11 13 15 17 19 21 23 Age (weeks) Age (weeks) Supplementary Figure 1. Lean phenotypes in mice regardless
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
McAlpine PERK-GSK3 regulates foam cell formation. Supplemental Material. Supplementary Table I. Sequences of real time PCR primers.
 Mclpine PERK-GSK3 regulates foam cell formation Supplemental Material Supplementary Table I. Sequences of real time PCR primers. Primer Name Primer Sequences (5-3 ) Product Size (bp) GRP78 (human) Fwd:
Mclpine PERK-GSK3 regulates foam cell formation Supplemental Material Supplementary Table I. Sequences of real time PCR primers. Primer Name Primer Sequences (5-3 ) Product Size (bp) GRP78 (human) Fwd:
Free Fatty Acid Assay Kit (Fluorometric)
 Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice.
 Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used for PCR and qpcr Primer Name
 Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
TITLE: Adipose Estrogen and Increased Breast Cancer Risk in Obesity: Regulation by Leptin and Insulin
 D ward Number: W81XH--1-497 TITLE: dipose Estrogen and Increased Breast Cancer Risk in Obesity: Regulation by Leptin and Insulin PRINCIPL INVESTIGTOR: Dr. Fahumiya Samad CONTRCTING ORGNIZTION: La Jolla
D ward Number: W81XH--1-497 TITLE: dipose Estrogen and Increased Breast Cancer Risk in Obesity: Regulation by Leptin and Insulin PRINCIPL INVESTIGTOR: Dr. Fahumiya Samad CONTRCTING ORGNIZTION: La Jolla
Supplementary Materials
 Supplementary Materials 1 Supplementary Table 1. List of primers used for quantitative PCR analysis. Gene name Gene symbol Accession IDs Sequence range Product Primer sequences size (bp) β-actin Actb gi
Supplementary Materials 1 Supplementary Table 1. List of primers used for quantitative PCR analysis. Gene name Gene symbol Accession IDs Sequence range Product Primer sequences size (bp) β-actin Actb gi
EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL MIGRATION IN PANCREATIC CANCER CELLS
 EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL MIGRATION IN PANCREATIC CANCER CELLS VIDYA MEDEPALLI ADVISOR: Maria Eugenia Sabbatini, PhD PHI KAPPA PHI RESEARCH CONFERENCE MARCH 8 th, 2017 Pancreatic
EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL MIGRATION IN PANCREATIC CANCER CELLS VIDYA MEDEPALLI ADVISOR: Maria Eugenia Sabbatini, PhD PHI KAPPA PHI RESEARCH CONFERENCE MARCH 8 th, 2017 Pancreatic
Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways
![Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic Agent that May Act through IRS-1, Akt and GSK-3β Pathways](/thumbs/96/128474991.jpg) Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2016 Supplementary Data Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic
Electronic Supplementary Material (ESI) for MedChemComm. This journal is The Royal Society of Chemistry 2016 Supplementary Data Synthesis of Substituted 2H-Benzo[e]indazole-9-carboxylate as Potent Antihyperglycemic
ab Adipogenesis Assay Kit (Cell-Based)
 ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION
 Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Supplementary Information POLO-LIKE KINASE 1 FACILITATES LOSS OF PTEN-INDUCED PROSTATE CANCER FORMATION X. Shawn Liu 1, 3, Bing Song 2, 3, Bennett D. Elzey 3, 4, Timothy L. Ratliff 3, 4, Stephen F. Konieczny
Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows
 FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
control kda ATGL ATGLi HSL 82 GAPDH * ** *** WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi iwat gwat ibat
 body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
SUPPLEMENT. Materials and methods
 SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
SUPPLEMENT Materials and methods Cell culture and reagents Cell media and reagents were from Invitrogen unless otherwise indicated. Antibiotics and Tet-certified serum were from Clontech. In experiments
Cell Signaling (part 1)
 15 Cell Signaling (part 1) Introduction Bacteria and unicellular eukaryotes respond to environmental signals and to signaling molecules secreted by other cells for mating and other communication. In multicellular
15 Cell Signaling (part 1) Introduction Bacteria and unicellular eukaryotes respond to environmental signals and to signaling molecules secreted by other cells for mating and other communication. In multicellular
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Berberine Sensitizes Human Ovarian Cancer Cells to Cisplatin Through mir-93/ PTEN/Akt Signaling Pathway
 Chen Accepted: et al.: February Berberine 24, Sensitizes 2015 Ovarian Cancer Cells to Cisplatin www.karger.com/cpb 956 1421-9778/15/0363-0956$39.50/0 Original Paper This is an Open Access article licensed
Chen Accepted: et al.: February Berberine 24, Sensitizes 2015 Ovarian Cancer Cells to Cisplatin www.karger.com/cpb 956 1421-9778/15/0363-0956$39.50/0 Original Paper This is an Open Access article licensed
ZL ZDF ZDF + E2 *** Visceral (g) ZDF
 Body Weight (g) 4 3 2 1 ** * ZL ZDF 6 8 1 12 14 16 Age (weeks) B * Sub-cutaneous (g) 16 12 8 4 ZL ZDF Visceral (g) 25 2 15 1 5 ZL ZDF Total fat pad weight (g) 4 3 2 1 ZDF ZL Supplemental Figure 1: Effect
Body Weight (g) 4 3 2 1 ** * ZL ZDF 6 8 1 12 14 16 Age (weeks) B * Sub-cutaneous (g) 16 12 8 4 ZL ZDF Visceral (g) 25 2 15 1 5 ZL ZDF Total fat pad weight (g) 4 3 2 1 ZDF ZL Supplemental Figure 1: Effect
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTAL MATERIALS AND METHODS. Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of
 SUPPLEMENTAL MATERIALS AND METHODS Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of cysteine and methionine and then treated with 10 μm puromycin in depletion medium
SUPPLEMENTAL MATERIALS AND METHODS Puromycin-synchronized metabolic labelling - Transfected HepG2 cells were depleted of cysteine and methionine and then treated with 10 μm puromycin in depletion medium
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
D CD8 T cell number (x10 6 )
 IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
WISP1 mediates IL-6-dependent proliferation in primary human lung fibroblasts
 WISP mediates IL-6-dependent proliferation in primary human lung fibroblasts Klee S, Lehmann M, Wagner DE, aarsma H, Königshoff M Comprehensive Pneumology Center, Helmholtz Zentrum München, Munich, Germany;
WISP mediates IL-6-dependent proliferation in primary human lung fibroblasts Klee S, Lehmann M, Wagner DE, aarsma H, Königshoff M Comprehensive Pneumology Center, Helmholtz Zentrum München, Munich, Germany;
Page 39 of 44. 8h LTA & AT h PepG & AT h LTA
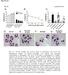 Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Mesenchymal Stem Cells Reshape and Provoke Proliferation of Articular. State Key Laboratory of Bioreactor Engineering, East China University of
 Mesenchymal Stem Cells Reshape and Provoke Proliferation of Articular Chondrocytes by Paracrine Secretion Lei Xu, Yuxi Wu, Zhimiao Xiong, Yan Zhou, Zhaoyang Ye *, Wen-Song Tan * State Key Laboratory of
Mesenchymal Stem Cells Reshape and Provoke Proliferation of Articular Chondrocytes by Paracrine Secretion Lei Xu, Yuxi Wu, Zhimiao Xiong, Yan Zhou, Zhaoyang Ye *, Wen-Song Tan * State Key Laboratory of
Glucose. Glucose. Insulin Action. Introduction to Hormonal Regulation of Fuel Metabolism
 Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL PROLIFERATION IN PANCREATIC CANCER CELLS
 EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL PROLIFERATION IN PANCREATIC CANCER CELLS VIDYA MEDEPALLI ADVISOR: Maria Eugenia Sabbatini, PhD PHI KAPPA PHI RESEARCH CONFERENCE MARCH 18 TH, 2016
EFFECTORS IMPLICATED IN THE AC1 INHIBITORY EFFECT ON CELL PROLIFERATION IN PANCREATIC CANCER CELLS VIDYA MEDEPALLI ADVISOR: Maria Eugenia Sabbatini, PhD PHI KAPPA PHI RESEARCH CONFERENCE MARCH 18 TH, 2016
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
RayBio KinaseSTAR TM Akt Activity Assay Kit
 Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Effect of High-fat or High-glucose Diet on Obesity and Visceral Adipose Tissue in Mice
 ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
ACTA ACADEMIAE MEDICINAE SINICAE 410011 0731-85295846 lixia2014@vip. 163. com 4 C57BL /6 20-1 mrna 20 P < 0. 05 O 20 P > 0. 05 M2 P < 0. 05-1 mrna P < 0. 05 P > 0. 05 R589. 2 DOI 10. 3881 /j. issn. 1000-503X.
NF-κB p65 (Phospho-Thr254)
 Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation
 Chemistry Volume 2015, Article ID 171570, 4 pages http://dx.doi.org/10.1155/2015/171570 Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation Jin Taek Hwang,
Chemistry Volume 2015, Article ID 171570, 4 pages http://dx.doi.org/10.1155/2015/171570 Research Article Inhibitory Effects of 4-(4-Methylbenzamino)benzoate on Adipocyte Differentiation Jin Taek Hwang,
Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line
 Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line Endah Puji Septisetyani, Muthi Ikawati, Barinta Widaryanti and Edy Meiyanto* ) Cancer Chemoprevention Research Center,
Apoptosis Mediated Cytotoxicity of Curcumin Analogues PGV-0 and PGV-1 in WiDr Cell Line Endah Puji Septisetyani, Muthi Ikawati, Barinta Widaryanti and Edy Meiyanto* ) Cancer Chemoprevention Research Center,
Role of cyclooxygenase-2 in H5N1 viral pathogenesis and the potential use of its inhibitors
 Title Role of cyclooxygenase-2 in HN viral pathogenesis and the potential use of its inhibitors Author(s) Lee, MY; Cheung, CY; Peiris, JSM Citation Hong Kong Medical Journal, 2, v. 9 n. Suppl. 4, p. 29-
Title Role of cyclooxygenase-2 in HN viral pathogenesis and the potential use of its inhibitors Author(s) Lee, MY; Cheung, CY; Peiris, JSM Citation Hong Kong Medical Journal, 2, v. 9 n. Suppl. 4, p. 29-
colorimetric sandwich ELISA kit datasheet
 colorimetric sandwich ELISA kit datasheet For the quantitative detection of human IL5 in serum, plasma, cell culture supernatants and urine. general information Catalogue Number Product Name Species cross-reactivity
colorimetric sandwich ELISA kit datasheet For the quantitative detection of human IL5 in serum, plasma, cell culture supernatants and urine. general information Catalogue Number Product Name Species cross-reactivity
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
The rabbit femoral artery was prepared and each arterial ring was permeabilized
 Online Supplement Nakmura et al. cgmp-dependent relaxation of smooth muscle Materials and Methods Measurement of tension The rabbit femoral artery was prepared and each arterial ring was permeabilized
Online Supplement Nakmura et al. cgmp-dependent relaxation of smooth muscle Materials and Methods Measurement of tension The rabbit femoral artery was prepared and each arterial ring was permeabilized
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Acetone and methanol fruit extracts of Terminalia paniculata inhibit HIV-1 infection in vitro Ankita Durge a, Pratiksha Jadaun a, Ashish Wadhwani a, Ashish A. Chinchansure b, Madhukar
SUPPLEMENTARY MATERIAL Acetone and methanol fruit extracts of Terminalia paniculata inhibit HIV-1 infection in vitro Ankita Durge a, Pratiksha Jadaun a, Ashish Wadhwani a, Ashish A. Chinchansure b, Madhukar
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Supporting Information. Supporting Tables. S-Table 1 Primer pairs for RT-PCR. Product size. Gene Primer pairs
 Supporting Information Supporting Tables S-Table 1 Primer pairs for RT-PCR. Gene Primer pairs Product size (bp) FAS F: 5 TCTTGGAAGCGATGGGTA 3 429 R: 5 GGGATGTATCATTCTTGGAC 3 SREBP-1c F: 5 CGCTACCGTTCCTCTATCA
Supporting Information Supporting Tables S-Table 1 Primer pairs for RT-PCR. Gene Primer pairs Product size (bp) FAS F: 5 TCTTGGAAGCGATGGGTA 3 429 R: 5 GGGATGTATCATTCTTGGAC 3 SREBP-1c F: 5 CGCTACCGTTCCTCTATCA
p = formed with HCI-001 p = Relative # of blood vessels that formed with HCI-002 Control Bevacizumab + 17AAG Bevacizumab 17AAG
 A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
A.. Relative # of ECs associated with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 ol b p < 0.001 Relative # of blood vessels that formed with HCI-001 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 l b p = 0.002 Control IHC:
Lecture Outline. Hormones & Chemical Signaling. Communication Basics: Overview. Communication Basics: Methods. Four methods of cell communication
 Lecture Outline Hormones & Chemical Signaling Communication Basics Communication Overview Communication Methods Signal pathways Regulation (modulation) of signal pathways Homeostasis... again Endocrine
Lecture Outline Hormones & Chemical Signaling Communication Basics Communication Overview Communication Methods Signal pathways Regulation (modulation) of signal pathways Homeostasis... again Endocrine
